1. Background
Contrast-induced nephropathy (CIN) as a severe complication of iodine media administration is common after angiography/angioplasty, especially in high-risk patients, including chronic kidney disease (CKD) and diabetic patients (1). The CIN, as the third cause of acute renal failure in the hospital setting, is considered when there is a rise in serum creatinine (SrCr) and blood urea nitrogen (BUN) or a decline in the estimated glomerular filtration rate (eGFR) within 24 - 72 hours after contrast media (CM) exposure (2). The CIN is also associated with increased mortality, hospital stay, and long-term adverse events (3).
Up to 3% of patients experience CIN following angiography/angioplasty according to the Risk, Injury, Failure, Loss, and End-stage renal failure, the Acute Kidney Injury Network (AKIN), and the Kidney Disease Improving Global Outcomes criteria (4, 5). The notable fluctuation of CIN incidence declared in previous studies within the range of 1 - 50% [half of which is among coronary angiography and percutaneous coronary intervention (PCI) cases] (6, 7) is explainable by the lack of a standardized definition of contrast-induced acute kidney injury (CI-AKI) and understanding the meaning of population-based incidence rates of CIN; accordingly, some risk factors, including diabetes, volume depletion, congestive heart failure, CKD, prior CM exposure, CM volume, and nephrotoxic drug history, put the particular population at higher risk of developing CI-AKI; therefore, vulnerable population and general population significantly differ statistically (1, 4, 7-10).
According to the previous studies, AKI is defined by an SrCr absolute rise ranging from 0.3 to 0.5 mg/dL or 25% to 50% for a relative increase (11). However, in recent studies, due to long-term major adverse events, CIN is presumed as an increase in SrCr ≥ 0.3 mg/dL from the baseline values or any slight decrement in renal function within 48 hours of CM administration in the absence of other causes (12-14). The SrCr is not the ideal biomarker for the early detection of AKI. The factors, including body mass index (BMI), measurement techniques, and medications, might influence SrCr concentration (15).
Several biomarkers, including neutrophil gelatinase-associated lipocalin (NGAL), cystatin C, kidney injury molecule-1, and interleukin-18, have been proposed for the early detection of CIN (16). Although no ideal marker is available for the early detection of CIN in patients undergoing angiography/angioplasty, several recent studies showed that NGAL might be a valuable biomarker for the early detection of CIN (17, 18). Overall, the guidelines by the American Society of Radiology defined CIN as one of the following criteria: (1) an absolute SrCr increase of ≥ 0.3 mg/dL; (2) a 50% increase in SrCr; (3) urine output of < 0.5 ml/kg/hour for at least 6 hours within 48 hours after CM administration (19).
Two critical mechanisms mentioned in previous studies are renal ischemia and direct cytotoxicity. The increased production of endothelin and adenosine, along with decreased nitric oxide by endothelial cells, is responsible for renal vasoconstriction (20). The CM administration also increases the release of reactive oxygen species (ROS), resulting in renal tubular direct cytotoxicity, inflammation, and induction of renal cell apoptosis. Taking the crucial role of ROS in CIN pathogenesis into account potentiates the use of free radical scavenger compounds and antioxidants as a promising CIN prevention strategy (20).
Saffron (derived from Crocus sativus L. dried stigmas) is a traditional spice, food coloring, and herbal medicine with a potent antioxidant activity that has extensively been used for different indications. The four main bioactive components of saffron are picrocrocin, crocin, safranal, and crocetin. Crocin, the water-soluble carotenoid, is responsible for the distinct color of saffron (21). Crocin is a potent antioxidant that ameliorates oxidative stress and potentiates the antioxidant defense system, thereby exhibiting several features, including antitumor (22), antihypertensive (23), antidepressant (24), and anti-inflammatory activity (25) plus neuroprotective (26), renoprotective (27), and cardioprotective (28) effects. The rat model investigations of crocin have proven efficacy in chronic stress-induced kidney damage, nephropathy, and renal ischemia/reperfusion damage (29-31). However, the role of crocin in CIN prevention has not yet been investigated.
2. Objectives
This study was designed to evaluate the crocin role in the prevention of CIN in angiography/angioplasty candidates in addition to standard treatment.
3. Methods
3.1. Study Population
The study was designed as an open-label, randomized controlled trial in a single center to evaluate the efficacy and safety of crocin in the prevention of CIN in CKD patients candidate for either diagnostic coronary angiography or PCI. The sample size was calculated based on the pilot study. A total of 115 patients with CKD stage 3 according to the modification diet in renal disease (MDRD) formula were enrolled in this study within February 2019 to July 2020. Permuted block randomization (block size: 4) was used to randomly allocate a participant to an intervention or control group.
The inclusion criteria were the age over 18 years, CKD stage 3 defined as the eGFR between 30-60 mL/min based on the MDRD formula, and systolic blood pressure of greater than 90 mmHg. The patients were excluded from the study due to a history of hypersensitivity to saffron, any contraindication to adequate intravenous hydration therapy, end-stage renal disease on dialysis (the eGFR less than 30 mL/min), acute renal failure, pregnancy or lactation, emergent coronary angiography, cardiogenic shock, platelets less than or equal to 100,000/mm3, left ventricular ejection fraction less than 30%, life-threatening condition, and a history of a kidney transplant.
3.2. Study Protocol
The eligible patients were randomly allocated into either control or intervention group, using a predesigned balanced permuted block randomization method. For the purpose of not depriving individuals of the most effective CIN prevention method, which is hydration therapy, the patients in both groups received standard hydration therapy based on cardiac ejection fraction either with intravenous isotonic normal saline (0.9%) or half saline (0.45%) at a rate of 1 mL/kg/hour 12 hours before up to 12 hours after the procedure. The patients in the intervention group received three consecutive oral doses of 30 mg crocin tablets (each tablet containing 15 mg crocin; Samisaz Co., Mashhad, Iran) 1 day before up to 1 day after the procedure. All physicians, catheterization staff, nurses, and laboratory staff were blinded to the study.
The patients’ demographic and clinical data, including age, gender, weight, BMI, drug history, laboratory data, medical history, and echocardiography findings, were recorded in a predesigned data collection checklist. The interventional procedures were performed according to standard clinical practice using a radial or femoral approach. Due to the lower rate of CIN, the iso-osmolar iodinated contrast agents (iodixanol, 320 mg/mL visipaque®, Marlborough, MA: GE Healthcare) are the preferred CM in Imam Ali hospital, affiliated to Kermanshah University of Medical Sciences. Low-osmolar iodinated contrast agents (e.g., iopamidol 300 or 370 mg/mL and iopromide® 300 ultravist®) were the alternatives.
3.3. Measurements
Three blood samples for each patient were collected at the time of admission, before the coronary angiography, and 48 hours after contrast exposure to measure the levels of BUN and SrCr. The SrCr level was measured by the Jaffe colorimetric method. In addition, for all the allocated patients, the Mehran Risk Score (MRS) was calculated (32). For the determination of the urinary NGAL, two urine samples were collected before and 12 hours after coronary angiography. The first urine sample was collected after 12 hours of hydration. The samples were centrifuged at 1500 × g at 4°C for 15 minutes, frozen in a polypropylene tube, and stored at -20°C for further analysis. The urinary NGAL concentration was determined by the Hycult Biotech® NGAL ELISA kit (Hycult Biotech, Netherlands). The minimum concentration which can be measured was 0.4 ng/mL. All the measurements were performed based on the manufacturer’s instructions.
3.4. Study Endpoints
The AKIN criteria were used for the classification of CIN (Appendix 1) (33). The primary outcome of the study was CIN occurrence, which was defined as an absolute rise in SrCr by more than 0.3 mg/dL within 48 hours of CM exposure in both groups. The secondary endpoints were the value of the urinary NGAL for the prediction of CIN and SrCr level changes. The patients were followed for potential crocin-related side effects during the study.
3.5. Study Analysis
The data were analyzed using SPSS software (version 21). The categorical variables were expressed as numbers and percentages. The Chi-square test and Fisher’s exact test were used to compare these variables between the two groups. The continuous variables were reported as mean ± standard deviation. The Kolmogorov-Smirnov test was used to determine the normality of the distributed variables. Normally distributed continuous variables were compared by the Student t-test, and nonnormally distributed variables were compared by the Mann-Whitney rank-sum test. A p-value less than 0.05 was considered statistically significant.
3.6. Ethical Statement
The current study was directed according to the Helsinki Declaration. This study was approved by the Ethics Committee of Kermanshah University of Medical Sciences, Kermanshah, Iran (ethical approval No.: KUMS.REC.1398.1152) and registered in the Iranian Registry of Clinical Trials (registered code No.: IRCT20200202046335N1). Informed consent was obtained from all the patients before the study.
4. Results
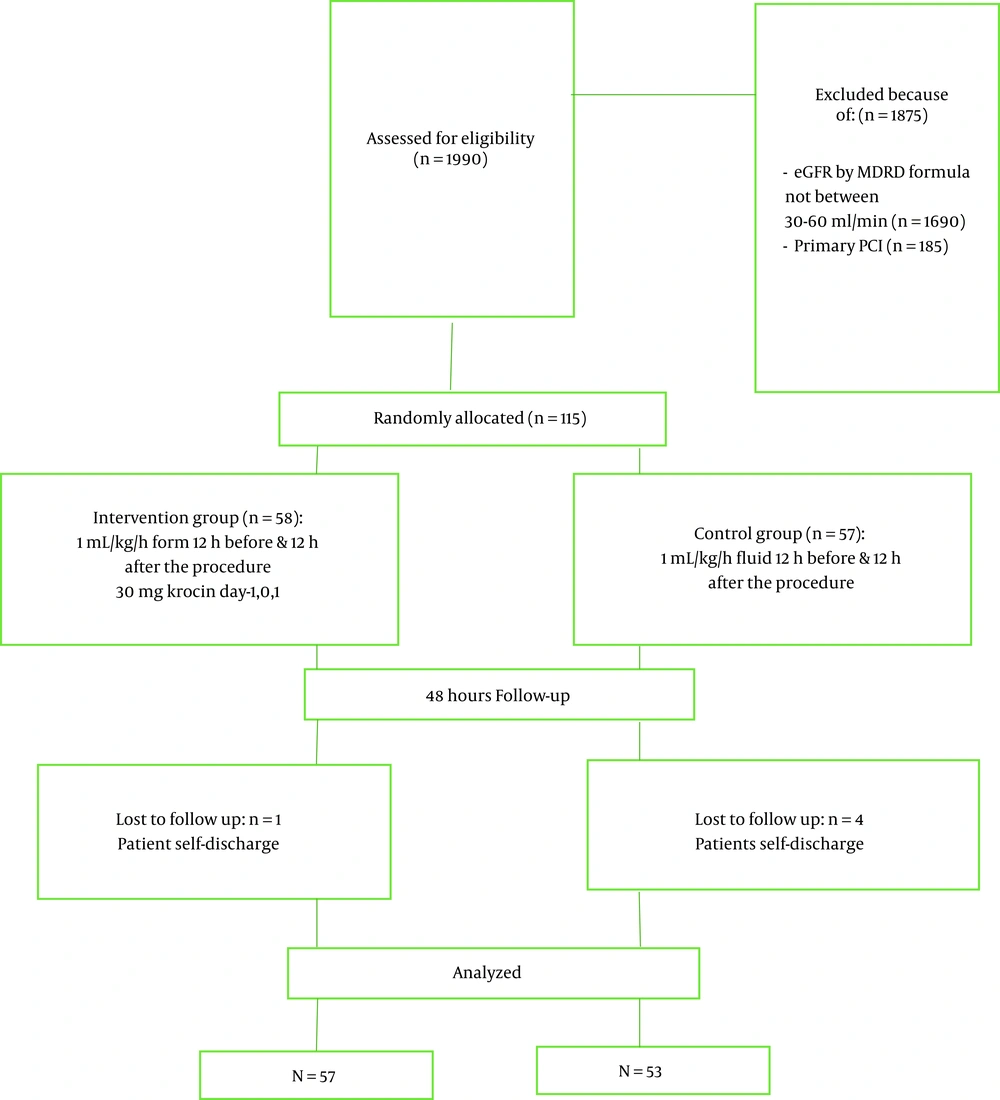
During the study period, 1,990 patients were nominated for angiography/angioplasty (Figure 1). Totally 1,875 patients did not meet the inclusion criteria. Of 115 patients randomly allocated, 5 patients were excluded due to self-discharge after the procedure and missing 24-hour follow-up. Table 1 shows the baseline clinical and laboratory data of the two groups. The mean age of the participants was 65.62 ± 9.05 years, and the majority of them were male (64.54%).
| Characteristics | Intervention Group (n = 57) | Control Group (n = 53) | P-Value |
|---|---|---|---|
| Age (y, mean ± SD) | 65.07 ± 9.01 | 66.21 ± 9.14 | 0.51 a |
| Gender | 0.16 b | ||
| Male | 33 (57.9) | 38 (71.7) | |
| Female | 24 (42.1) | 15 (28.3) | |
| Creatinine (mg/dL) | 1.25 ± 0.16 | 1.26 ± 0.19 | 0.69 c |
| BUN (mg/dL) | 46.16 ± 14.58 | 43.06 ± 11.21 | 0.22 a |
| Weight (kg) | 75.65 ± 14.55 | 75.25 ± 13.73 | 0.96 c |
| Height (cm) | 165.39 ± 9.92 | 164.23 ± 8.82 | 0.52 a |
| Serum NGAL (μg/L) | 23.89 ± 9.55 | 19.75 ± 10.19 | 0.020 c |
| BSA (m2) | 1.86 ± 0.20 | 1.84 ± 0.19 | 0.80 c |
| BMI (kg/m2) | 27.77 ± 5.52 | 27.84 ± 4.64 | 0.94 a |
| eGFR (mL/min) | 47.46 ± 7.14 | 49.70 ± 7.24 | 0.10 a |
| Contrast volume (mL) | 89.04 ± 56.89 | 92.45 ± 58.54 | 0.72 c |
| Mehran risk score | 6.23 ± 2.76 | 5 ± 2.91 | 0.016 c |
| Diabetes | 24 (42.1) | 16 (30.2) | 0.24 b |
| Dyslipidemia | 9 (15.8) | 5 (9.4) | 0.39 b |
| Anemia | 17 (29.8) | 12 (22.6) | 0.52 b |
| Heart failure | 17 (29.8) | 11 (20.75) | 0.27 b |
| Prior myocardial infarction | 8 (14.04) | 6 (11.32) | 0.67 b |
| Prior coronary angiography | 9 (15.79) | 7 (13.21) | 0.68 d |
| Procedure type | 0.56 | ||
| Angiography | 49 (85.96) | 48 (90.57) | |
| Angioplasty | 8 (14.04) | 5 (9.4) | |
| Access type | 0.85 | ||
| Femoral | 33 (57.89) | 29 (54.72) | |
| Radial | 24 (42.10) | 24 (45.28) | |
| Nephrolithiasis | 16 (28.07) | 14 (26.42) | > 0.99 |
| Drug History | |||
| Loop diuretics | 35 (71.4) | 37 (78.7) | 0.48 b |
| ACEI or ARB | 34 (59.6) | 35 (66) | 0.56 b |
| NAC | 2 (3.5) | 3 (5.7) | 0.67 d |
| Sodium bicarbonate | 1 (1.8) | 0 (0) | 1.00 d |
| Statin | 38 (66.7) | 35 (66) | 1.00 b |
| Aspirin | 37 (64.91) | 41 (77.36) | 0.15 b |
| Clopidogrel | 15 (26.32) | 18 (33.96) | 0.38 b |
| Type of contrast agent | 0.56 | ||
| Iodixanol | 51 (89.47) | 46 (86.79) | |
| Iopamidol | 3 (5.26) | 2 (3.77) | |
| Iopromide | 3 (5.26) | 5 (9.4) | |
| History of smoking | 10 (17.54) | 14 (26.42) | 0.26 b |
| Opium addiction | 5 (8.77) | 8 (15.09) | 0.30 b |
Abbreviations: ACEI, angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blockers; BMI, body mass index; BUN, blood urea nitrogen; BSA, body surface area; eGFR, estimated glomerular filtration rate; NAC, N-acetyl cysteine; NGAL, neutrophil gelatinase-associated lipocalin; SD, standard deviation.
a T-test.
b Chi-square test.
c Mann-Whitney U test.
d Fisher’s exact test.
All the baseline characteristics were similar between the two groups except the MRS and urinary NGAL. The SrCr, BUN, and NGAL were measured multiple times. No significant differences were observed in SrCr and BUN levels between the two groups before angiography/angioplasty. All the recruited patients had CKD stage 3; therefore, the eGFR was similar in both groups. The mean volume of contrast agents and infused fluid were similar between the control (P = 0.72) and intervention (P = 0.99) groups. During the study, SrCr decreased significantly in the intervention group (-0.04 ± 0.14; P = 0.012) but not in the control group (0.08 ± 0.18; P = 0.008) (Table 2).
| Parameters and Groups | Before the Study | After the Study | P-Value a |
|---|---|---|---|
| Creatinine (mg/dL) | |||
| Intervention group | 1.25 ± 0.16 | 1.20 ± 0.19 | 0.012 |
| Control group | 1.25 ± 0.19 | 1.33 ± 0.24 | 0.003 |
| eGFR (mL/min) | |||
| Intervention group | 47.46 ± 7.14 | 59.30 ± 14.25 | < 0.001 |
| Control group | 49.70 ± 7.24 | 55.32 ± 17.3 | 0.011 |
| NGAL | |||
| Intervention group | 23.89 ± 9.55 | 26.47 ± 8.44 | 0.18 |
| Control group | 19.75 ± 10.19 | 23.79 ± 6.33 | 0.56 |
Abbreviations: eGFR, estimated glomerular filtration rate; NGAL, neutrophil gelatinase-associated lipocalin; SD, standard deviation.
a Wilcoxon signed-rank test.
The SrCr measured 48 hours after the administration of contrast agents was significantly higher in the control group than in the intervention group (P = 0.001; Table 3). Therefore, the eGFR was higher in the intervention group at the end of the study than in the control group (P = 0.009; Table 3). The measured urinary NGAL 12 hours after the angiography/angioplasty was significantly higher in the crocin group than in the control group but not clinically significant (P = 0.006; Table 3).
Abbreviations: AKI, acute kidney injury; eGFR, estimated glomerular filtration rate; NGAL, neutrophil gelatinase-associated lipocalin; SD, standard deviation.
a Mann-Whitney U test.
b Fisher’s exact test.
Although the MRS was significantly lower in the control group than in the intervention group (P = 0.016), CIN was lower in the intervention group (1.75% and 13.21%; P = 0.028; Table 3). In the crocin group, there was a significantly lower rate of CIN (relative risk: 13%; 95% CI: 1.01 - 1.26). No patients experienced adverse effects during the study.
5. Discussion
The incidence of CIN among the general population undergoing angiography/angioplasty is about 1 - 2% in the general population and might increase up to 50% for high-risk subgroups (34), which is in concordance with the findings of the current study. In the present study, the incidence of CIN was 7.3% in the total population. The population of this study was selected from patients with CKD stage 3; as a result, they were at higher risk of CIN than the normal population. Although the MRS in the intervention group was significantly higher than in the control group, CIN incidence in the crocin group was significantly lower. The analysis of the results revealed a significant AKI risk reduction in patients receiving contrast agents by crocin administration.
To the best of our knowledge, this study has been the first randomized clinical trial that evaluated the protective effects of crocin on CIN. Although recent studies have attributed renoprotective features to crocin, no clinical trial has been conducted on the effect of crocin on CIN prevention in CKD patients undergoing coronary angiography/angioplasty. Crocin is a potent antioxidant and ROS scavenger with renal protection properties due to renal ischemia amelioration (35), oxidative stress attenuation (27), lipid peroxidation prevention (35), anti-apoptotic effect (36), and endothelin reduction (37). It also has vasodilatory and Antioxidant properties (37). Ascorbic acid, N-acetyl cysteine, statins, sodium bicarbonates, nicorandil, alpha-tocopherol, alprostadil, allopurinol, adenosine antagonists, sarpogrelate, silymarin, isosorbide dinitrate, pentoxifylline, L-arginine, beta-blockers, and theophylline have been used to reduce CIN by other researchers (38).
The results of the current study showed that crocin could prevent CM-associated nephropathy. Although this reduction was statistically significant but not clinically, this study could prevent the SrCr rise in the crocin group. Since hydration remains the mainstay of treatment and the standard by which other CIN prevention strategies are judged, both groups received adequate hydration therapy (19). Consequently, in both groups, the eGFR was improved statistically but not clinically. Other factors, including contrast agents, contrast volume, and N-acetylcysteine consumption, were similar between the two groups.
Inflammation, ethnicity, gender, hydration, age, BMI, renal function, chronic illness, detection methods, and medications might influence SrCr concentration (39). During the last decades, the biomarkers, including NGAL, cystatin C, β2 microglobulin, α-1 microglobulin, and urinary insulin-like growth factor binding protein 7, have been proposed for the early detection of AKI. For instance, urinary NGAL is increased within 2 hours after CM administration and can predict the occurrence of CIN approximately 24 hours before SrCr increase (40). However, in the present study, no significant difference in urinary NGAL concentration was observed between the two groups. It should be noted that an increase in NGAL concentration is associated with a poor prognosis following CM administration (41). No patient in the current study needed renal replacement therapy. Only patients with CKD stage 3 were included in this study. Accordingly, patients with stage 3 had a higher risk of AKI, and crocin pretreatment significantly decreased CIN.
5.1. Limitations
The patients were discharged within 24 hours after angiography leading to not having more samples (e.g., 6, 24, and 48 hours after CM administration) to calculate the kinetics of urinary NGAL.
5.2. Conclusion
Crocin pretreatment is associated with a lower rate of CIN. Furthermore, studies with a larger sample size are needed to confirm the results of the present study. Based on the literature, there is controversy concerning whether SrCr could be a better predictor of CIN than NGAL as a new biomarker. In this study, urinary NGAL could not predict and diagnose AKI. One possibility to explain NGAL inefficiency in this study was the probable degradation and reduction of NGAL concentration in the urine samples due to long-term (6 months) storage at -24°C and freeze-thawing cycles. The NGAL in urine samples is stable at 4°C for up to 24 hours and -80°C for up to 5 years; however, it was not possible to provide -80°C. Moreover, the small sample size and missing some urine samples in the collection process might account for NGAl’s incompetence with the literature confirming its predictor role in CIN. Another reason that NGAL was not a good predictor of AKI in our study was that urine samples were collected 12 hours before and after procedure. However, measured NGAL 24 hours after contrast exposure could be a more promising biomarker for predicting CIN.
-thumbnail.webp)