1. Context
Eradicating Helicobacter pylori has been recommended to treat different upper gastrointestinal disorders. Although combination therapy is suggested for H. pylori eradication, using multiple drugs can decrease patients' adherence to pharmacotherapy regimens and may increase the risk of adverse drug reactions (ADRs) (1). Moreover, failure to respond to antibiotics is another concern, and choosing the most efficacious treatment for infected patients has become increasingly important (2).
The popularity of using complementary therapies/dietary supplements to treat gastrointestinal disorders is on the rise. Most studies regarding the effect of complementary therapies in gastrointestinal disorders have been done in vitro (3). The present review article aimed to provide information regarding the use of dietary supplements for eradicating H. Pylori, particularly in humans.
2. Evidence Acquisition
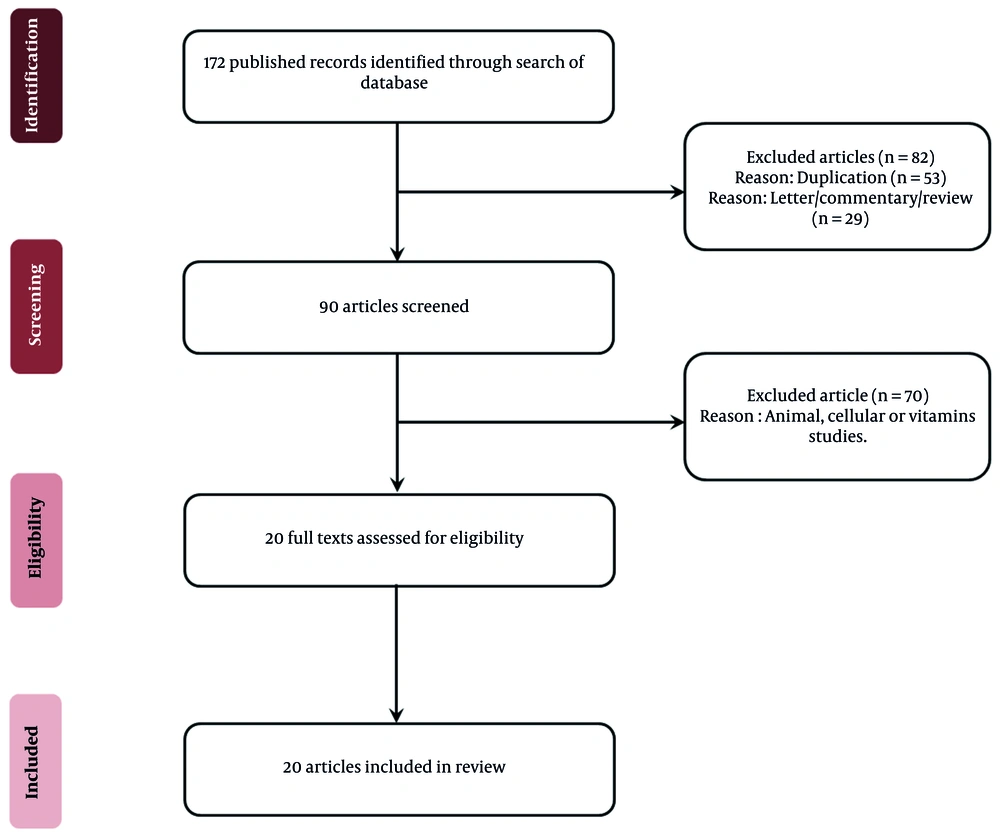
A search for human studies published before June 2021 was performed in the SCOPUS, Medline/PubMed, and EMBASE databases. All the authors participated in the search process. The authors searched for the terms “Helicobacter pylori" with any of the following: "Dietary supplements," "complementary," and "herbal." We focused on the clinical studies published in English (full text or abstract). Data analysis was done by two specialists (a gastroenterologist and a clinical pharmacist) who assessed the title and summary of the search results separately to omit duplicates and case reports. The inclusion criterion was following a human clinical study design to investigate the effect of any herbal medicine or dietary supplement on H. pylori eradication. Results of the studies were not the point of selection, and all clinical studies that evaluated the outcome of any complementary or dietary supplement against H. pylori eradication were included. Finally, a total of 20 eligible articles were retrieved. The flow of the search is shown in Figure 1.
3. Results and Discussion
3.1. Green and Black Tea (Camellia sinensis)
An animal study carried out on Mongolian gerbils by administering green tea catechins solution adsorbed to sucralfate reported that the colony count of H. pylori was significantly reduced (4). Another study compared a plate of H. pylori strains and green tea-embedded discs or control. After incubation, green tea-embedded showed the growth inhibition of H. pylori; moreover, the study showed that green tea administration could prevent gastric mucosal inflammation in animal models (5). There is no published clinical trial regarding the effect of green/black tea on eradicating H. pylori in the medical literature. Boyanova et al. (6) evaluated the impact of dietary habits on the H. pylori infection rate in 150 patients diagnosed with dyspepsia. They examined all the participants endoscopically and with the urea breath test (UBT). The infection rate was less (45.2%) in patients consuming green/black tea one day weekly than in other patients (64.8%). Logistic regression confirmed that green/black tea was correlated with a remarkably lower H. pylori positivity rate (OR, 0.45; 95% CI, 0.21 - 0.95).
3.2. Black Cumin (Nigella sativa)
Pharmacological studies, such as Ali and Blunden (7), suggested different properties of Nigella sativa to have anti-inflammatory, antioxidative, and antimicrobial effects. Several studies have mentioned that anti-H. pylori effects may be related to numerous ingredients such as thymoquinone, thymol, nigellidine, and carvacrol. The specific environment of the stomach causes the growth of H. pylori, and suppressing stomach acid and changing its pH cause H. pylori to leave the antrum and make it easier to be eradicated. Nigella sativa expresses a potent anti-secretory effect (7-9).
There are two published articles about N. sativa administration against H. pylori in humans. Salem et al. (10) evaluated the effect of N. sativa in suppressing H. pylori in patients diagnosed with Non-Ulcer Dyspepsia (NUD). The study was carried out on 88 patients diagnosed with H. pylori using histopathology and the rapid urease test (RUT). The patients were randomly distributed into four groups, as follows: group 1: triple therapy comprising clarithromycin, amoxicillin, and omeprazole; group 2: 1 g N. sativa plus omeprazole (40 mg/d); group 3: 2 g N. sativa plus omeprazole (40 mg/d); and group 4: 3 g N. sativa plus omeprazole (40 mg/d). Eradication rates were evaluated using the fecal antigen test after four weeks. They reported that H. pylori elimination was 82.6, 47.6, 66.7, and 47.8% in the groups 1 to 4, respectively. Elimination rates with 2g N. sativa and triple therapy were equal statistically, whereas H. pylori elimination with other doses was remarkably less than that with triple therapy (P< 0.05). Interestingly, the H. pylori elimination rate was more with N. sativa 2 g/d (66.7%) than with N. sativa 3 g/d (47.8%). In the same vein, an in vitro study described a similar consequence, where a lower concentration of N. sativa extract exerted more antibacterial effect than a higher concentration of it (11). Symptoms of dyspeptic patients were alleviated in all the groups to a similar extent. They reported that a combination of N. sativa plus omeprazole possessed a clinically useful antibacterial effect, comparable to triple pharmacotherapy. In another randomized human study, Alizadeh-Naini et al. (12) determined the effects of adding N. sativa to bismuth-based quadruple therapy on H. pylori elimination, dyspeptic symptoms, biochemical markers, and quality of life (QoL) in patients infected with H. pylori. Fifty-one patients with a diagnosis of NUD were randomized into treatment (bismuth-based quadruple therapy plus 2 g/day N. sativa) or placebo (bismuth-based quadruple therapy with 2 g/day placebo) arm for two months. Concentrations of interleukin-8, high-sensitivity C-reactive protein (hs-CRP), malondialdehyde, and QoL were evaluated before and after the trial. Finally, the H. pylori elimination rate was more in the N. sativa arm against the placebo (P = 0.01). Also, the QoL score was more significant in the treatment arm versus the placebo arm (P < 0.05). Biochemical markers and dyspeptic symptoms were not different between the two arms. They concluded that the addition of N. sativa may have beneficial effects on H. pylori eradication and QoL.
3.3. Honey
Boyanova et al. (6) assessed the effect of honey consumption on the H. pylori infection rate in 150 patients who suffered from dyspepsia. The patients were evaluated using endoscopy and UBT. They mentioned that the infection rate was lower (50.6%) in those consuming honey more than one day per week than in others (70.8%). They concluded that honey intake was related to a lower H. pylori infection rate. Most of the published articles focused on evaluating the effect of honey against H. pylori in vitro. In a systematic review on the in vitro effect of honey against H. pylori, Quraisiah et al. (13) searched databases such as Medline via Ovid Medline, Scopus, and ScienceDirect to identify relevant articles published from 2000 to 2018. Associated published data were assessed and chosen based on the criteria on the effects of honey on ulcers in the stomach or duodenum caused by H. pylori existence. A total of 53 articles were selected finally. All the articles showed the positive role of honey against ulcers induced by H. pylori. Most of the articles reported that a minimum of 10% honey concentration was effective against H. pylori. They mentioned the need for future in vitro studies to find the active component and exact mechanism of honey before clinical trials could be done to deliver valid evidence. There is no published article about the net effect of honey on H. pylori elimination. Hashem-Dabaghian et al. (14) assessed the effect of a combination of N. sativa and honey in eliminating H. pylori infections of the stomach. Nineteen participants were requested to intake one teaspoon of the mixture (6 g/day of N. sativa as ground seeds and 12 g/day of honey) three times a day after meals for 14 days. Helicobacter pylori elimination was determined using UBT at the baseline and after the study. The duration of the study was four weeks. Dyspeptic symptoms were determined at the baseline and after the study and compared. Finally, fourteen participants finished the study. Negative UBT was determined in 57.1% of the patients after providing the intervention. The median and interquartile range (IQR) of total dyspepsia symptoms were remarkably reduced from 5.5 to 1 (P = 0.005). All the participants tolerated the N. sativa plus honey mixture well, except for one omitted from the study due to mild diarrhea.
3.4. Cranberry (Vaccinium macrocarpon)
In vitro studies suggested that cranberry could inhibit H. pylori adhesion to the mucosa and reduce colonization of H. pylori (15, 16). In a randomized, double-blinded clinical trial, Gotteland et al. (17) evaluated the effect of the regular intake of a mixture of cranberry juice and the Lactobacillus johnsonii La1 to eradicate H. pylori in infants. Eligible infants, who were H. pylori positive, according to UBT, were included in the trial. The infants were divided into four arms: Cranberry juice plus La1 (CB/La1 arm), placebo juice plus La1 (La1 arm), cranberry juice plus heat-killed La1 (CB arm), and placebo juice plus heat-killed La1 (control arm). Cranberry juice (200 mL) and La1 product (80 mL) were administered daily for three weeks, after which a second UBT was done. A third UBT was carried out after 30 days of washout in infants who were negative in the second UBT. Finally, 271 participants completed the treatment course. A different rate of eradication has been shown in four arms: 1.5% in the control arm versus 14.9%, 16.9%, and 22.9% in the La1, CB, and CB/La1 arms, sequentially (P < 0.01); the latter group showed a slight but not significant increase when compared with the other arms. The third UBT was done only in 19 infants who were negative in the second UBT, and H. pylori existed in 80% of them. They concluded that the regular intake of cranberry juice or La1 may be useful in treating asymptomatic infants infected with H. pylori; however, no additive inhibitory property on H. pylori eradication was observed when both agents were simultaneously consumed. Li et al. (18) conducted a human study to determine the dosage effect of daily cranberry intake on H. pylori elimination over time in infected persons. A randomized, placebo-controlled blinded trial was conducted on 522 participants infected with H. pylori. The study assessed the dose-response effects of proanthocyanidin-standardized cranberry juice, cranberry powder, or identical placebos on eliminating H. pylori at two and eight weeks using UBT at 45 days post-intervention. Helicobacter pylori-negative rates in placebo, low-proanthocyanidin, medium-proanthocyanidin, and high-proanthocyanidin cranberry juice arms were 13.24%, 7.58%, 1.49%, and 13.85% after 14 days and 7.35%, 7.58%, 4.48%, and 20.00% after eight weeks, sequentially. The intake of high-proanthocyanidin juice twice a day for eight weeks resulted in reduced H. pylori infection rate by 20% compared with other dosages and the placebo (P < 0.05). The percentage of H. pylori-negative patients elevated from two to eight weeks in subjects who consumed 44 mg proanthocyanidin/day juice once or twice daily, showing a statistically notable positive trend over time. In addition, encapsulated cranberry powder doses were not significantly successful at either time. The participants tolerated cranberry juice or powder well in the mentioned trial. The authors concluded that the consumption of proanthocyanidin-standardized cranberry juice twice a day may effectively contribute to H. pylori elimination.
3.5. Mastic Gum (Pistacia lentiscus)
Before discovering H. pylori, some studies intended to show the effects of mastic gum on the healing of gastrointestinal diseases such as Crohn's disease (19). There are only some hints for reducing colonization due to an acidic fraction of this substance. There are two contrasting ideas for mastic gum efficacy against H. pylori: (1) ideas stating that positive effects of mastic gum can reduce H. pylori colonization, and (2) ideas expressing that anti-H. pylori potency is related to its acidic fractions.
Dabos et al. (20) designed a study in which participants were randomly divided into four arms to intake 350 mg of pure mastic gum three times a day (A), 1.05 g of pure mastic gum three times a day (B), 20 mg of pantoprazole twice a day plus 350 mg of pure mastic gum three times a day (C), or a triple therapy consisting of pantoprazole, amoxicillin, and clarithromycin (D). For all the patients, confirmation of H. pylori was performed using UBT. The duration of the regimens was 14 days in the A, B, and C arms and 10 days in the D arm. Five weeks later, H. pylori eradication was tested using a repeated UBT. The eradication of H. pylori was confirmed in 30% and 38% of the A and B arms, respectively. No patient in the C arm achieved eradication, whereas 77% of patients in the D arm had a negative UBT. There were no statistically significant differences concerning the mean UBT values in A, B, and C arms, although there was a trend in A (P = 0.08) and B (P = 0.064). Also, the difference was eminent in the D arm (P = 0.01). On the whole, mastic gum was tolerated well, and there was no dropout during the trial.
3.6. Olive Oil (Olea europaea)
Castro et al. (21) designed and finalized two different pilot human studies to investigate the effect of virgin olive oil on the H. pylori elimination rate. In the first trial, 30 volunteers infected with H. pylori agreed to intake 30g of washed virgin olive oil for 14 days, and after 30 days, they took 30 g of unwashed virgin olive oil for another 14 days. In the second trial, 30 infected participants received 30 g of a different virgin olive oil for two weeks. In both trials, H. pylori infection was assessed using UBT. In the first trial, 27% and 40% of the volunteers became negative by intention to treat per protocol, respectively, while for the second trial, these values were 10% and 11%, sequentially. Thirteen subjects did not tolerate virgin olive oil because of taste and nausea drawbacks, and they dropped out the trial. Castro et al. concluded that the intake of virgin olive oil had little efficacy against H. pylori, although further large trials are needed to confirm these preliminary assumptions.
3.7. Licorice (Glycyrrhiza glabra)
Licorice potent has antioxidant, anti-cancer, anti-inflammatory, secretin-secreting, and anti-adhesive properties that can be used against H. pylori. It can also inhibit DNA gyrase28. An in vitro study showed the anti-H. pylori effect of Licorice (22).
Momeni et al. (22) evaluated the effect of licorice on 60 positive H. pylori patients diagnosed with peptic ulcer disease in a randomized clinical trial. Those in the treatment arm received a combination of omeprazole, amoxicillin, metronidazole, and licorice, and those in the control arm received a combination of omeprazole, bismuth subsalicylate, amoxicillin, and metronidazole, which were prescribed for two weeks in each group. Six weeks later, UBT was done on all the patients. The response rate to medical therapy was almost similar (67% in the treatment arm and 57% in the control arm) (P > 0.05). They concluded that licorice may be effective as bismuth in eliminating H. pylori; therefore, licorice can be substituted with bismuth safely in a quadruple therapy. In a similar small trial by Decker, individuals with peptic ulcer disease positive for H. pylori infection (diagnosed by endoscopy and biopsy with positive rapid urease test) were treated with quadruple therapy or quadruple therapy plus licorice as a substitution for bismuth subsalicylate. After four weeks of medical treatment, eradicating H. pylori infection was comparable in both groups (23). Following a clinical trial design, Hajiaghamohammadi et al. (24) investigated 120 patients with positive rapid urease tests who were allocated to receive a triple therapy consisting of clarithromycin (control arm) or licorice in addition to the same triple therapy (treatment arm) for two weeks. The eradication rate of H. pylori was determined six weeks after ending pharmacotherapy. Response to treatment was 83.3% and 62.5% in the study and control arms, respectively, and the difference was considerable. They concluded that the addition of licorice to the triple therapy consisting of clarithromycin increased H. pylori elimination.
3.8. Garlic (Allium sativum)
Several studies evaluated the effect of garlic against H. pylori in vitro (25, 26). Few clinical trials investigated the effect of garlic on eradicating H. pylori. McNulty et al. (27) evaluated the effect of garlic oil on the treatment of patients diagnosed with dyspepsia who were infected with H. pylori as a pilot study. The patients were asked to intake a capsule consisting of 4 mg garlic oil four times per day for two weeks. Five dyspeptic patients with positive antibodies against H. pylori were confirmed using UBT at the baseline and two weeks later completed the study. If there was a negative UBT at both follow-up evaluations, H. pylori elimination would be successful. UBT was used for eradication follow-up. No document of either eradication of H. pylori or symptom relief was observed in this trial. They postulated that garlic oil at this dose did not eliminate H. pylori. They mentioned that a higher dose of garlic for a longer duration might be effective.
3.9. Cinnamon Extract (Cinnamomum verum)
In vitro studies reported the efficacy of cinnamon extract on H. pylori growth. This inhibitory effect is due to various compounds such as eugenol, carvacrol, cinnamaldehyde, and anti-inflammatory/antimicrobial activity (28).
In a pilot study, Nir et al. (29) investigated the efficacy of the cinnamon extract in 15 participants infected with H. pylori who received either 40 mg of cinnamon extract twice a day or a placebo for 28 days. The colonization rate of H. pylori was measured using UBT before and after the intervention. Cinnamon extract was tolerated well, and there was a minimal adverse reaction. At the second UBT, eight patients showed a decrease in the colony count of H. pylori, while seven subjects showed an increase in the colony count. In the cinnamon group, the readings in the second UBT increased over the first UBT. The baseline readings were somewhat higher in the control group than in the other groups, although not significantly. In another randomized clinical trial, Imani et al. (30) evaluated the effect of cinnamon extract on eradicating H. pylori. They enrolled 98 healthy volunteers and H. pylori-infected patients approved by the endoscopic procedure in the trial. The patients received multiple antibiotic regimens plus cinnamon extract capsules or multiple antibiotic regimens plus a placebo. UBT was done three months after the initiation of the treatment. A notable difference was shown in the H. pylori eradication rate, where the rate was 73.47% and 53.06% in the cinnamon and placebo arms, respectively (P = 0.036). They concluded that the combined use of cinnamon and antibiotic regimens could increase the rate of H. pylori eradication.
3.10. Propolis
It has been postulated that an active phenolic compound of propolis is related to the inhibition of peptide deformylase, which is an essential enzyme for bacterial growth (31). In contrast to various reports regarding the effect of propolis against H. pylori in vitro, few clinical trials have investigated this issue. A pilot clinical study on 18 volunteers by Coelho et al. (32) determined that propolis had no effects on the eradication rate. Helicobacter pylori infection was confirmed in the participants using histology and UBT. The patients were asked to intake 20 drops from Brazilian green propolis elixir three times a day for one week. Clinical assessment and UBT were carried out 40 days later to evaluate the H. pylori eradication rate. According to the findings, 83% of the volunteers did not succeed in suppressing or eliminating H. pylori.
3.11. Lycopene
Helicobacter pylori increase the cleavage of Poly [ADP-ribose] polymerase 1, an enzyme that plays a crucial role in DNA recovery. Therefore, H. pylori induce apoptosis in gastric epithelial cells. Jang et al. (33) claimed that lycopene inhibited reactive oxygen species and that it could cause modification in the cell cycle of gastric epithelial cells. As a result, lycopene has the potential to treat H. pylori-induced gastric diseases.
In a quasi-control trial designed by Shidfar et al. (34), a group of patients received a standard quadruple regimen. The other group received a similar quadruple regimen plus lycopene (30 mg/daily). Thirty days after the intervention, the patients were examined for H. pylori elimination using RUT. Although the elimination rate was more in the lycopene group, the bivariate analysis statistically revealed no difference between the two arms.
3.12. Melatonin
A study demonstrated the antioxidative and immunomodulatory activities of melatonin against H. pylori. The mRNA expression levels of arylalkylamine-n-acetyl transferase and acetyl serotonin methyl transferase were assayed in the mucus of the stomach (35). Forkhead box P3 (Foxp3) and transforming growth factor-beta1 (TGF-β1) are immuno-regulators, which are essential targets in anti-H. pylori therapy. Melatonin interferes in inflammatory responses by inhibiting the expression of Foxp3 and TGF-β1 (36).
There are two published clinical trials about the impact of melatonin in eliminating H. Pylori in the medical literature. In a study conducted by Celinski et al. (37), three groups of participants, each including infected patients with gastric or duodenal ulcers, were randomly allocated to intake omeprazole 20 mg twice daily plus placebo (group A), melatonin 10 mg/d (group B), or tryptophan 500 mg/d (group C). All the patients underwent routine endoscopy at the baseline, in which the stomach biopsy was taken for the existence of H. pylori. Ulcer alleviation was evaluated using endoscopy at 7, 14, and 21 days after initiating the medications. On day 21, all ulcers were alleviated in participants of the groups B and C, but only 42% of them were cleared in the group A. They concluded that melatonin or tryptophan added to omeprazole could remarkably accelerate the alleviation rate of H. pylori-infected chronic peptic ulcers compared to the sole administration of omeprazole. Abdi et al. (38) designed a randomized clinical trial to determine the effect of melatonin addition on the H. pylori eradication rate. The trial contained a 14-day quadruple eradication regimen (omeprazole, bismuth subsalicylate, amoxicillin, and metronidazole) supplemented with melatonin 3 mg daily or a comparable placebo. Helicobacter pylori elimination rates were 73% in the melatonin arm and 65% in the placebo using intention-to-treat analysis (ITT; n = 118). Elimination rates of per-protocol analysis (PP; n = 98) were 80% and 79% in the melatonin and placebo arms, respectively. The statistical survey did not show any critical difference between the two groups using either ITT or PP analysis (P = 0.74 and P = 0.91, respectively). They concluded that melatonin 3 mg daily did not have any synergistic impact on the H. pylori elimination rate.
3.13. Nickel-Free Diet
Nickel is a chemical element that plays a vital role in the existence of H. pylori. Nickel activates the urease enzyme, which is essential for metabolism, virulence, and colonization of H. pylori. H. pylori NikR (HpNikR) is a nickel-responsive transcription factor that regulates urease expression (39). A hypothesis claims that nickel free diet (NFD) can be effective against H. pylori infection. It is a type of diet in which foods with high nickel, such as apricots, figs, pears, plums, and asparagus, should be avoided (40).
Campanale et al. (41) compared the H. pylori eradication rate of an NFD associated with standard triple therapy and standard triple therapy consisting of lansoprazole, clarithromycin, and amoxicillin (LAC) in 52 patients diagnosed with H. pylori infection. The patients followed 30 days of an NFD plus a week of LAC regimen starting from day 15 of the diet. A remarkable higher elimination rate was observed in the NFD plus LCA arm (22/26) versus the LCA alone arm (12/26) (P < 0.01). Regarding adverse drug reactions, all regimens were tolerated well, and there was no dropout in either the NFD plus the LCA arm or the LCA arm. Further clinical trials are necessary to confirm this preliminary observation.
3.14. Polyunsaturated Fatty Acids
In vitro studies showed the efficacy of polyunsaturated fatty acids (PUFA) against H. pylori’s growth and treatment of duodenal ulcer disease. PUFAs prevent gastrointestinal cancer due to their anti-inflammatory and rejuvenating effects against H. pylori (42). It has been shown that PUFAs inhibit Interleukin-8, mRNA, and protein expression in cells infected with H. pylori (43).
Few clinical studies investigated the efficacy of PUFA against H. pylori. In a human study, 40 patients diagnosed with duodenal ulcers and infected with H. pylori received H2 blockers. They were randomized to receive either polyunsaturated fatty acids (PUFA arm) or an identical placebo (control arm). The efficacy of drug regimens is evaluated endoscopically by examining ulcer healing, while the H. pylori status was assessed by taking a biopsy from the antrum via RUT and histology. The concentration of prostaglandin E2 (PGE2) and leukotriene B4 (LTB4) was quantified in the antrum tissue. Despite a remarkable difference in linoleic acid intake (19.9 ± 1.6 g) for the PUFA arm versus controls (6.7 ± 0.8 g) (P < 0.01) and linolenic acid (2.6 ± 0.2 g in the PUFA arm versus 0.6 ± 0.03 g in the control arm) (P < 0.01), there was no remarkable change in either the severity of H. pylori elimination or prostaglandin concentrations in either arm after six weeks. An appreciable amount of PUFA does not prevent the stomach colonization of H. pylori, nor does it alter the inflammatory features of H. pylori gastritis (44). In another clinical trial on H. pylori-positive patients (diagnosed with histology and RUT) with mild functional dyspepsia, Frieri et al. (45) evaluated the effect of dietary PUFA supplementation on H. pylori. They asked patients to consume two grams of a dietary mixture of fish oil and black currant seed oil daily for eight weeks. The eradication rate of H. pylori was determined at the end of the eight weeks. Eight (out of 15) patients (53%) were negative for H. pylori after the study period.
4. Conclusions
Table 1 presents the summary of 20 eligible studies regarding the role of supplementary medicine in H. pylori eradication. There is a great deal of interest in complementary and herbal medicines globally. However, there is no consistent evidence to support their efficacy in eradicating H. pylori. More high-quality randomized clinical trials are needed in general or for each complementary intervention or usage in the H. pylori eradication regimens. We believe that the findings provide an additional benchmark for further trials regarding the effect of dietary medicine on H. pylori eradication.
| Active Component/ Dose/ Duration | First Author (Year/ Country) (Reference Number) | Putative Anti- Helicobacter pylori Properties | Type of Studies | Number of Patients | Intervention Group | Concurrent Antibiotics Eradication Regimen | Effect |
|---|---|---|---|---|---|---|---|
| Honey and green black tea/ Dose: N/A/ One day weekly | Boyanova et al. (2015/ Bulgaria) (6) | Polyphenolic for inhibition of the growth of H. pylori for honey | Retrospective study | 150 | untreated dyspeptic patients | No | Lower H. pylori positivity in patients |
| Nigella sativa/ powder A) 1 g as a capsule of 500mg; B) 2 g as capsule 500mg; C) 3 g as capsule 500mg/ Four weeks | Salem et al. (2010/ Saudi Arabia) (10) | Anti-inflammatory/ Anticancer/ Antimicrobial activity/Inhibition of the growth of H. pylori | Randomized, open, clinical trial | 88 | Nonulcer dyspeptic patients | Yes | Increased eradication rate/ Two g of N. sativa has the same effectiveness as triple therapy/ One g, and 3g are less than that |
| N. sativa/ 2 g daily/ Eight weeks | Alizadeh-Naini et al. (2020/ Iran) (12) | Antioxidative/Anti-inflammatory effect of thymoquinone on NFκB | Randomized, double-blind, placebo-controlled, clinical trial | 51 | Patients with functional dyspepsia with positive H. pylori | Yes | Significantly increasing the eradication rate/ A significant improvement in patients' quality of life/No differences in biochemical markers and dyspepsia between the two groups |
| The mixture of N. sativa and honey/ a teaspoon (N. sativa 6 g/day; honey 12 g/day)/ three times a day/ Two weeks | Hashem-Dabaghian et al. (2016/Iran) (14) | Nigella sativa oil: Antibacterial activity of the phenolic fractions and the urease enzyme inhibition potency of Iranian N. sativa; Honey: Anti-H. pylori activity | Pilot clinical trial | 19 | Positive H. pylori (UBT) without a history of peptic ulcer, gastric cancer, or gastrointestinal bleeding | No | Helicobacter pylori eradication in 57% of infected patients/ Significantly reduced score of dyspepsia symptoms |
| Cranberry juice/ 200 mL and Lactobacillus acidophilus product 80 mL/ Three weeks | Gotteland et al. (2008/ Chile) (17) | Probiotics have been proposed to produce organic acid and bacteriocins capable of inhibiting H. pylori growth / Cranberries have inhibiting activity against H. pylori because it has proanthocyanidins, and cranberry contains polyphenols that are antioxidant and anti-inflammatory | Multicentric, randomized, double-blind, placebo-controlled, clinical trial | 295 | UBT-positive children | No | No synergism effects observed when simultaneously administering the cranberry juice and Lactobacillus /Temporary H. pylori inhibition by consuming cranberry juice or probiotic/A regular intake of cranberry juice and La1 may be useful in colonizing a symptomatic population |
| Cranberry juice or juice-based powder/ dosage (44 mg/240-mL serving) / 2 - 8 weeks | Li et al. (2021/ China) (18) | Proanthocyanidin for suppression of H. pylori infection | Randomized, double-blind, placebo-controlled, clinical trial | 522 | Helicobacter pylori -positive adults | Yes | Decreased viability of GC cell lines/Reduced esophageal adenocarcinoma/ H. pylori suppression |
| Pure mastic gum/ 350 mg TID/ 1.05 mg TID/ 14 days | Dabos et al. (2010 / Greece) (20) | Medicinal product for gastrointestinal upsets with an acidic fraction that can kill H. Pylori in stomach | Randomized controlled clinical trial | 52 | A patient suffering from an H. pylori infection | Yes | No effect of a combination of mastic gum and pantoprazole on H. pylori/ Antibacterial activity of mastic gum alone against H. pylori/ No effect on the eradication rate |
| 1st study: Washed virgin olive oil/ 30 g/ 14 days followed by unwashed virgin olive oil/ 30 g/ 14 days; 2nd study: different extra virgin olive oil/ 30 g/ 14 days | Castro et al. (2012/ Spain) (21) | Antibacterial activity because of phenolic compounds | Pilot clinical trial | 1st study: 30; 2nd study: 30 | Helicobacter pylori ‐infected subjects | No | Moderate effectiveness in eradicating H. pylori |
| D-Reglis/ 380 mg licorice tablet) twice daily/ Four weeks | Momeni et al. (2014/ Iran) (22) | Efficacious for H. pylori eradication/ Anti-inflammatory actions | Randomized, double-blind, placebo-controlled, clinical trial | 60 | Patients with abdominal pain or dyspepsia and gastric ulcer, duodenal ulcer, gastritis or duodenitis in upper endoscopy and H. pylori positivity | Yes | As efficient as bismuth in H. pylori eradication |
| Licorice extract/ 380 mg twice daily/ Two weeks | Hajiaghamohammadi et al. (2016/ Iran) (24) | Antioxidant/Anti-cancer/Anti-inflammatory/ Secreting secretin/ Anti-adhesive effect against H. pylori by inhibiting DNA gyrase | Randomized controlled clinical trial | 120 | Patients suffering from dyspepsia either with peptic ulcer disease (PUD) or non-ulcer dyspepsia (NUD) with a positive rapid urease test | Yes | Higher eradication in patients with PUD/No difference in patients with dyspepsia |
| Garlic oil/ 4 mg capsule four times per day/ Two weeks | Mcnulty et al. (2001/ UK) (27) | Reduced risk of gastric cancer | Pilot clinical trial | 5 | Dyspeptic patients with positive serology for H. pylori | No | No evidence of H. pylori eradication and suppression |
| Alcoholic cinnamon extract/ 40 mg twice daily/ Four weeks | Nir et al. (2000) (29) | Inhibition of the growth of H. pylori | Pilot, randomized, placebo-controlled, clinical trial | 23 | Patients with positive Campylo Bacter Urease Test (CUT) | No | Did not demonstrate a decline in the colonization rate |
| Cinnamon extract/ 40 mg capsule twice daily/ 14 days | Imani et al. (2020/ Iran) (30) | Anti-inflammatory/ Antimicrobial activity | Randomized, double-blind, placebo-controlled, clinical trial | 98 | Helicobacter pylori infected patient | Yes | Increased eradication rate/Reduced complications of H. pylori treatment and increased efficacy of antibiotics |
| Alcoholic preparation of Brazilian green propolis/ 20 drops three times/ Seven days | Coelho et al. (2007/ Brazil) (32) | Phenolic components inhibiting H. pylori growth | Pilot clinical trial | 18 | Positive UBT patients | No | No effect on the eradication rate in 83 percent of the patients |
| Lycopene/ 30 mg daily/ 30 days | Shidfar et al. (2012/ Iran) (34) | Interfering oxidative processes | Parallel group quasi-control trial | 54 | Patients had been referred to the endoscopy with a positive rapid urease test | Yes | No significant effect on the eradication rate |
| Melatonin: 5mg; Tryptophan: 250mg/ 21days | Celinski et al. / 2012/ Poland) (37) | Potent endogenous antioxidants/Acting as a common mediator of inter-organ communication between the upper and lower portions of the gut | Randomized, placebo-controlled, clinical trial | 42 | Helicobacter pylori positive, Gastric or ulcer disease | Yes | Significant effect on the eradication rate (when added to omeprazole treatment) |
| Melatonin/ 3 mg daily/ 14 days | Abdi et al. (2018/ Iran) (38) | Patient healing lesions like stomatitis, Oesophagitis, Peptic ulcers/ Cytoprotection/ Antioxidant/Free radical scavenger activity | Randomized, double-blind, placebo-controlled, clinical trial | 118 | Patients with positive rapid urease test via an endoscopy | Yes | No significant effect on the eradication rate in ITT or PP analysis |
| Nickel-free diet/ N/A / 30 days | Campanale et al. (2014/ Italy) (41) | Activating Urease and Hydrogenase | Pilot, randomized, controlled, clinical trial | 52 | Patients who had been diagnosed with H. pylori infection by UBT | Yes | Increased eradication rate |
| High-PUFA (Efamol)/ Four 500mg capsules Four times daily/ 35 days | Duggan et al. (1997/ England) (44) | PUFAs inhibiting gastric acid secretion while increasing cytoprotective prostaglandins | Randomized, double-blind, placebo-controlled, clinical trial | 40 | Patients with proven infection with H. pylori/ endoscopic evidence of past or present duodenal ulcer disease | Yes | No significant change in either the severity of H. pylori infection or prostaglandin levels |
| PUFAs/ 2g mixture of fish oil and black currant seed oil/ Eight weeks | Frieri et al. (2000/ Italy) (45) | Inhibiting the growth of H. pylori, increasing PGE production, increasing the effectiveness of the mucosal barrier, and hindering bacterial adhesion to the gastric epithelium | Blinded clinical trial | 15 | Helicobacter pylori positive patients with mild functional dyspepsia | Yes | No effect on eradication; Modification of cellular membranes of both bacterium and host; Reduced H. pylori virulence |
Summary of Human Studies on the Effect of Complementary and Dietary Supplements on Helicobacter pylori Eradication
-thumbnail.webp)