1. Background
Bladder cancer ranks as the tenth commonest cancer worldwide, with estimated 573,000 incident cases in 2020 (1). There are distinct gender disparities in bladder cancer incidence, mainly attributed to gender-specific risk factor exposure and regulation of the sex steroid hormone pathway (2). Generally, bladder cancer can be divided into non-muscle-invasive bladder cancer (NMIBC) and muscle-invasive bladder cancer (MIBC), and the former occurs in approximately 70% of the patients with organ-confined bladder cancer (3). Transurethral resection is recommended for the primary treatment of NMIBC, usually followed by immediate intravesical immunotherapy or chemotherapy according to the risk of tumor progression or recurrence (4). Radical cystectomy, together with cisplatin-based neoadjuvant chemotherapy, can bring survival benefits to MIBC patients (5).
Daurisoline is a bisbenzylisoquinoline alkaloid extracted from the rhizome of the traditional Chinese herb Menispermum dauricum DC. Benzylisoquinoline alkaloids are a structurally diverse group of specialized plant metabolites, including well-established narcotic morphine, antimicrobials sanguinarine and berberine, and the antitussive and anticancer drug noscapine (6, 7). A structural dimer of 1-benzylisoquinolines is bisbenzylisoquinoline alkaloid. The pharmacological properties of bisbenzylisoquinoline alkaloid have been explored over the years. For instance, tetrandrine, originated from Stephania tetrandra and other related species of Menispermaceae, has been pre-clinically tested to have effects against multiple cancers (8). Dauricine is another bisbenzylisoquinoline alkaloid extracted from M. dauricum DC, and has been detected to have tumor-suppressive effects in pancreatic cancer cells (9) and renal carcinoma cells (10). Dauricine and daurisoline have sensitized cancer cells to camptothecin by acting as autophagy inhibitors (11). A recent study has reported that daurisoline could suppress lung cancer tumorigenesis by disrupting the heat shock protein 90 (HSP90)-mediated stability of β-catenin (12). However, whether daurisoline can act as a tumor suppressor in bladder cancer is unknown.
Heat shock protein 90 plays an important role in the correct folding and stability maintenance of its client proteins, among which E3 ubiquitin-ligase HAKAI is newly discovered (13). HAKAI participates in the ubiquitination and endocytosis of E-cadherin (E-cad), thereby modulating the acquisition of mesenchymal phenotype in epithelial cells. HAKAI promotes non-small-cell lung cancer cell growth and invasion and increases the cisplatin sensitivity of the cancer cells (14). Also, HAKAI can facilitate hepatocellular carcinoma progression by mediating the stability of Ajuba (15), but the role of HAKAI in bladder cancer is poorly studied. Based on the collected evidence, daurisoline might promote HAKAI protein degradation through the possible mediation of HSP90.
2. Objectives
The present study sheds light on the pharmacological role of daurisoline and the underlying mechanism in bladder cancer.
3. Methods
3.1. Ethics Statement
Human tissues were obtained with informed consent from the patients and approval from the Ethics Committee of a local hospital. The experiments involving humans were in accord with the Declaration of Helsinki. Animal experiments were permitted by a local hospital and conformed to the standards and ethical requirements for managing laboratory animals.
3.2. Clinical Samples
Tumor tissues and adjacent normal tissues (about 2 cm away from the tumor) were collected from the patients (non-referral) with bladder cancer who underwent surgical resection from September 2012 to June 2014 at the local hospital. The tissues were washed with PBS, snap-frozen in liquid nitrogen, and then stored at -80°C for gene and protein expression detection. Inclusion criteria: Patients diagnosed with primary bladder cancer by pathological examinations and not receiving preoperative radiotherapy, chemotherapy, or immunotherapy. Exclusion criteria: Patients who were unwilling or unable to cooperate in the follow-up; patients suffering from mental illness or other serious diseases. A total of 12 patients with bladder cancer were excluded from this study, and 40 samples were included. Survival curves were plotted to analyze the 5-year survival of all the included patients. The clinical research was retrospective.
3.3. Cell Cultivation
Human bladder cancer cell lines (5637, T24, and EJ), immortalized normal ureter epithelial cell line (SV-HUC-1), HUVECs, and human embryonic kidney (HEK)-293T cells were all purchased from the American Type Culture Collection (ATCC, Manassas, Virginia, USA). These cells were cultured in DMEM (Gibco, Grand Island, NY, USA) with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin in 5% CO2 at 37°C.
3.4. Drug Treatment
Daurisoline and HSP90 inhibitor geldanamycin were obtained from Selleck Chemicals (Houston, TX, USA) and dissolved in DMSO. Cells were treated with daurisoline (0, 1, 2, 5, 10, or 20 μM) or 10 μM geldanamycin for 24 h. Animal experiments applied intragastric administration of 40 mg/kg daurisoline 2 days a time.
3.5. MTT Assay
Five-six-three-seven, T24, EJ, and SV-HUC-1 cells at logarithmic phases were digested by trypsin and seeded into a 96-well plate in which each well contained 5 × 103 cells and had 3 duplicates. After being cultured in 5% CO2 and saturated humidity at 37°C for 24 h, the cells were treated with daurisoline (0, 1, 2, 5, 10, or 20 μM). After the daurisoline treatment, the supernatant in the wells was discarded, and the plate was washed with a culture medium 3 times. The cells were incubated in a serum-free medium (100 μL/well) for 4 h. After the removal of the supernatant, each well was added with 10 μL of DMSO-dissolved MTT (5 mg/mL). Optical density (OD) was measured at 570 nm.
3.6. Cell Transfection
Five-six-three-seven and T24 cells were transfected with 2 μg of pcDNA3.1-HAKAI or pcDNA3.1, and accordingly assigned into the HAKAI and pcDNA3.1 groups. The plasmids were synthesized by GenePharma (Shanghai, China) and transfected into the cells using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Briefly, cells were cultured in a serum-free medium 1 day before transfection. The cell confluency was about 90% at the time of transfection. Plasmid DNA (2 μg) was diluted in 50 μL of Opti-MEM medium, and Lipofectamine 2000 was diluted in an equal volume of Opti-MEM medium. The two diluted solutions were mixed and left at room temperature for 20 min. The mixture was added to the cells, and the expression of genes was detected after the cells were cultured at 37°C for 24 h.
3.7. qRT-PCR
Total RNA was extracted using TRIzol (Invitrogen, Carlsbad, CA, USA) and reverse transcribed using corresponding kits (TaKaRa, Tokyo, Japan). Gene expression was detected using SYBR Green Mix (Roche Diagnostics, Indianapolis, IN) in the LightCycler 480 system (Roche, Indianapolis, IN, USA). Thermal cycling was set as follows: 10 s at 95°C; 45 cycles of 5 s at 95°C, 10 s at 60°C and 10 s at 72°C; 5 min at 72°C. Each reaction was performed in triplicate. GAPDH acted as an internal reference. Data were analyzed using the 2-ΔΔCt method (ΔΔCt = (Cttarget gene - Ctinternal reference)experiment group - (Cttarget gene - Ctinternal reference)control group). Primers used for the PCR and their sequences are presented in Table 1.
| Name of Primer | Sequences |
|---|---|
| HSP90-F | GCTCCAAGGGTTGACATGGT |
| HSP90-R | TGTAACTCATGGACGCAGGG |
| HAKAI-F | GCGAGCCGAATCATGGATCA |
| HAKAI-R | ATCACCAGGTGGAGCCTTTG |
| E-cad-F | GCTGGACCGAGAGAGTTTCC |
| E-cad-R | CAAAATCCAAGCCCGTGGTG |
| N-cad-F | TGGGAAATGGAAACTTGATGGC |
| N-cad-R | TGGAAAGCTTCTCACGGCAT |
| Vimentin -F | TCCGCACATTCGAGCAAAGA |
| Vimentin-R | TGAGGGCTCCTAGCGGTTTA |
| Snail-1-F | AAGATGCACATCCGAAGCCA |
| Snail-1-R | CATTCGGGAGAAGGTCCGAG |
| GAPDH-F | AATGGGCAGCCGTTAGGAAA |
| GAPDH-R | GCGCCCAATACGACCAAATC |
Primer Sequences
3.8. Western Blot
The proteins were extracted using RIPA lysis buffer (Beyotime, Shanghai, China). The protein concentration was measured using a BCA kit (Beyotime). The proteins were added with a loading buffer and denatured in a hot bath for 3 min. The electrophoresis was run at 80 V for 30 min and continued at 120 V for 1 ~ 2 h after bromophenol blue came into the separation gel. The proteins were transferred onto a membrane in an ice bath at 300 mA for 60 min. The membrane was soaked in a blocking buffer at room temperature for 60 min or at 4°C overnight. The proteins were incubated with primary antibodies against GAPDH (5174S, 1: 1000, CST, Boston, USA), HSP90 (4877S, 1: 1000, CST), HAKAI (A311-100A, 10 μg, Bethyl Laboratories, Montgomery, TX, USA), E-cad (14472S, 1: 1000, CST), N-cadherin (N-cad) (13116S, 1: 1000, CST), vimentin (5741S, 1: 1000, CST) and Snail-1 (3879S, 1: 1000, CST) on a shaking bed at room temperature for 1 h. The proteins were incubated with a secondary antibody at room temperature for another 1 h. The membrane was added with a developing solution to visualize protein expression.
3.9. Transwell Invasion Assay
Cells were resuspended in a serum-free medium, and about 3 × 105 cells in 100 μL of medium were added onto the top of a matrigel-coated filter membrane in a transwell insert (354480, Corning, NY, USA). The basolateral chamber was added with 600 μL of medium containing 10% FBS. The transwell insert was taken out 24 h after cell incubation at 37°C. The medium and remaining cells that did not invade from the top of the membrane were removed using wet cotton swabs. The cells in the plate were fixed in 4% paraformaldehyde for 30 min and stained in Giemsa solution for 20 min. The number of the cells that had invaded through the membrane was counted under a microscope.
3.10. Matrigel-based Tube Formation Assay
A 48-well plate (Millipore, Billerica, MA, USA) and pipette were pre-chilled, and matrigel (Corning, Tewksbury, MA, USA) was dissolved at 4°C. The matrigel (100 μL) was added into the plate using the pipette. The plate was placed into a 37°C incubator for 30 min to solidify the matrigel. Each well was added with 2 × 104 HUVECs cultured in a conditioned medium for 24 h. The HUVECs were cultured in the plate for 6 h and then photographed. The number of branches was analyzed by Image J (NIH, Bethesda, MD, USA).
3.11. Drug Affinity Responsive Target Stability Assay
Protein concentration was measured using a BCA kit. Cell lysates were mixed with daurisoline or DMSO for 3 h at room temperature and then incubated with pronases for 15 min (pronase: protein = 1: 1000). After polyacrylamide gel electrophoresis, the expression of HSP90 in the enzymatic hydrolysates was detected by western blotting.
3.12. Co-immunoprecipitation (co-IP) Assay
The cells were lysed in a pre-chilled RIPA lysis buffer, and the lysates were centrifuged at 14,000 g, 4°C for 15 min. The supernatant was collected and placed into another centrifuge tube. The tube was added with 500 μL of diluted rabbit or mouse antibodies against HSP90 (ab203126, 1: 100), E-cad (ab1416, 1: 100), or IgG (ab172730, 1: 20) (Abcam, Cambridge, MA, USA), and placed on a shaking bed at 4°C overnight or gently shaken at room temperature for 2 h. The tube was added with 100 μL of 50% protein A/G-agarose beads to capture antigen-antibody complexes and then shaken on a shaking bed at 4°C overnight or maintained at room temperature for 1 h. The mixture was centrifuged at 14,000 rpm for 5 s and the supernatant was removed. The antigen-antibody complexes were washed with 800 μL of pre-chilled RIPA buffer 3 times and resuspended in 60 μL of 2 × loading buffer. The loading samples were boiled for 5 min and then centrifuged at 14,000 g. The supernatant was boiled for 5 min before electrophoresis. Protein expression was detected by western blotting.
3.13. Heterotopic Bladder Carcinoma in Nude Mice
Specific pathogen-free (SPF) BALB/c nude mice (n = 18, 4 ~ 6 weeks, 16 ± 2 g) were purchased from Shanghai Experimental Animal Center, Chinese Academy of Sciences. All mice were raised in a sterile SPF laminar airflow room at 22°C ~ 26°C with 55 ± 5% humidity. The nude mice were randomized into 3 groups (pcDNA3.1, HAKAI, and daurisoline + HAKAI groups) and anesthetized by intraperitoneal injection of 100 mg/kg ketamine and 10 mg/kg xylazine. The mice in the pcDNA3.1 group were given hypodermic inoculation of 1 × 106 T24 cells transfected with blank pcDNA3.1. The mice in the HAKAI and daurisoline + HAKAI groups were transplanted with 1 × 106 HAKAI-overexpressing T24 cells, which were selected using 1000 μg/mL G418 and stabilized by 500 μg/mL G418. The daurisoline + HAKAI group was given intragastric administration of 40 mg/kg daurisoline 2 days a time after 2 weeks. Tumors were taken out from the mice after 28 days. The tumor volume was measured using the following formula:
After volume measurement, the tumors were embedded in paraffin and sectioned.
3.14. Immunohistochemistry
Tumors isolated from mice were fixed in 4% formaldehyde for 48 h and made into paraffin sections (4 μm). The sections were roasted for 20 min and dewaxed in routine xylene, followed by a wash in distilled water. After being washed with PBS 3 times, the sections were treated with 3% H2O2 at room temperature for 10 min. After 3 PBS washes, the sections were subjected to thermal antigen retrieval. After another round of PBS washes, the sections were soaked in normal goat serum-blocking buffer at room temperature for 20 min. The primary antibodies against Ki67 (ab15580, 1: 500) and CD31 (ab28364, 1: 50) (Abcam, Cambridge, MA, USA) were added to the sections and incubated at 4°C overnight. The secondary antibody was incubated at room temperature for 1 h. After 3 PBS washes, the sections were treated with DAB for 1 ~ 3 min. Cell nuclei were stained with hematoxylin for 3 min. The sections were dehydrated, transparentized, and mounted.
3.15. Statistical Analysis
Data were statistically analyzed using GraphPad Prism 7 and SPSS and presented as mean ± standard deviation. The sample size was estimated before experiment, and the experiment and data analysis were completed by different individuals. A normality test and a test for homogeneity of variance were carried out before the data were analyzed. The data that did not conform to the normal distribution were excluded, and all the included data met the homogeneity of variance. t-test was used to compare 2 groups. One-way or two-way analysis of variance was used to compare 3 or more groups, followed by Tukey’s multiple comparisons test. Kaplan-Meier survival curve was used to analyze the difference between the 5-year overall survival rates of bladder cancer patients with high HAKAI expression and low HAKAI expression, and SPSS software was used to perform univariate and multivariate Cox regression analyses. P < 0.05 was considered of statistical significance.
4. Results
4.1. High Expression of HAKAI in Bladder Cancer Tissues
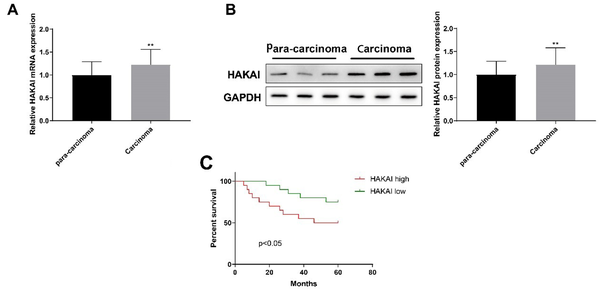
First, we detected the expression of HAKAI in tumor tissues and adjacent normal tissues of 40 bladder cancer patients using qPCR and western blot. The tumor tissues showed higher expression levels of HAKAI mRNA and protein than adjacent normal tissues (Figure 1A and B, P < 0.01). According to the median value of HAKAI expression in the tumor tissues, the patients were divided into HAKAI high expression group (n = 20) and HAKAI low expression group (n = 20). The Kaplan-Meier survival analysis showed that the HAKAI high expression group had a lower survival rate (Figure 1C, P < 0.05). The univariate and multivariate Cox regression analyses also indicated a significant difference in survival between these 2 groups. The above results suggest that HAKAI may promote the development of bladder cancer.
HAKAI is highly expressed in bladder cancer and associated with poor prognosis; A, qRT-PCR; and B, western blot were used to detect the expression of HAKAI mRNA and protein in bladder cancer; C, Kaplan-Meier survival analysis was used to analyze the relationship between HAKAI expression and patient survival. Paired t-test was used for the two-group comparisons. ** P < 0.01, carcinoma group vs. para-carcinoma group.
4.2. Daurisoline Suppressing Viability of Bladder Cancer Cells
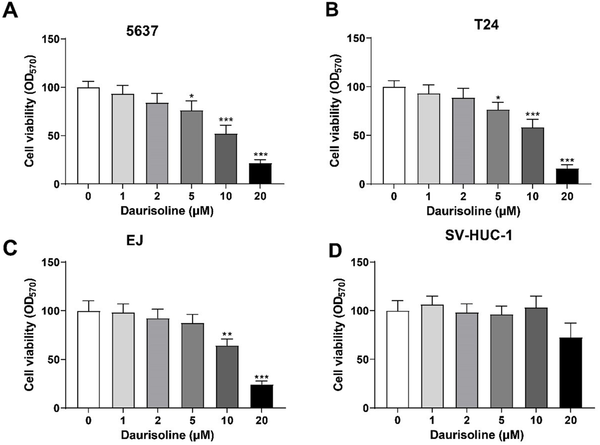
Five-six-three-seven, T24, EJ, and SV-HUC-1 cells were treated with daurisoline (0, 1, 2, 5, 10, or 20 μM) for 24 h. MTT assay tested the toxicity of daurisoline. Daurisoline exerted dose-dependent suppression on the viability of 5637, T24, and EJ cells. Daurisoline significantly reduced the viability of 5637 and T24 cells at a concentration of 5 μM and above and inhibited that of EJ cells at a concentration of 10 μM and above; the viability of the three bladder cancer cell lines was reduced with the increase in daurisoline concentration (Figure 2A - C, P < 0.05). Daurisoline at a concentration of 10 μM and below had no suppressive effect on the viability of SV-HUC-1 cells (Figure 2D). Only when the concentration reached 20 μM could the viability of SV-HUC-1 cells be partially suppressed (Figure 2D, P > 0.05). The measurement of the MTT assay demonstrates that daurisoline inhibits the viability of bladder cancer cells.
Daurisoline suppresses the viability of bladder cancer cells; MTT assay tested the toxicity of daurisoline to A, 5637; B, T24; C, EJ; and D, SV-HUC-1 cells. One-way analysis of variance was used for the multi-group comparisons, followed by Tukey’s multiple comparisons test. * P < 0.05, ** P < 0.01, and *** P < 0.001, daurisoline (1, 2, 5, 10, or 20 μM) group vs. daurisoline (0 μM) group.
4.3. Daurisoline Suppressing Invasion and Epithelial-Mesenchymal Transition of Bladder Cancer Cells and Angiogenesis of Human Umbilical Vein Endothelial Cells
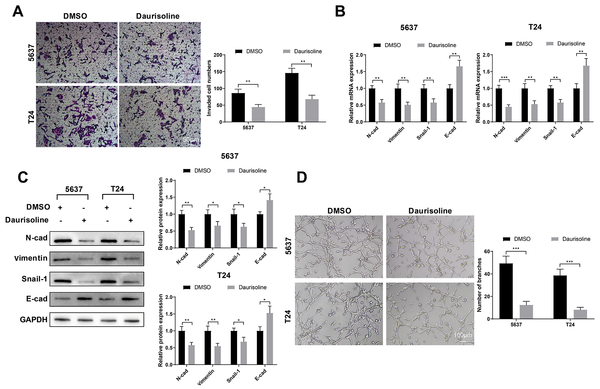
Five-six-three-seven and T24 cells were treated with 10 μM daurisoline or DMSO (negative control) for 24 h. Transwell assay showed that daurisoline suppressed the invasion of 5637 and T24 cells (Figure 3A, P < 0.01). Daurisoline decreased N-cad, vimentin, and Snail-1, and increased E-cad in 5637 and T24 cells, suggesting inhibited EMT (Figure 3B and C, P < 0.05). Human umbilical vein endothelial cells were cultured in a conditioned medium of daurisoline or DMSO-treated 5637 and T24 cells. Matrigel-based tube formation assay detected that the length of new-formed vessels was decreased in the daurisoline group compared with the DMSO group (Figure 3D, P < 0.001). From the above, daurisoline suppresses the invasion and EMT of bladder cancer cells and associated HUVEC angiogenesis.
Daurisoline suppresses invasion and epithelial-mesenchymal transition of bladder cancer cells and angiogenesis of human umbilical vein endothelial cells; A, Transwell assay assessed the invasion of 5637 and T24 cells treated with daurisoline or DMSO; B, qRT-PCR; and C, western blotting measured the expressions of E-cad, N-cad, vimentin, and Snail-1 in treated 5637 and T24 cells; D, matrigel-based tube formation assay assessed the angiogenesis of HUVECs cultured in conditioned medium of treated 5637 and T24 cells. An unpaired t-test was used for the two-group comparisons.
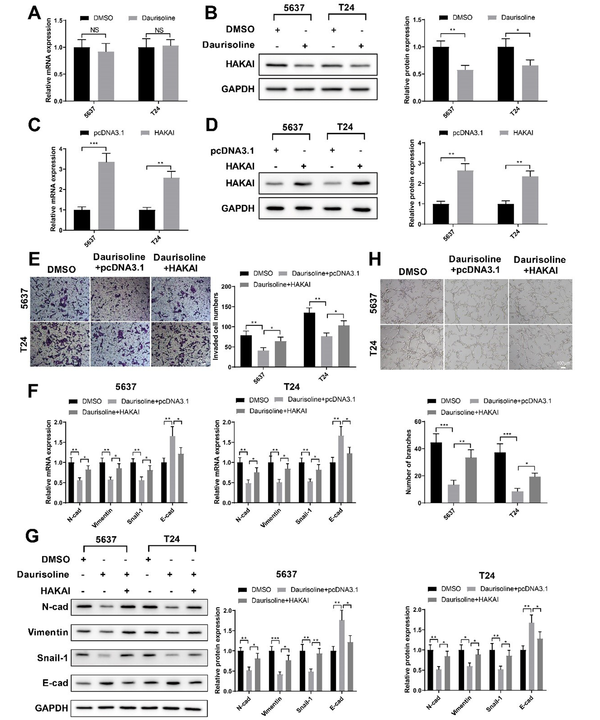
4.4. Daurisoline Exerting Tumor Suppressive Effects by Inhibiting HAKAI Protein in Bladder Cancer Cells
qRT-PCR and western blotting detected the expression of HAKAI in 5637 and T24 cells treated with daurisoline or DMSO. Daurisoline had no impact on HAKAI mRNA level (Figure 4A) but significantly reduced HAKAI protein level (Figure 4B, P < 0.01). Five-six-three-seven and T24 cells were transfected with pcDNA3.1-HAKAI and later treated with daurisoline to investigate whether daurisoline acted by mediating HAKAI. qRT-PCR and western blotting detected that both mRNA and protein levels of HAKAI were elevated after transfection of pcDNA3.1-HAKAI (Figure 4C and D, P < 0.01).
Daurisoline exerts tumor-suppressive effects by inhibiting HAKAI protein in bladder cancer cells; A, qRT-PCR; and B, western blotting measured the expression of HAKAI in 5637 and T24 cells treated with daurisoline or DMSO; C and D, the expression of HAKAI in 5637 and T24 cells transfected with pcDNA3.1-HAKAI or pcDNA3.1. After cell transfection and daurisoline treatment, E, transwell assay assessed the invasion of 5637 and T24 cells; F, qRT-PCR; and G, western blotting measured the expressions of E-cadherin (E-cad), N-cadherin (N-cad), vimentin, and Snail-1 in 5637 and T24 cells; H, matrigel-based tube formation assay assessed the angiogenesis of human umbilical vein endothelial cells (HUVECs) cultured in conditioned medium of 5637 and T24 cells. An unpaired t-test was used for the two-group comparisons. One-way analysis of variance was used for the multi-group comparisons, followed by Tukey’s multiple comparisons test.
Transwell assay showed that the invasion of 5637 and T24 cells were promoted in the daurisoline + HAKAI group compared with the daurisoline + pcDNA3.1 group (Figure 4E, P < 0.05). The expressions of N-cad, vimentin, and Snail-1 were increased, while the expression of E-cad was decreased in the daurisoline + HAKAI group compared with the daurisoline + pcDNA3.1 group (Figure 4F and G, P < 0.05). Matrigel-based tube formation assay showed that the angiogenesis of HUVECs was also promoted in the daurisoline + HAKAI group compared with the daurisoline + pcDNA3.1 group (Figure 4H, P < 0.05). Taken together, HAKAI overexpression reverses the suppressive effects of daurisoline on the invasion and EMT of bladder cancer cells and tumor-associated angiogenesis. Daurisoline acts on bladder cancer cells by inhibiting HAKAI.
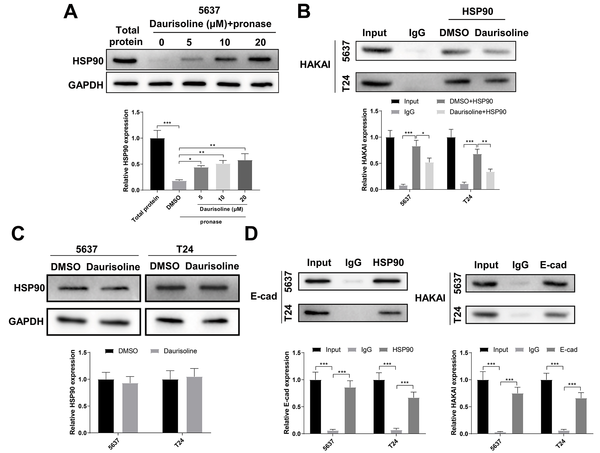
4.5. Daurisoline Targeting Heat Shock Protein 90 to Promote the Degradation of HAKAI Protein
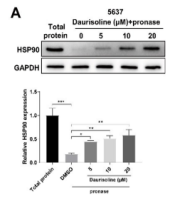
The mechanism underlying the effect of daurisoline on HAKAI was further investigated. Drug affinity responsive target stability (DARTS) assay was applied to determine the interaction of daurisoline with HSP90. Western blot analysis showed that HSP90 protein was degraded in the lysates of DMSO-treated 5637 cells while HSP90 could be detected in the presence of daurisoline (Figure 5A). Co-IP assay was performed to verify whether HSP90 could bind to HAKAI in bladder cancer cells. HAKAI protein was enriched in the complexes captured by the antibody against HSP90 in 5637 and T24 cells, and the expression of HAKAI protein enriched by the HSP90 antibody was down-regulated in the presence of daurisoline (Figure 5B, P < 0.05). Daurisoline had no impact on the expression of HSP90 protein in 5637 and T24 cells (Figure 5C). Therefore, daurisoline disrupts the binding of HSP90 to HAKAI by directly targeting HSP90, and promotes the degradation of HAKAI. Furthermore, HAKAI can induce ubiquitination of E-cad. E-cadherin was enriched in the complexes captured by the antibody against HSP90 in 5637 and T24 cells, and HAKAI was enriched in the complexes captured by the antibody against E-cad (Figure 5D, P < 0.001), indicating that HAKAI could bind to E-cad.
Daurisoline targets HSP90 to promote the degradation of the HAKAI protein; A, Drug affinity responsive target stability (DARTS) assay verified the binding between daurisoline and heat shock protein 90 (HSP90) in 5637 cells; B, co-immunoprecipitation (Co-IP) assay verified the binding between HSP90 and HAKAI in 5637 and T24 cells; C, Western blotting detected the expression of HSP90 protein in daurisoline-treated 5637 and T24 cells; D, Co-IP assay verified the targeting relationships among HSP90, HAKAI, and E-cadherin (E-cad) in 5637 and T24 cells. An unpaired t-test was used for the two-group comparisons. One-way analysis of variance was used for the multi-group comparisons, followed by Tukey’s multiple comparisons test.
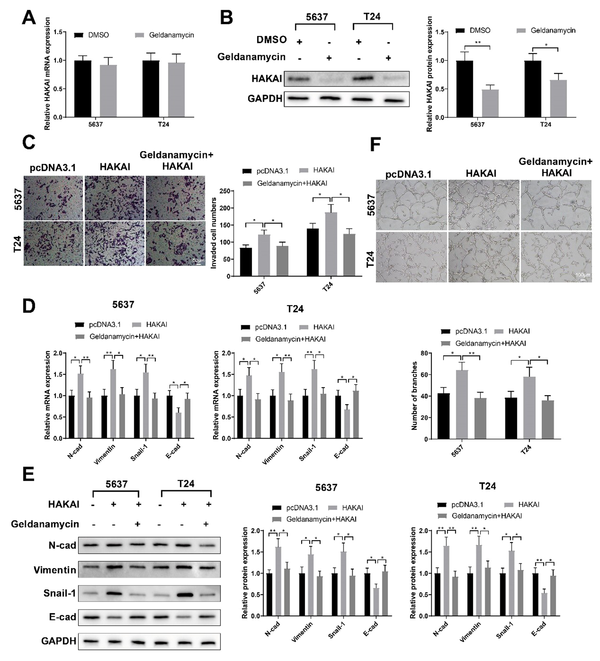
4.6. Heat Shock Protein 90 Inhibition Offsetting the Effects of HAKAI on Bladder Cancer Cells
To investigate the interaction of HSP90 with HAKAI in invasion and EMT of bladder cancer cells and tumor-associated angiogenesis, 5637 and T24 cells were treated with 10 μM HSP90 inhibitor geldanamycin for 24 h, and HAKAI was overexpressed in the cells. Geldanamycin treatment did not affect the HAKAI mRNA level (Figure 6A) but significantly reduced the HAKAI protein level (Figure 6B, P < 0.05). Transwell assay showed that the invasion of 5637 and T24 cells was suppressed in the geldanamycin + HAKAI group compared with the HAKAI group (Figure 6C, P < 0.05). The expressions of N-cad, vimentin, and Snail-1 were decreased, while the expression of E-cad was increased in the geldanamycin + HAKAI group compared with the HAKAI group (Figure 6D and E, P < 0.05). Matrigel-based tube formation assay showed that the angiogenesis of HUVECs was also inhibited in the geldanamycin + HAKAI group compared with the HAKAI group (Figure 6F, P < 0.05). Taken together, HSP90 inhibition suppresses the promotive effects of HAKAI on invasion and EMT of bladder cancer cells and tumor-associated angiogenesis.
Heat shock protein 90 (HSP90) inhibition offsets the effects of HAKAI in bladder cancer cells; A, qRT-PCR; and B, western blotting measured the expression of HAKAI in 5637 and T24 cells treated with geldanamycin or DMSO. After cell transfection and geldanamycin treatment, C, transwell assay assessed the invasion of 5637 and T24 cells; D, qRT-PCR; and E, western blotting measured the expressions of E-cadherin (E-cad), N-cadherin (N-cad), vimentin, and Snail-1 in 5637 and T24 cells; F, matrigel-based tube formation assay assessed the angiogenesis of human umbilical vein endothelial cells (HUVECs) cultured in conditioned medium of 5637 and T24 cells. An unpaired t-test was used for the two-group comparisons. One-way analysis of variance was used for the multi-group comparisons, followed by Tukey’s multiple comparisons test.
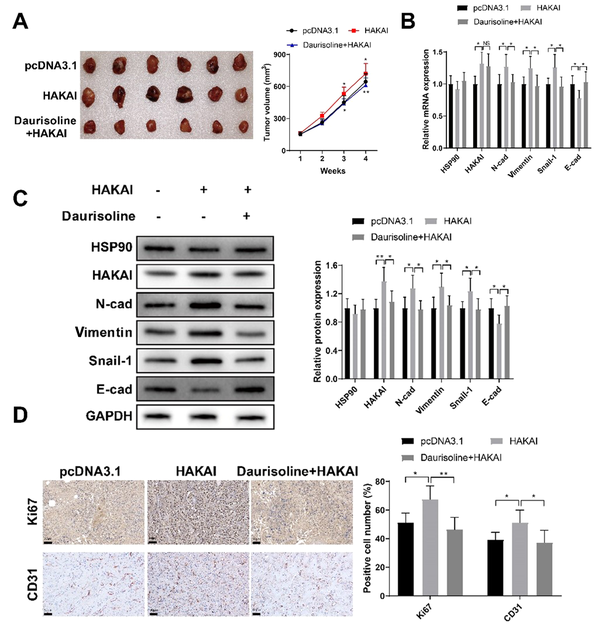
4.7. Daurisoline Suppressing Bladder Carcinoma Growth in Nude Mice
Nude mice were given hypodermic injection of T24 cells transfected with pcDNA3.1 or pcDNA3.1-HAKAI. Tumors of the HAKAI group were significantly larger than those of the pcDNA3.1 group, and daurisoline treatment reduced the tumor volume (Figure 7A, P < 0.05). The expressions of HAKAI, N-cad, vimentin, and Snail-1 were increased in the HAKAI group compared to the pcDNA3.1 group, while the protein levels of these genes were lessened in the daurisoline + HAKAI group compared to the HAKAI group; E-cad level was decreased in the HAKAI group but elevated in the daurisoline + HAKAI group (Figure 7B and C, P < 0.05). Immunohistochemistry displayed that Ki67 and CD31 positive cells were increased in the HAKAI group compared to the pcDNA3.1 group, while the positive rates were decreased in the daurisoline + HAKAI group compared to the HAKAI group (Figure 7D, P < 0.05).
Daurisoline suppresses bladder carcinoma growth in nude mice; nude mice were given hypodermic injection of T24 cells with stably overexpressed HAKAI or negative control T24 cells. The mice in the daurisoline + HAKAI group were later treated with daurisoline. A, Tumor volume; B and C, the expressions of heat shock protein 90 (HSP90), HAKAI, E-cadherin (E-cad), N-cadherin (N-cad), vimentin, and Snail-1 in the tumors; D, positive rates of Ki67 and CD31 in the tumors. One-way or two-way analysis of variance was used for the multi-group comparisons, followed by Tukey’s multiple comparisons test.
5. Discussion
Systematic treatment of bladder cancer has been developed over the years. Chemotherapy plays a key role in the prevention of MIBC metastasis as well as NMIBC reoccurrence and progression. Benzylisoquinoline alkaloids, widely distributed in a variety of Chinese herbs, possess potent pharmacological functions. Internationally, daurisoline was first reported to inhibit the ability of calmodulin to activate cyclic nucleotide phosphodiesterase among bisbenzylisoquinoline compounds (16). Daurisoline was later found to act as an antagonist of N-type and P-type calcium channels (17, 18). Since daurisoline has been discovered as a calcium channel blocker, investigations have been made into the pharmacological effect of daurisoline in calcium-mediated physiological and pathophysiological processes. For instance, daurisoline suppresses early afterdepolarizations by inhibiting L-type calcium current in a rabbit model of myocardial hypertrophy (19). Moreover, daurisoline has been reported to increase P-glycoprotein ATPase activity on the blood-brain barrier (20) and facilitate acidification of acid-sensing ion channel 1a (21). In recent years, studies have discovered an anti-tumor effect of daurisoline. Daurisoline disturbs autophagic flux and vasculogenic mimicry formation in hepatocellular carcinoma cells and sensitizes the cells to cisplatin- or sorafenib-induced apoptosis (22, 23). However, the effect of daurisoline on bladder cancer remains elusive to date. This study experimentally verifies the suppressive effect of daurisoline and related mechanisms in bladder cancer.
First, we tested the toxicity of daurisoline to bladder cancer cells and normal ureter epithelial cells. Daurisoline only suppressed the viability of bladder cancer cells, and the suppression was dose-dependent. Therefore, we further investigated the effect of daurisoline on bladder cancer cells. Epithelial-mesenchymal transition is characterized by the induction of mesenchymal markers such as N-cad, vimentin, and Snail-1 and the downregulation of E-cad, allowing polarized epithelial cells to adopt mesenchymal properties, including enhanced migratory and invasive capabilities (24). Transwell assay showed that daurisoline inhibited bladder cancer cell invasion. Moreover, the levels of N-cad, vimentin, and Snail-1 were decreased, and E-cad was increased in bladder cancer cells after daurisoline treatment. Furthermore, the angiogenesis of HUVECs cultured in a conditioned medium of daurisoline-treated bladder cancer cells was also inhibited.
HAKAI was first identified as an E3 ubiquitin-ligase by Fujita et al. (25). They found that HAKAI interacted with E-cad in a tyrosine phosphorylation-dependent manner and induced ubiquitination of the E-cad complex. Therefore, HAKAI overexpression promoted endocytosis of E-cad in epithelial cells and disrupted cell adhesion. HAKAI was recruited for E-cad ubiquitination by basolateral CD147 to inhibit nuclear translocation of β-catenin and expression of partitioning defective 3 in hepatocellular carcinoma progress (26). Hakin-1, a new specific inhibitor of HAKAI, reduced the oncogenic potential of colorectal cancer cell lines by inhibiting HAKAI-dependent ubiquitination of E-cad (27). It is reasonable to assume that the expression of E-cad is mediated by HAKAI in bladder cancer cells.
We found that daurisoline significantly down-regulated the protein expression of HAKAI in bladder cancer cells. Overexpression of HAKAI reversed the suppression of daurisoline on bladder cancer cell invasion. N-cadherin, vimentin, and Snail-1 were increased, and E-cad was decreased in daurisoline-treated bladder cancer cells after HAKAI overexpression. Bladder cancer cell-induced angiogenesis was also promoted by overexpressed HAKAI in daurisoline-treated bladder cancer cells. Therefore, daurisoline down-regulated the protein expression of HAKAI to prevent degradation of E-cad and inhibit malignant behaviors of bladder cancer cells. HAKAI could also regulate proteins related to cytoskeleton dynamics and posttranscriptional regulation, and a massive loss of expression in several proteasome subunits was detected during HAKAI-driven EMT (28). There may be many undiscovered molecular targets for HAKAI during tumor progression.
We identified HSP90 as a direct target of daurisoline in bladder cancer cells. Heat shock protein 90 chaperone is a central modulator of proteostasis in many cellular processes, from protein folding to DNA repair, development, immune response, and neuronal signaling (29). Due to its potent regulation of cellular proteins, HSP90 has become a promising drug target for treating several diseases, including neoplasms. Heat shock protein 90 inhibitors potentiated the anti-tumor activity of chemotherapy and photothermal therapy in bladder cancer treatment (30, 31). Demethoxycurcumin induced apoptosis of bladder cancer cells overexpressing human epidermal growth factor receptor 2 (HER2) by disrupting the HSP90-dependent stability of HER2 (32).
Heat shock protein 90 mediates the folding and maturation of more than 300 client proteins, many of which are conducive to accumulating cancer hallmarks (33). Here, HAKAI was demonstrated to be a client protein of HSP90. The enrichment of HAKAI protein by HSP90 antibody was disrupted in the presence of daurisoline. Moreover, E-cad was targeted by HAKAI. Taken together, daurisoline increased the HAKAI-mediated expression of E-cad by binding to HSP90, and daurisoline did not affect the expression of HSP90. We later investigated the effect of geldanamycin, a specific inhibitor for HSP90, in bladder cancer cells. Geldanamycin decreased the post-transcriptional protein expression of HAKAI. Inhibition of HSP90 reversed HAKAI-dependent promotion of bladder cancer cell EMT and angiogenesis.
Furthermore, the pharmacological effect of daurisoline was validated in nude mice transplanted with bladder cancer cells. HAKAI overexpression increased the expressions of N-cad, vimentin, and Snail-1, and decreased that of E-cad in the tumor tissues. The expression patterns of EMT-related proteins and HAKAI protein in the tumor tissues were revered in the presence of daurisoline. The daurisoline treatment also impeded the HAKAI overexpression-induced increase of the positive rates of Ki67 and CD31 in the tumor tissues.
In summary, daurisoline suppresses bladder cancer progression by inhibiting HAKAI-mediated EMT and angiogenesis. Daurisoline directly targets HSP90 and disrupts the stability of the HAKAI protein, which is required for the ubiquitination of E-cad. The study first validates the pharmacological effect of daurisoline in bladder cancer, and daurisoline shows great potential as a new chemotherapeutic agent for bladder cancer.