1. Context
Acinetobacter baumannii was for the first time identified as Micrococcus calcaoceticus by Beijerinck, a Dutch bacteriologist, in 1911. After several years, Baumann et al. (1968) cited in Asif et al. categorized all isolates similar to A. baumannii into one genus as Acinetobacter. They are currently classified as Gammaproteobacteria (1) non-fermentative, aerobic, and Gram-negative bacteria with extended distribution in natural sources such as soil and water and medical environments (2, 3). Some species of Acinetobacter, as commensal residents, exist on the human skin. However, the pathogenic species (pittii, nosocomialis, bauamnnii) are involved in opportunistic life-threatening infections, covering septicemia, pneumonia, endocarditis, urinary tract, and wound infections (3-5). During the last two decades, A. baumannii have appeared as a major concern in health care units. There is 60% fatality due to wide-spectrum procurement of new antibacterial-resistance genes by horizontal gene transfer (HGT) process acquiring plasmids and phages, respectively. In 2017, the WHO declared that carbapenem-resistant A. baumannii (CRAB) was ranked first in the list of antibiotic-resistant bacteria (6). This pathogen has been confirmed to be a successful infection worldwide, and its risk factors include the wayward consumption of various antibiotics, ventilator sets, and a long period of being bedridden in ICU (7-9). Bacteria often are in pursue of amelioration of pathogenicity. They Preferably alter solitude lifestyle to collective forms if adequate bacterial density ("quorum") is sensed; hence, some modifications in the transcription of genes and bacterial behaviors can be observed. The biofilm architecture is directly harnessed by Quorum Sensing systems. Biofilm formation capability enables A. baumannii to persist when facing undesirable terms (e.g., dehydration and nutrient deficiency) both on biotic and abiotic surfaces (10, 11).
2. A Short Review on Biofilm History
For the first time, Van Leeuwenhoek using his primary microscope, observed some microbial aggregations on the surface of the teeth. After a while, Heukelekian and Heller examined the bottle effect on marine organisms and concluded that the activity of these organisms increases when they are attached to the surfaces (12). According to Zobell’s observations, the number of bacteria on surfaces was at high rates compared to those in the seawater. With the improvements of electronic microscopes, it was possible to investigate biofilms with a better resolution. Jones et al. used an electronic microscope to detect biofilms produced by marine life in wastewater. They could also detect the cells surrounded by a-matrix-material and called it "poly-saccharide." Later, some studies reported that these materials were resistant to disinfectants such as chlorine (13-15), and they described a theory of biofilm mechanism.
According to the theory, on any biotic or abiotic surface on which microorganisms are attached, the advantages increase for microorganisms in a relevant ecological niche (12, 15). Biofilm studies have been conducted in industry, ecological systems, and the environment during the past decades. However, in recent years, most observations on biofilms have used electronic microscope equipment for the subtle identification of biofilms. They include studies on genes associated with cell attachment and biofilm formation (16). The biofilm description has changed over 25 years; in 1976 Marshall was found that there 73 were extracellular polymers integrating bacterial cells on the surface (15, 16). They also observed bacterial aggregations and claimed that biofilms consist of individual cells, producing micro-colonies. Marshall and Chracklis (1990) presented a better explanation of biofilms, indicating that "the exclusivity and heterogeneity of organic and non-living material on surfaces contribute to biofilm formation." (16, 17). Afterward, Costerton and Lappin-Scott (1995) reported their observations and showed that biofilms cling onto the surfaces and initiate gene expression to produce necessary components for bacteria (18). Now, according to a new description of biofilms, bacteria form biofilms in several stages: reversible attachment of bacteria to surfaces, colonization and biofilm maturation, irreversible attachment, and finally distribution of some parts of biofilm. The first investigation of biofilm structures depended on working with optical microscopes; however, after a while, the transmission electronic microscope was used to further observe the biofilm structure. However, it is the crucial element with high resolution to survey and study the structures (16). Bacteria can exhibit biofilm production on surfaces such as live tissues, medical devices, and aquatic systems.
Interestingly, no similarity is observed among biofilm structures produced by bacteria on different surfaces. As an example, the produced biofilm on aquatic systems, because of some spoiling material such as clay and filamentous bacteria, shares no similar characteristics with the ones on medical devices. In contrast, the biofilms on medical devices include coccoid bacteria and extracellular polysaccharides (EPS) (12, 16).
3. Bacterial Attachments: Biofilm Architecture
The desired conditions (e.g., a solid or liquid (blood)) provide the chance for the organism to attach to surfaces and begin to colonize and proliferate. There are some factors, such as the cell surface hydrophobicity, existence or lack of flagella, and the production of EPS, which affect the microbial attachment rates. Bacterial fimbriae play a remarkable role in surface hydrophobicity and the attachment of cells through involvement in domination against electrostatic existing between the surface and cells (19-21). Moreover, Bendinger et al. reported that the other factors involved in cell-surface interactions, known as proteolytic enzymes, cause the distribution of the attached bacteria such as Corynebacterium, Mycobacteria, and Nocardia. According to the mentioned subjects about biofilm, the attachment of organisms to surfaces is a complicated process (22). Hence, further studies and investigations on the cell-surface interactions contribute to the better understanding of the biofilm structure and mechanisms to generate antibiofilm agents to combat stubborn bacterial infections.
Biofilms are composed of microbial cells, and EPS is considered as principal matrix substance in biofilm structures (23). The whole structure of biofilms is surrounded by a bacterial self-produced polysaccharide matrix (24). The main structure of EPS consists of bacterial poly-saccharides with neutralized electric charge or polyanion, like what is observed in gram-positive bacteria. In some cases, EPS plays an anti-antibiotic role in microbial communities; remarkable amounts of the antibiotic return by the attachment of EPS to these substances; hence, the biofilm is like a bumper for bacteria (22). In some gram-positive bacteria, such as Staphylococcus, the structure of EPS may vary in chemical composition and is also cationic. Furthermore, the concentration of hydrogen bonds in the EPS composition causes dehydration, thereby preventing desiccation in biofilms (23, 25). Several characterizations of EPS influence biofilms. First, the structure and composition of polysaccharides determine the conformation. Second, various EPSs existing in biofilms share no similarity. Leriche stated that various organisms synthesized different EPS rates, and this amount increases as time passes (26). Moreover, the generation of EPS fluctuates by nutrients in the environment. The better the access of bacteria to carbon, the higher the rates of the EPS-production. More importantly, the limitation of phosphate, nitrogen, and potassium reduces the EPS synthase (23).
Last but not least, the slow growth rate of bacteria determines the improved EPS level. The biofilm-related infections are produced on external surfaces and in the body. Moreover, biofilms are growing and expanding on surfaces such as the teeth, plaque, respiratory tract, heart valves (endocarditis), and otitis media (24, 27, 28). Inside biofilms, polysaccharides, proteins, and DNA originate from bacteria themselves; hence, the matrix origin has a particular effect on the stability and preservation of biofilms. The development of biofilms in the past years has been investigated by confocal scanning laser microscopy employing green fluorescent protein. Due to the creation of three-dimensional images of biofilm along with advanced silicon image analysis, the beginning of biofilm formation in vivo depends on the reversible attachment of planktonic bacteria over surfaces sometimes covered with the layers of protein-pellicles. As sensitivity to antibiotics, pathogen bacteria in this stage of biofilms are prone to being killed by antimicrobial agents (24). It is important to note that there are fluctuations in bacterial growth in biofilm structures. The strategy of the bacteria to combat antibiotic therapy is to lower growth rates in biofilms; hence, the adsorption of antibiotics is lower, leading to their survival.
Accordingly, Evans et al. used the cell culture to evaluate the biofilm lifestyle on the growth rate of Escherichia coli and reported that the bacteria were highly resistant to antibiotics (29, 30). Accordingly, the biofilm framework sometimes consists of water channels (31). In the clinical samples of bacteria (biopsy), biofilm structures can be observed via optical microscopes (32). Consequently, recognizing a specific strain of bacteria requires molecular techniques. Moreover, we need specific staining techniques for the better and detailed analysis of biofilms.
4. Gene Transformation in Bacterial Communities
Biofilms provide an ideal encasement for microbes to exchange their genetic treasury with the external environment by exotic chromosomal materials, known as plasmids. The conjugation is carried out through microbial cells; hence, the relevant bacterial strains possessing conjugative plasmids can communicate and exchange genetic materials. In general, bacteria go through lower mutations in their planktonic forms, in contrast to their communicational lifestyles, and most genetic interactions happen in biofilm structures (33, 34). The F-conjugative-pilus of E. coli, as an adhesion factor, furnishes cell-to-cell interaction (12, 33). The bacterial biofilms cause chronic infections in humans due to the severe resistance to different antimicrobial agents and phagocytosis. It can be an elucidation for the MDR bacteria (multi-drug-resistance), with the capability of rejecting well-known antibiotics such as beta-lactam, aminoglycosides, and fluoroquinolones (35, 36). According to what explained, bacteria within groups secrete some degradation enzymes to combat antibiotics to survive and colonize. For this purpose, the target site of the mentioned enzymes has reduced affinity to attract antimicrobial molecules (36). To take the idea one step further, the antibiotics, including positive charges such as aminoglycosides, cannot bind to polysaccharide matrix with a negative charge (24). Further, within biofilm communities, bacteria express more efflux pump-related genes. For example, in patients with cystic fibrosis, the Pseudomonas aeruginosa strains are mostly observed to mutate their genes in biofilms. Moreover, one of the reasons for the high resistance of Staphylococcus-induced infections is the biofilm produced by these pathogens, or P. aeruginosa causes health problems by producing mucoid biofilms (16). It is estimated that high gene transfer rates within the biofilm structures are the main reason for obtaining extracellular DNA, which confers resistant and highly pathogen strains.
5. Quorum Sensing in Bacteria as a Noticeable Target
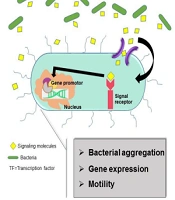
Principally, biochemical processes such as biofilm formation, bacteriocin production, conjugation, competence, pathogenesis, the secretion of signaling molecules, known as auto-inducers (AIs), and physiological mechanisms in bacteria are regulated by Quorum Sensing systems. Since the mass potency of bacteria is followed by the bacterial population changes in and around the microenvironments, it can be clearly recognized that monitoring microenvironment happens by signaling molecules. Accordingly, the secretion of signaling molecules provides a better cell-to-cell communication, which allows the bacterium to adapt environmental changes in a group of other bacteria (37, 38). Accordingly, there is a need to activate Quorum Sensing mechanisms to percept signals and cell density (39, 40). Cell-to-cell signal organizing is feasible by Quorum Sensing network components (38, 41). These components in Gram-positive bacteria are known as oligopeptides. The counterpart of oligopeptides is N-acyl-homoserine lactone, referred to as AHL mediating cell-to-cell connection in Gram-negative bacteria (42). A typical AHL system in Gram-negative bacteria is regulated by two proteins belonging to LuxI and LuxR protein families (40). Lux-I-type proteins generate AHLs directly acting with LuxR-type proteins. Then the mentioned complex coheres to the sequence of a promoter, called ‘ Lux-Box,’ so this process regulates the gene expression (9, 40). In general, the emission of the N-acyl homoserine lactone signal is referred to as an autoinducer (AI) (43). Synthesizing an autoinducer is mediated by a LuxI-type AI-synthase.
Furthermore, the threshold concentration of AI is associated with the high density of bacterial cells in the environment. To express the targeted genes, AI binds to a LuxR-type protein (43). AHL-signaling molecules are composed of homoserine lactone ring (HSL), which is bound to an acyl chain. The length of the acyl chain varies from C4 to C18. The hypothesis on the simple diffusion of AIs into bacterial cells was approved in a study on Vibrio fischeri luminescence characteristics (39, 44).
The eradication of bacterial biofilm-related infections is challenging for health authorities (30). Antibiotics are still at the center of bacterial therapy, both human and veterinary. All antibiotics share the same purpose of eradicating pathogenic bacteria (45, 46). Generally, in the presence of certain antibiotics, the sensitive bacteria are in the danger of eradication. Following this phenomenon, the resistant bacteria can survive and spread quickly, leading to the widespread antibiotic resistance (47). Pathogenic bacteria include a wide range of virulence factors to invade the host cells and finally disseminate relevant infections. Accordingly, providing new strategies to destroy the virulence factors and highlighting the significance of "quorum sensing" related factors would be more advantageous to combat infections instead of the total spoil of bacteria (45). As a result, the expression of virulence factors is controlled by a complex regulatory network, and providing strategies to inhibit the virulence factors attracts attention.
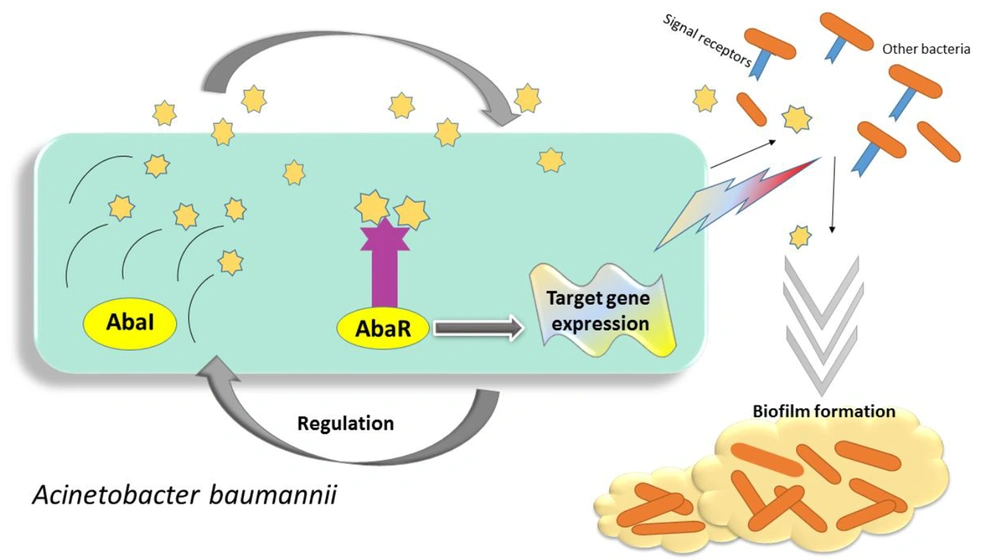
Quorum sensing is performed in several stages: (1) producing signaling molecules, (2) dissemination around the environment, (3) sensing and signaling molecules by membrane receptors, (4) attaching molecules to gene promotors, and (5) expressing virulence genes (48). As a result, bacteria trigger to evolve their pathogenesis or colonization on abiotic surfaces. Quorum sensing network in A. baumannii is an acyl-homoserine lactone (AHL)-dependent LuxIR-type system. The components of these systems are called AbaI as AHL-synthase, AbaR as transcriptional regulatory proteins, and AHL-receptors (3) (Figure 1). In many studies, the great relevance of Q.S networks with bacterial virulence has been confirmed. In this regard, a mutation in abaI genes has led to a significant reduction in biofilm formation and virulence compared to wild-type. Accordingly, the effects of quorum sensing can be considered as a significant factor in persistence on abiotic and biotic surfaces, and notably intrinsic antibiotic resistance of A. baumannii (49-52). Interestingly, in pursue of the quorum-sensing system in A. baumannii, the homologous of LuxI, which encodes AHL signaling molecules, has been described in Acinetobacter . According to the findings, they share 94% similarity in the amino acid content of the AbaI and AbaR in A. baumannii. In this regard, the mentioned components of the Quorum sensing system in Acinetobacter nosocomialis are termed as AnoR-(A. nosocomialis LuxI) and AnoI-(A. nosocomialis LuxR) (4).
The Quorums Sensing network in Acinetobacter baumannii is known as the LuxIR-system, consisting of AbaI and AbaR components, as described previously. This figure demonstrates the several steps of signal production of A. baumannii as follows: First, under a desirable condition, bacteria produce signaling molecules, then the surrounding bacteria begin to sense these signals and activate their specific receptors to receive the signals. Finally, the reception of signals leads to the expression of relevant virulence genes.
6. Relevance of Quorum Sensing and Quorum Quenching
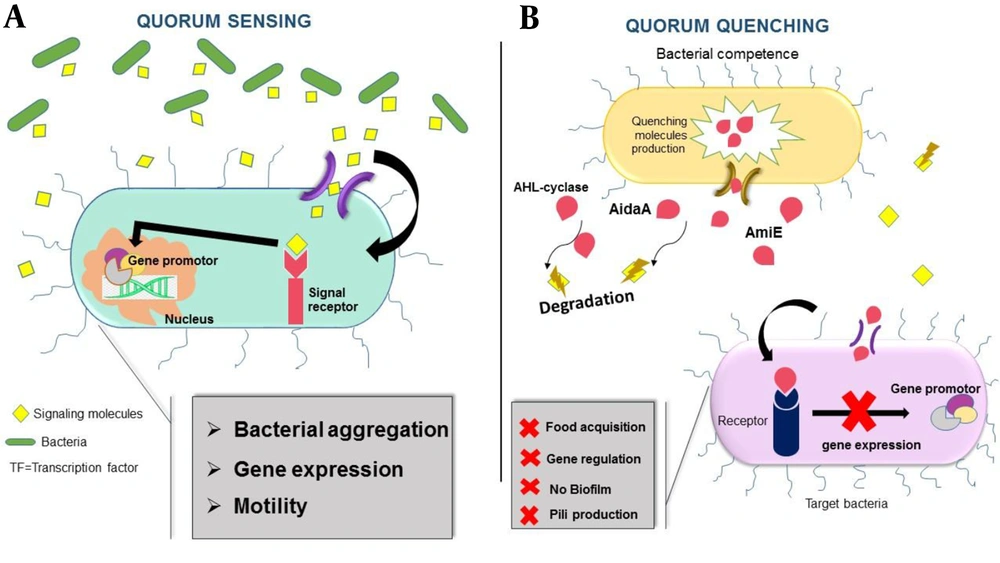
Pathogenic Gram-negative bacteria typically use Quorum networks to enhance their potentials to adapt to environmental tensions such as the regulation of resistance gene expression, genome transmission, and biofilm production and motility. Moreover, the bacterial invasion of the host cells is possible by the type VI secretion system (T6SS) (53). Aligned with Quorum Sensing mechanisms, bacteria prevent Quorum Sensing processes. Gram-negative bacteria in competitive communities exhibit unusual strategies to achieve superiority among other species (54). One of such strategies is releasing Quorum Quenching (QQ) molecules, which would affect the regulation of signal production, signal comprehension, and food acquisition mechanisms in competitor bacteria (54). Quorum Quenching molecules degrade signaling molecules (53); hence, the pathogenesis of bacteria will be decreased by disrupting quorum sensing (37). Hence, it can be concluded that various species of bacteria in microbial accumulations for superior competition possess both Quorum Sensing and Quorum Quenching networks (54). Generally, the strategies in the interference of Quorum sensing under the regulation of quorum quenching molecules are made up of two methods: non-enzymatic acting by Quorum sensing-inhibitors and an enzymatic method breaking down the signaling molecules and then extinguishing the Quorum Sensing system (55). A new quencher molecule, known as AidaA, is found in the clinical strains of A. baumannii. Furthermore, AHL-cylase and AmiE are the sensible kinds of Quenching molecules in Acinetobacter spps (54) (Figure 2).
A: Bacteria usually activate Quorum sensing systems to generate signaling molecules to link to other surrounding bacteria. These bacteria receive the signaling molecules by specific receptors and then activate nucleus transcriptional factors to express relevant genes. B: While bacteria under a multi-species condition include strategies to obtain some advantages for the acquisition of food and ecological niches, Quorum quenching mechanisms interfere with critical factors of quorum sensing networks and quench the activation of quorum network communication. AHL-cyclase, AmiE, and AidA enzymes as a Quorum Quenching mechanism affect inter-species relationships by hydrolyzing and degrading signaling molecules. Accordingly, the surrounding bacteria will not receive signals and express beneficial genes (pili production, biofilm formation).
7. Unsaturated Fatty Acids
By the continual occurrence of drug resistance in pathogens, the urgent demand for achieving alternative therapeutic agents to defeat infections is tangible (56); however, numerous efforts have been made to find suitable antibacterial agents targeting virulence factor regulators such as secretion systems, Quorum Sensing, biofilm formation, and the toxicity activity of the bacteria (57). One of the remarkable anti-biofilm agents containing antibacterial characteristics is unsaturated fatty acids (UFA). These chemical agents have attracted remarkable attention. Moreover, the absence of resistance mechanisms against these molecules is a prospective alternative to combat the virulence of microorganisms (58). Although the exact function of the recommended agents is still vague, the initial target of these molecules is the disruption of electron transport chain and oxidative phosphorylation in the cell membrane of bacteria (59).
Interestingly, the extraction of unsaturated fatty acids as small organic molecules has been proved in hetero-marine organisms (60, 61). We can conclude that UFAs are considered as new anti-biofilm agents to eradicate relevant infections. The administration of Virstatin, 4- [N-(1, 8-naphthalimide)]-n-butyric acid, to an animal model has proved its antibacterial effect on two major virulence factors of Vibrio cholera: Toxin co-regulated pilus (TCP) and cholera toxin (ctx), which impair the transcription regulator of relevant genes, known as ToxT. In other words, it prevents the dimerization of ToxT (56, 57). Furthermore, another study indicated that long-chain polyunsaturated fatty acids (LC-PUFA) were used to combat acne and dermal infections induced by Propionibacterium acnes and Staphylococcus aureus, respectively (58).
8. Certain Unsaturated Fatty Acids Block Quorum Sensing-Related Biofilm Formation
Recently, Virstatin's potential to inhibit biofilm production in A. baumannii, mediated by blocking pili synthesis, has been demonstrated (62). Meanwhile, in another study, its efficacy against biofilm structure in Acinetobacter, by involvement in Quorum Sensing system, has been studied (4, 56, 58). Concerning interference of UFAs and organic molecules in biofilm formation and motility of bacteria, which are under the regulation of Quorum Sensing networks, it can be assumed that they are a putative target for antibiofilm therapeutic agents. The following pathways can establish these strategies: first, prevention of signaling molecules production, and second, quenching and deactivation of signaling molecules (40). As to prevention of the ToxT interaction with DNA by PoA (Palmitoleic Acid), which is one of the regulatory proteins to transcript the coding genes of toxin co-regulated and cholera toxin in V. cholera (63), in another study, the efficacy of two unsaturated fatty acid molecules, i.e., myristoleic Acid (MoA) and Palmitoleic Acid (PoA), has been reported to decrease the biofilm production in A. baumannii ATCC17978 (61) ( Table 1).
Anti-biofilm Effects of Different Fatty Acids
4. Conclusions
The widespread drug resistance of the A. baumannii genus is a major trouble in clinical settings and accounts for high morbidity rates. The biofilm formation by these pathogens is an effective factor of antimicrobial failure to eradicate relevant infections. The collected data in this review have highlighted the specific role of Quorum Sensing and signaling processes between the microbes following biofilm production in microenvironments. Accordingly, understanding the role of this network and the performance of AHL signaling molecules can be of great importance in producing signaling inhibitors to prevent severe biofilm-related infections. Moreover, figuring out the constitution of biofilms and their backbone materials makes us generate other drugs with altered specific sites. Accordingly, interference in the expression of communication and signaling mechanism-associated genes can impress the bacteria's aggregative lifestyles since using new alternative therapeutic agents that target the quorum networks and their performance leads to the better control of A. baumannii infections in the community and hospitals.