1. Background
Toxoplasma gondii is an obligate intracellular parasite of members of apicomplexan order. This parasite is the most common parasitic infection in humans and many warm-blooded animals in the world (1, 2). Infection by this parasite in developing countries is estimated between 30 to 60 percent (3). About one-third of the world's population is infected with this parasite (1, 4). Humans and warm-blooded animals are intermediate hosts for T. gondii. Cats are playing a definitive host role in this parasitic cycle. This parasite forms cysts in various tissues of intermediate hosts and has sexual procreation in the intestine cells of the definitive host. Toxoplasma gondii is transmitted through sporulated oocysts excreted by cats, fruits, water, and soil contaminated with oocyst (5-7). This disease in people with a healthy immune system has mild and transient symptoms such as fever, enlarged lymph nodes, or no symptoms at all. But in pregnant women who are in their first pregnancy, it is dangerous and can be transmitted to the infant. In infants, it causes severe symptoms in brain problems such as microcephaly. Other groups at risk are AIDS, malignant problems, cancer patients, and so on (8-10). Studies show that there are several risk factors for toxoplasmosis, such as contact with soil, residence in a rural region, consumption of drinking water, etc. Therefore, military personnel are exposed to the infection during operations and maneuvers due to the lack of access to healthy drinking water and contact with soil (11).
The first-line drugs for this parasitic disease are sulfadiazine and pyrimethamine, which have various side effects, including bone marrow suppression and parasite drug resistance. We are looking for drugs with the least side effects on patients and the most effect on parasitic bradyzoites phase and cysts in body tissues (12). In recent years, nanotechnology has been used to diagnose and treat various diseases. The materials produced include microcapsules, conjugates, nanocomposites, and nanoparticles (13). Antiparasitic compounds in the form of nanosuspension, such as atovaquone nanosuspension, have a stronger effect on absorption and killing of more parasites than conventional drugs (14-16). In a research conducted by KarimiPourSaryazdi et al., the effects of silver nanoparticles on Toxoplasma parasites were evaluated. Silver nanoparticles have lethal effects on the parasite, leading to about 55% of apoptosis in Toxoplasma parasite (17). In another experiment performed by Tavakoli, the effect of manganese oxide (Mn2O3) nanoparticles on Leishmania was investigated, which resulted in lethal effects of the parasite at different concentrations. The results were confirmed by MTT assay, flow cytometry, and promastigote counting tests (18). Nanoparticles have various properties, including the production of free radicals that lead to the destruction of infectious organisms. With very small size, they can cross membrane barriers to exert their effects inside the cell, including the prevention of some intracellular chemical reactions (19, 20).
2. Objectives
This study aimed to investigate the different antiparasitic effects of tellurium oxide (TeO2) nanoparticles using different tests such as tachyzoite assay, MTT assay, and flow cytometry.
3. Methods
3.1. Synthesis of Silver Nanoparticles
Tellurium oxide (TeO2) NPs were synthesized by the Department of Pharmaceutical Biotechnology Research and the Research Center for Pharmaceutical Sciences, Faculty of Pharmacy, Tarbiat Modares University, Tehran. Different concentrations of nanoparticles (400, 200, 100, 50, 25, 12.5, 6.25 μg/mL) were prepared for MTT assay and tachyzoite assay.
3.2. Preparation of Toxoplasma gondii (RH Strain)
Toxoplasma gondii (RH strain) was prepared by Tarbiat Modares University, Department of Parasitology. To proliferate the parasite and preserve it, we injected the parasite into the peritoneum of BALB/c mice. When the parasite multiplied enough, it caused clinical symptoms in mice, including fever, swollen lymph nodes, and closed eyes. We injected sterile physiological serum into the mice peritoneum with 2.5 mL syringe to suspend the parasites in the serum. Physiological serum containing the parasite was removed from the peritoneum with syringe (21).
3.3. Tachyzoite Assay
For this experiment, different concentrations of nanoparticles (400, 200, 100, 50, 25, 12.5, 6.25 μg/mL) were prepared in microtiter plate (MP). Two wells were considered for control drugs, sulfadiazine, and pyrimethamine, and one well was used as a control (parasite without any drug). The solution was then centrifuged (1,000 g), and 6 × 105 tachyzoites (counted with hemocytometer slide) were added to all wells. Tachyzoites were explored for morphology and number of parasites in each well three, six, and 24 hours after exposure to nanoparticles. Each test was repeated three times (21).
3.4. MTT Assay for Macrophage
This experiment was used to evaluate the effects of nanoparticles and to evaluate cytotoxicity. To prepare MTT solution, five micrograms of MTT powder (Tetrazolium salt) (Gibco, Germany) were dissolved in 1 mL of PBS solution in the dark condition. We added in three separate plates 100 μL of RPMI 1640 (Gibco, Germany) solution plus 10% FBS (Gibco, Germany) composed with streptomycin (100 U/mL) (Gibco BRL) and penicillin (100 U/mL) (Gibco BRL) containing macrophages in each well. Also, we added different concentrations of nanoparticles in the volume of 100 microliters to all wells except the negative control well (macrophages without nanoparticles and drugs). We incubated them at 37°C with 5% CO2. After 24 hours, the surface fluid of the wells was removed, and 20 microliters volume of MTT solution was added to all wells and incubated again for four hours. Subsequently, 100 μL of DMSO solution was added to the wells and placed in an ELISA reader with 540 nm wavelength to calculate the wells absorption. We performed all steps of this experiment in a dark room (22, 23).
3.5. Flow Cytometry
In this research, flow cytometry technique with double staining [annexin V (V-FLUOS) and propidium iodide (PI)] was used to evaluate the type of cell death (apoptosis or necrosis). In this technique, we have four parts in the output of the device results. The different parts are as follow: the lower right was stained with annexin V (apoptotic cells), the upper right was stained with annexin V and PI (delayed or secondary apoptosis), the lower left was colorless (living cells), and the upper left was stained PI (necrosis cells) (24). In our research, Annexin V-FITC apoptosis (Bio vision) kit was used for flow cytometry. According to the kit instructions, we exposed 2 × 105 tachyzoites with 93 μg/mL concentration (IC50 concentration) of tellurium oxide nanoparticles. After 24 hours of incubation at 24°C, we first added 500 μL of buffer and placed the samples on the ice complex for 15 minutes. In the next step, after adding five μL of PI and Annexin V, the samples were explored by flow cytometry device.
4. Results
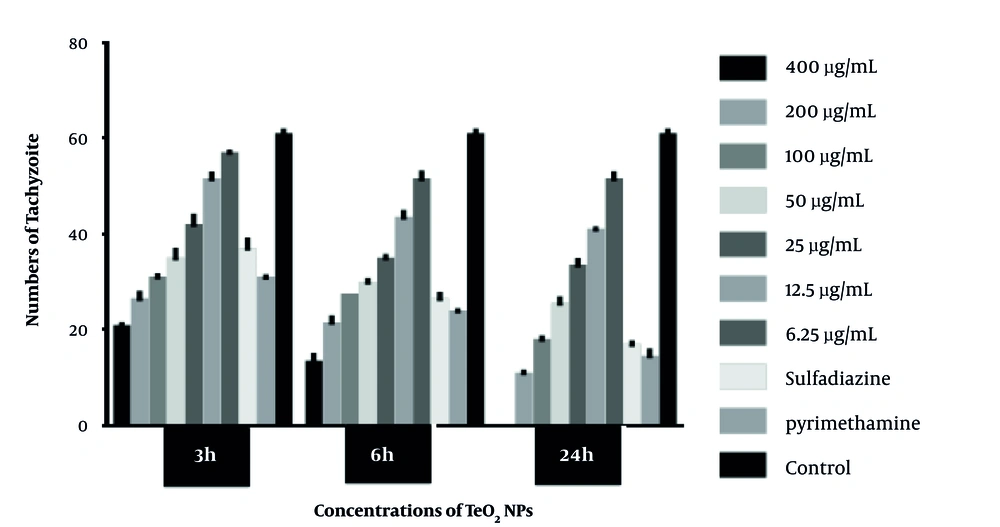
4.1. Tachyzoite Assay
Tachyzoites were counted with a light microscope at 3, 6, and 24 hours, and the results are plotted in Figure 1. The IC50 concentration of TeO2 nanoparticles in this study was estimated at 93 μg/mL, according to ocular count. According to this experiment, it was found that nanoparticles in high concentrations have a more lethal effect on the parasites. Also, the longer the parasite is exposed to nanoparticles, the greater the lethal effect. The results of light microscopy showed deformation of parasites exposed to nanoparticle concentrations.
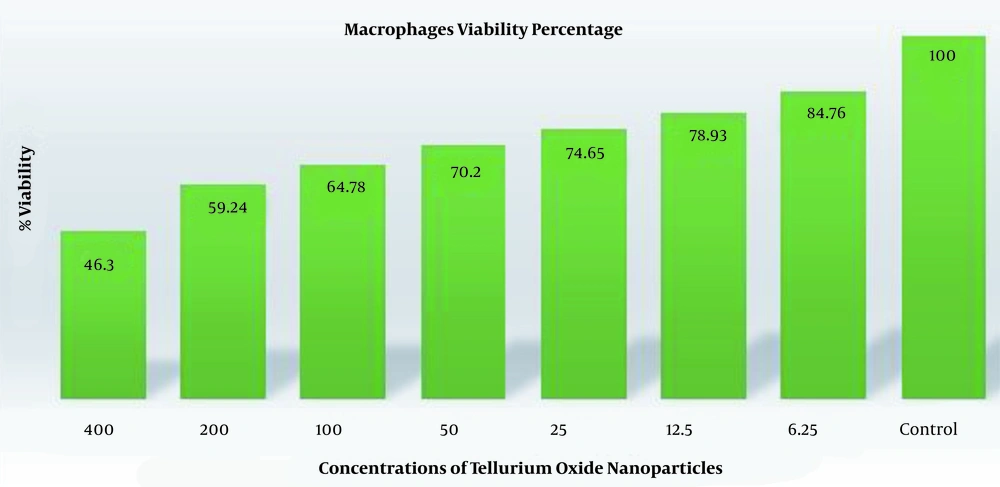
4.2. MTT Assay for Macrophage
The results of this experiment showed that concentrations such as 400 μg/mL was the most toxic effect on macrophages. At concentrations close to the IC50 of the parasite, most macrophages were alive. More information can be seen in Figure 2.
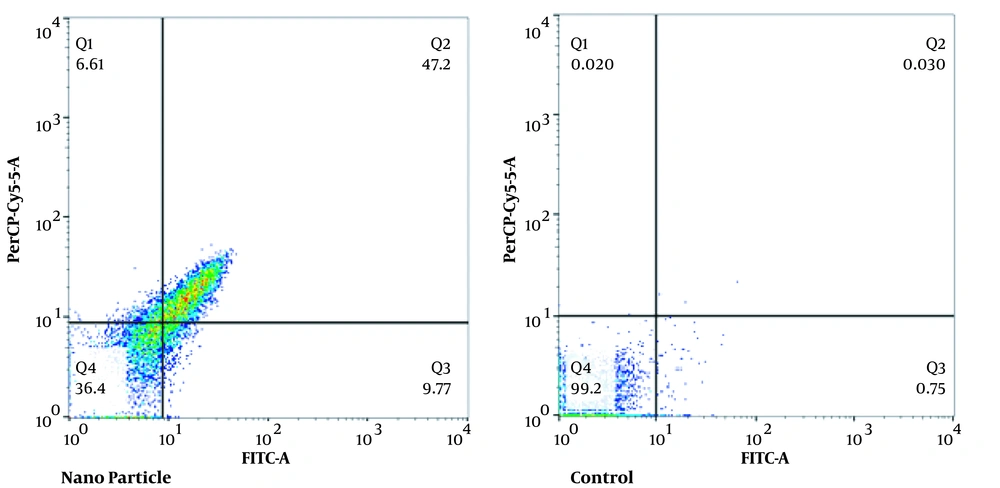
4.3. Flow Cytometric Analysis
This test was performed to evaluate apoptosis and necrosis. At 93 μg/mL of nanoparticle, the percentages of apoptotic (primary and secondary), necrotic, and living cells were 56.97, 6.61, and 36.4%, respectively. In the control group, the percentage of apoptotic (primary and secondary), necrotic, and living cells were 0.78, 0.02, and 99.2%, respectively (Figure 3).
5. Discussion
Toxoplasma gondii is a parasite with high prevalence in developing countries (about 30 to 60%) (3). About one-third of the world's population is infected with this parasite (1, 4). Routine anti-Toxoplasma gondii drug therapy cannot eradicate parasitic cysts. Immunocompromised patients cannot tolerate these drugs (12). The first-line drugs for the treatment and prevention of toxoplasmosis diseases are pyrimethamine and sulfadiazine. These drugs have various side effects, including neutropenia, leukopenia, thrombocytopenia, severe platelet count, blood disorders, allergic reactions, elevated serum creatinine, and elevated serum liver enzymes (25). The aim of this study was to investigate the destructive effects of tellurium oxide (TeO2) nanoparticles on T. gondii tachyzoites in vitro. According to the count of tachyzoites with hemocytometer slide and light microscope, the lethal effect of nanoparticles increases after exposing the parasites to different concentrations of nanoparticles (particularly high concentrations). In this study, the IC50 was 93 μg/mL. Also, we used MTT assay to investigate the possible toxic effect of nanoparticles on macrophages. We found that the survival rate of macrophages is high at low nanoparticle concentrations and has a little toxic effect on macrophages. The present research is the first research in this field, and resemblance research has not been performed using TeO2 nanoparticles on T. gondii. A study published in 2012 investigated anticancer effects of tellurium-complex compounds (AS101 and SAS). These compounds induced their antitumor and anticancer effects by inhibiting cysteine proteases e.g., cathepsin B, inhibiting tumor marker proteins, survivin, and constraining tumor e.g., IL-10 production (26). In our study, TeO2 nanoparticles led to 56.97% of apoptosis in T. gondii parasites. The nanoparticle induces apoptosis by activating the enzyme caspase-3. A study by Pandey in 2016 evaluated the effects of platinum tellurium nanowires with lasers on cancer cells such as A549. Tumor growth was significantly reduced after photothermal treatment through a combination of TePt and laser nanoparticles. Flow cytometry showed that it killed many cancer cells. Therefore, the importance of these new nanomaterials for the photothermal treatment of cancer was proven (27).
5.1. Conclusions
According to the findings of this study, TeO2 nanoparticles have a toxic effect on T. gondii and led to apoptosis in this parasite. We suggest that our research be conducted on other parasites.