1. Introduction
Coronavirus disease 2019 (COVID-19) (1) is an emerging viral disease, which was first reported in December 2019 in Wuhan, China, to the office of the World Health Organization (WHO) based in China as a cause of unknown pneumonia in three individuals (2).
The most common symptoms include fever, cough, and shortness of breath (3). Other symptoms include fatigue, muscle aches, diarrhea, sore throat, decreased sense of smell, and abdominal pain (3, 4). The incubation period is usually 5 days from exposure to onset, but may vary from 2 to 14 days (5). While most patients are asymptomatic or experience only a mild cold-like illness such as fever, cough, fatigue, and shortness of breath (6). some patients develop viral pneumonia and multiple-organ failure, and experience a severe disease (2).
In addition, various studies have shown that liver complications are observed in 2 - 11% of patients with COVID-19, and elevated liver alanine aminotransferase (ALT) and aspartate aminotransferase (AST) are observed in 14 - 53% of patients (7).
2. Case Presentation
The patient was a 3-year-old boy with symptoms of cholestasis, jaundice, and abdominal pain for two weeks referred to Ali-Ibn-Abitaleb Hospital in Zahedan, Iran. During this time, due to fear of COVID-19, the patient had been treated at home in the traditional way and experienced nausea and vomiting over the past three days, along with high-grade fever. Upon entering the emergency room, the patient was examined, which was monitored with a slight decrease in the level of consciousness (LOC) [based on the AVPU (alert, verbal, pain, unresponsive) system] and hospitalized with the diagnosis of hepatic encephalopathy.
At admission to the emergency room, the child was monitored, and his vital signs were examined. The patient's RR was 32, PR was 137, and his core body temperature was 38.9°C rectal. The patient's systolic and diastolic blood pressure was 76 and 45 mmHg, respectively. The patient had not taken any other potent medication except a history of taking unknown herbal medicines rubbed on the abdomen prescribed by a local midwife. No history of underlying liver or brain disease was reported, and the family had a moderate socioeconomic status. The decrease in the level of consciousness of the child during the last two days in the form of sleep disorders and lack of orientation to those around him caused the parents to refer to our medical center with confusion. The child's developmental and neural developments were normal (weight 16 kg, height 77 cm). No family history of diseases such as Wilson disease, metabolic diseases, or other liver diseases were reported. No history of contact with a patient with jaundice or suspected hepatitis detected.
At the initial examination, the patient was completely icteric and had abdominal distention. In addition, the examination of the mucous membranes and sclera of the eyes were icteric, and purpuric lesions were also found in the patient's mouth and back.
Examination of physically demonstrable symptoms of meningitis (Neck Redor, brudzinski sign, and kernig sign) was also negative in the child. The patient's PGCS (Pediatric Glasgow Coma Scale) was 12. The patient has normal muscle tone, and examination of the deep tendon reflexes (DTR) decreased slightly by +1. Lymphadenopathy was not found anywhere else on either side of the neck. On examination of the patient's limb, the lower limb underwent a permeable edema of +1, but no other positive findings were found.
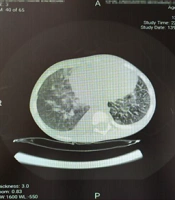
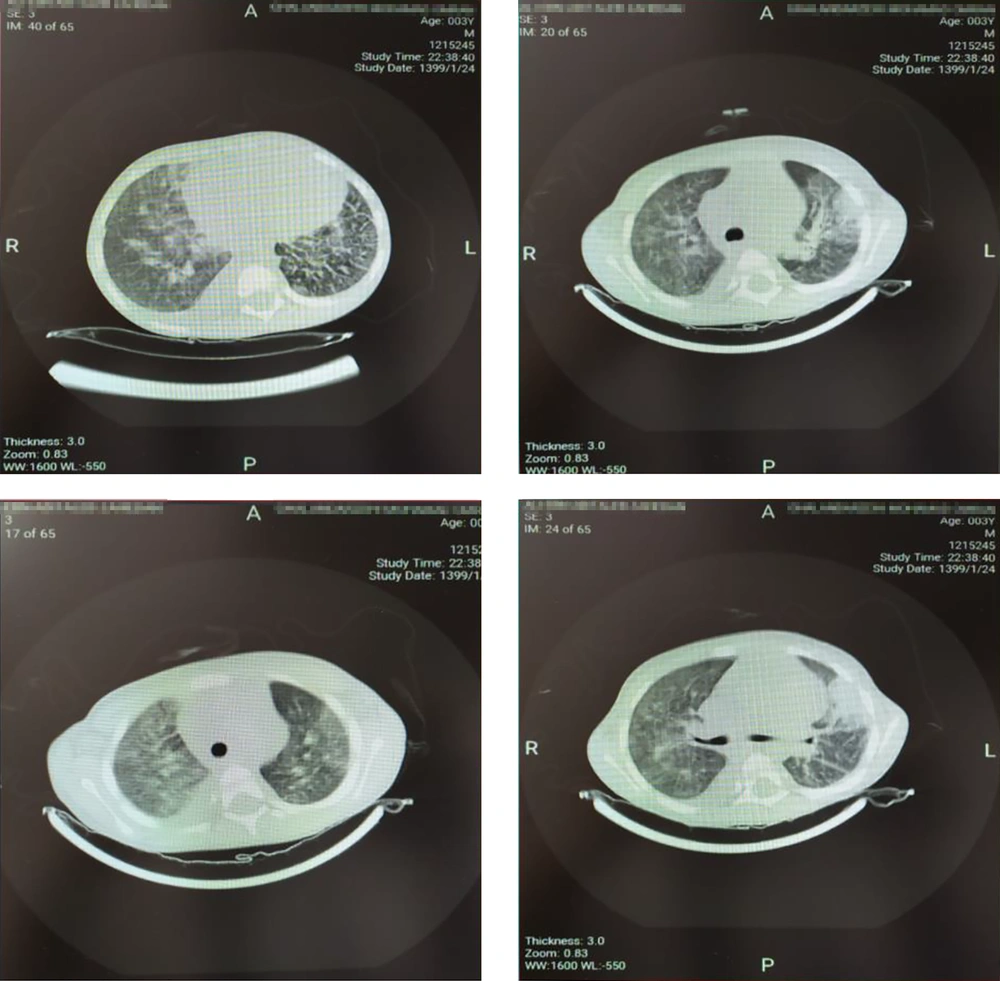
Examination of the patient's abdomen detected hepatomegaly about 6 cm below the edge of the ribs (moderate hepatomegaly), and other organs were normal. In the lung auscultation, crackles were heard in the base of the lungs. The patient's tests were sent with a possible diagnosis of hepatitis A and treatment of hepatic encephalopathy was started immediately (lab data are given in Table 1). Due to the patient's high-grade fevers, the patient was given the antibiotic cefotaxime, and the patient continued to be monitored by the nurses. Due to the patient's accompanying respiratory symptoms, a chest CT-scan was performed (Figure 1). In CT scan, bilateral patch infiltrations were seen in the lungs.
| Lab Data | First Day | Third Day | Ref. Value |
|---|---|---|---|
| AST | 280 | 405 | 0-41 |
| ALT | 647 | 530 | 0 - 37 |
| Alkaline phosphatase | 545 | 492 | 180 - 1200 |
| Bilirubin (T,D) | 1.2/0.7 | 2.3/0.9 | 0.2 - 1.2/0 - 0.4 |
| Blood Glucose | 135 | 127 | - |
| ABG (PH, PCO2, HCO3) | 7.47/26/19 | 6.82/38/6 | - |
| HAV Igm | Negative | - | - |
| Albumin | 3.1 | 2.3 | |
| Total Protein | 4.5 | 4.6 | |
| LDH | 1228 | - | 225 - 500 |
| WBC | 15.6 | 23.8 | 4.0 - 10 |
| Neutrophil count | 62% | 58% | - |
| Hb | 8.4 | 7.7 | 14 - 18 |
| Plt | 73 | 36 | 140 - 440 |
| PT | 14.5 | 21 | 10 - 13 |
| PTT | 22 | 55 | 28 - 38 |
| INR | 1 | 2.7 | 1 - 1.3 |
| Potassium | 4.6 | 6.7 | 3.5 - 5.2 |
| Sodium | 138 | 132 | 135 - 145 |
| Urea | 16 | 24 | 7 - 21 |
| Creatinine | 06 | 0.6 | 0.7 - 1 |
| Calcium | - | 8.3 | 8.5 - 10.5 |
Due to the high prevalence of COVID-19 in the region and to investigate the disease, polymerase chain reaction (PCR) was performed by sampling of the throat and nasal, which was positive after 12 hours. Immunoglobulin hepatitis A response was also negative during hospitalization. To confirm other diseases, the 24-hour urine copper, G6PD, viral hepatitis B, and C were also checked, all of which were reported to be normal and negative. The patient was transferred to the intensive care unit (ICU) prepared for COVID-19 patients and underwent a possible diagnosis of hepatic encephalopathy following COVID-19 and was treated with hydroxychloroquine and lopinavir/ritonavir [as prescribed by the treatment protocol (8)].
On the third day of hospitalization in the ICU, the patient's general condition and level of consciousness improved, and oral nutrition was started for the patient. The patient continued to be icteric and the patient's abdominal distension was also established. Patient tests on the third day of hospitalization are listed in Table 1. On ultrasound of the patient's liver, his liver was reported to be hetrogen, and the gallbladder wall to be 5 mm thick. Images of several lymph nodes with a maximum sagittal abdominal diameter (SAD) diameter of 6 mm were also seen in porta hepatis, and moderate free fluid was also seen in the abdominal and pelvic spaces.
On the fifth day of hospitalization, despite treatment for the patient's underlying disease and encephalopathy, the patient developed cardiac arrest, and unfortunately, the patient died after 45 minutes CPR. Finally, we may mention that the patient's family did not provide a consent for the liver biopsy; hence, the presence of the virus in the patient's liver cells remained a vague issue.
3. Discussion
Liver cell destruction and subsequent damage are usually caused by direct infection of liver cells by viral agents. In a study by Yeo et al. about 10% of patients with COVID-19 who had diarrhea were diagnosed with RNA in the feces and blood at the same time, which could confirm the possibility of liver cells being infected (9).
Extensive research has shown that coronavirus, using its surface glycoprotein called spike, is highly susceptible to angiotensin-converting enzyme 2 (ACE2) enzymes attached to type 2 alveolar cells and exerts its destructive effects (10). The virus also affects the organs of the gastrointestinal tract, as ACE2 is abundant in the cells of the gastric epithelium, duodenum, and rectum, as well as endothelial cells and small intestinal enterocytes (11). A preliminary study found that ACE2 receptors were also present in small amounts in cholangiocytes, (12) which increased the risk of liver involvement with SARS-CoV-2, but none of the studies found the virus in the liver unless some case presentations proved hepatic failure and encephalopathies due to COVID-19 (13). Pathological studies in severe acute respiratory syndrome (SARS) patients showed a small presence of the virus in liver cells, which was due to the low number of ways the virus entered (14). However, no disease was reported in the liver in Middle East respiratory syndrome (MERS) cases.
In the case report serial study by Abel et al. in 2020, four children with COVID-19 who presented with neurological symptoms and encephalopathy were examined. All four children were observed to have inflammatory symptoms in splenium callus on neuroimaging (15).
Various cases of COVID-19 encephalopathy have been reported. Sejal et al. reported a case of adolescent resulting in encephalopathy (16).
Liver disease is much more likely to be contracted by viruses in patients with chronic liver disease such as Wilson's, alcoholic and non-alcoholic fatty liver disease, and chronic hepatitis. However, our patient was also screened for all possible chronic liver diseases, and no evidence of a history of chronic liver disease was found. Our patient's liver ultrasound did not report fatty liver and described all the findings as acute and related to acute illness. Since the outbreak of COVID-19, no severe liver involvement has been reported in patients with the virus, or death, or encephalopathy associated with the virus, and this has not been studied in pediatric patients.
We are currently collecting information on patients admitted to this center to provide a more comprehensive study to the scientific community in the near future by examining the possible complications of the disease on patients' livers. According to the case, the patients' liver function (especially pediatrics) and possible complications, mortality, risk of secondary infection, hepatic encephalopathy, gastrointestinal bleeding, and liver damage are very important. We recommend that researchers pay more attention to these issues in their future studies.