1. Background
Tonsillectomy is one of the most common surgeries in children. This surgery is associated with complications such as pain, nausea and vomiting, and bleeding. Pain is one of the main side effects of tonsillectomy, especially in children (1), and if not controlled effectively, it can prolong recovery time and hospital discharge (2). Also, it can impair eating and drinking and lead to dehydration. On the other hand, considering that most of patients in this group are children, this complication can have psychological effects on them (1, 3).
Various studies have suggested methods and drugs for reducing pain after tonsillectomy, including opioids, non-steroidal anti-inflammatory drugs (NSAIDs), steroids, and acetaminophen. The use of NSAID drugs after tonsillectomy is controversial because they impair platelet aggregation and function and intensify postoperative bleeding (4).
Steroids, such as dexamethasone, are used to control nausea and vomiting, and pain after tonsillectomy, but its side effects such as high blood pressure and hormonal and gastrointestinal disorders have limited the use of this class of drugs (4).
The use of opioids to control pain after tonsillectomy has been limited due to respiratory depression since these patients are usually treated on an outpatient basis and are discharged from hospital soon (5).
Bupivacaine is a widely used medication, which works to prevent pain by inhibiting peripheral nociceptive excitation following tissue damage and stop the sensitization of the central nervous system (6). Numerous studies have been performed on the efficacy of bupivacaine in reducing pain after tonsillectomy. However, some studies did not find the use of this drug to be effective in preventing pain and noted a number of risks, including life-threatening upper airway obstruction, visual loss, cervical osteomyelitis, and vocal cord paralysis (1). Based on the contradictory results concerning the effects of bupivacaine on postoperative pain, we examined the effect of topical administration of bupivacaine on postoperative pain and nausea and vomiting (PONV) after tonsillectomy.
2. Methods
2.1. Trial Design
This prospective, double-blind, randomized clinical trial was conducted using a parallel assignment of patients into treatment and placebo groups after approval of the Ethics Committee of Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran, from December 2016 to January 2018 at Imam Khomeini Hospital, Ahwaz, Iran (AJUMS.REC. 1393.146). The RCT code of this study was: IRCT20180909040979N6. Written informed consent was collected from parents of the pediatric patients.
2.2. Participants
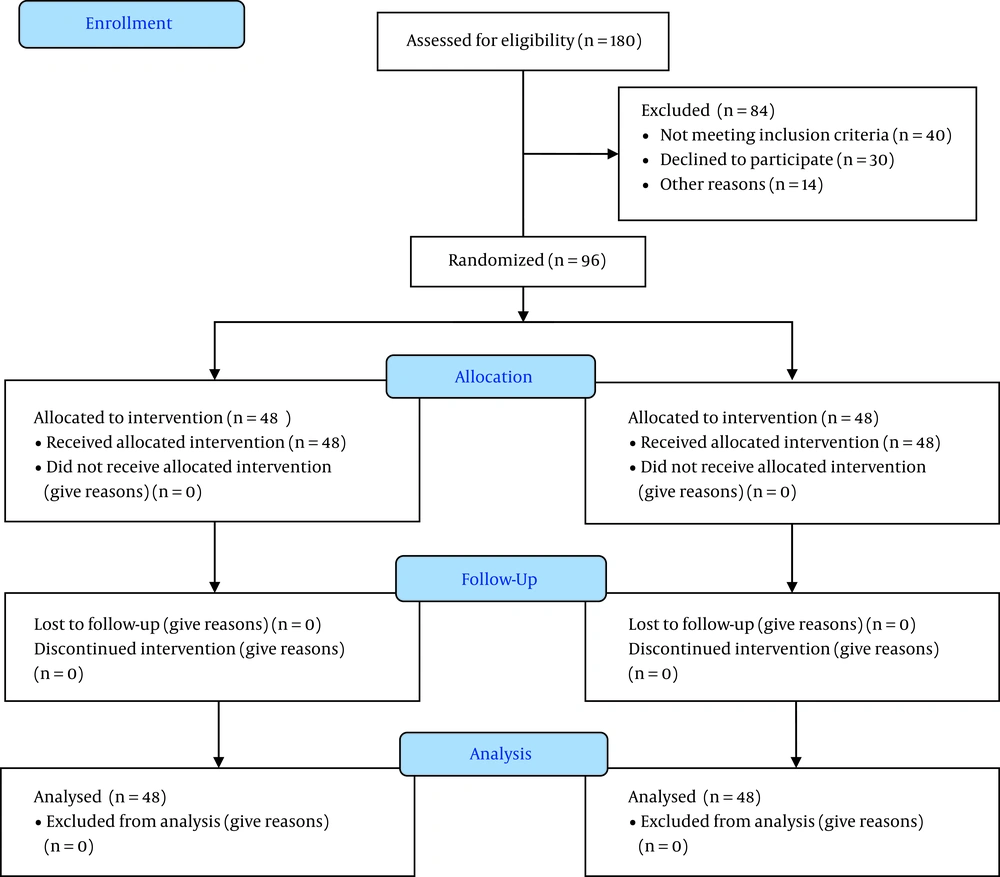
Of the 180 children undergoing elective tonsillectomy, 96 were suitable to contribute to the study. Thus, they were assigned into the two groups of bupivacaine (48 patients) and placebo (48 patients) (Figure 1)
Patients were included in the study if they met the following inclusion criteria: American Society of Anesthesiologists (ASA) I, elective tonsillectomy, and age between 7 - 12 years.
Patients were excluded from the study if they met the following exclusion criteria: coagulation disorder, difficult intubation, symptoms of acute pharyngitis, hypersensitivity to bupivacaine, regular use of analgesic, hypnotic, or sedative medications, the presence of rhonchi or pyrexia, and failure to comprehend the visual analog scale (VAS).
2.3. Sample Size
We calculated the sample size required for this study by using the sample size estimation formula and a confidence interval (CI) of 95%. Based on previous data (7), 90 patients were participated in this study.
2.4. Randomization
Patients were divided into the bupivacaine and control groups with simple random sampling on a 1:1 ratio. Randomization was performed by one of the investigators who did not have a role in the treatment of the participants.
2.5. Study Settings and Interventions
All the patients were evaluated before surgery by an anesthesiologist who was blinded to the research. Relevant laboratory investigations (complete blood count, coagulation profile) were conducted. All the patients were kept fasting, according to standard fasting guidelines. Patients who participated in the study were educated on how to use a 0 - 10 VAS, which consisted of a 10‐cm horizontal column that was separated into ‘no pain’ on the left and ‘worst pain’ on the right (8). The patients were instructed by a nurse who was blinded to the study at the Outpatient Surgery Unit of the hospital.
In the operation room, the patients were divided into bupivacaine (Group B) or control group (Group C) with simple random sampling.
After insert of standard monitoring (noninvasive blood pressure, ECG, and pulse oximetry), both groups were put under similar anesthesia: 0.02 mg/kg atropine, 2 μg/kg fentanyl, 4 - 5 mg/kg thiopental sodium, and 0.5 mg/kg atracurium, and underwent nasotracheal intubation. Capnograph was used for confirmation of the placement of the endotracheal tube. The patients were kept under anesthesia using a combination of nitrous oxide with oxygen (50% - 50%), as well as 1% isoflurane. After the patients were stabilized, the intervention was performed by a single otolaryngologist who was blinded to the study. The patients received either 0.5% bupivacaine (Astra Zeneca, France) 1 mg/kg with adrenaline 1: 200000 or an equivalent volume of saline (control group) in the lateral and upper areas of peritonsillar space. Prescriptions were provided by an anesthesiologist according to the randomization protocol.
The snare‐dissection technique for tonsillectomy was performed by a board‐certified otolaryngologist. Following the completion of the surgery, the effects of the administered neuromuscular drug were reversed using a combination of neostigmine (0.05 mg/kg) and atropine (0.02 mg/kg). Once the patients met the requirements for weaning, they were extubated. After extubation, the patients were transferred to the recovery room and laid in the left lateral position.
Visual analogue score assessment was performed by the patients per instructions. If VAS was more than 3, 0.25 mg/kg pethidine was prescribed. In case of PONV, ondansetron 0.1 mg/kg was prescribed.
The primary outcome was pain intensity measured based on VAS score and was measured in recovery period on admission to the recovery room as 0 and at 1, 2, 4, 8, 12, and 24 hours after surgery. The secondary outcome was PONV
The duration of being pain-free, total pethidine consumption, the time of drinking liquids, and the time of eating soft foods were recorded. As the VAS is a self-reported measure, the pain intensity levels reported by the patients were subjective. In this measure, the pain was measured using a visual analogue pain scale, where 0 represents no pain, and 10 represents unbearable pain. The amount of nausea and vomiting experienced by the patients was evaluated utilizing the post-operation nausea and vomiting scale; a PONV of 0 indicates no nausea or vomiting, 1 nausea but no vomiting, 2 vomited once in 30 minutes, and 3 as two or more episodes in 30 minutes (9).
Patients were discharged in the afternoon if the surgery was performed in the morning, and patients were discharged the next day if the surgery was performed in the evening. For their return home, the patients were prescribed acetaminophen elixir 15 mg/kg of body weight at 4-hour time intervals, but if required, the patients could take it as required for analgesia. Furthermore, after their return home, the patients were contacted by the researchers to obtain a VAS on their first postoperative day to assess pain levels during eating and drinking.
2.6. Statistical Analysis
To analyze the data, SPSS version 22 and GraphPad Prism 8.4.2 were used. For comparisons between the groups, we used chi-squared test or Student's t-test. To ascertain the correlation between post-operation time to the first food ingestion and post-operation pain levels, we used unpaired Student's t-test. The significance threshold was set at ≤ 0.05.
3. Results
During the study period, 96 patients were enrolled in our study. There were no significant differences between the groups regarding demographic information, duration of surgery, and anesthesia (Table 1).
| Variables | Bupivacaine Group (n = 48) | Control Group (n = 48) | P |
|---|---|---|---|
| Age (y) | 7.83 ± 1.85 | 8.38 ± 2.05 | 0.17 |
| Sex (m/f) | 29/19 | 29/19 | 0.58 |
| Weight (kg) | 26.77 ± 7.10 | 27.00 ± 9.24 | 0.89 |
| Duration of surgery (min) | 35.97 ± 9.85 | 45.52 ± 13.99 | 0.80 |
| Duration of anesthesia (min) | 57.50 ± 10.15 | 67.81 ± 16.72 | 0.10 |
aValues are expressed as mean ± SD unless otherwise indicated.
bThe statistical tests used were t-test for continuous data and chi-square test for qualitative data.
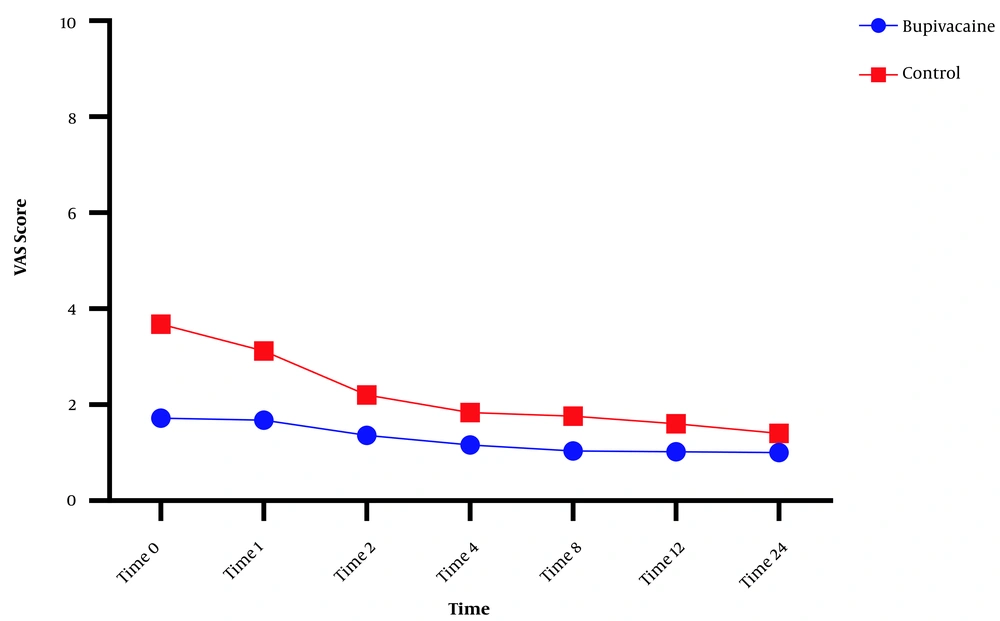
Visual analogue scale score was measured on admission to the recovery room at 0, 1, 2, 4, 8, 12, and 24 hours after surgery, which was significantly lower in the bupivacaine group than in the placebo group (P < 0.001; Figure 2).
Mean visual analogue scale (VAS) score for the bupivacaine and control groups during 24 hours. Data are expressed as mean ± SD. The statistical test used was t-test. *P < 0.05 is considered as significant level. At all times, the mean VAS score in the control group was significantly higher than in the bupivacaine group (P < 0.001).
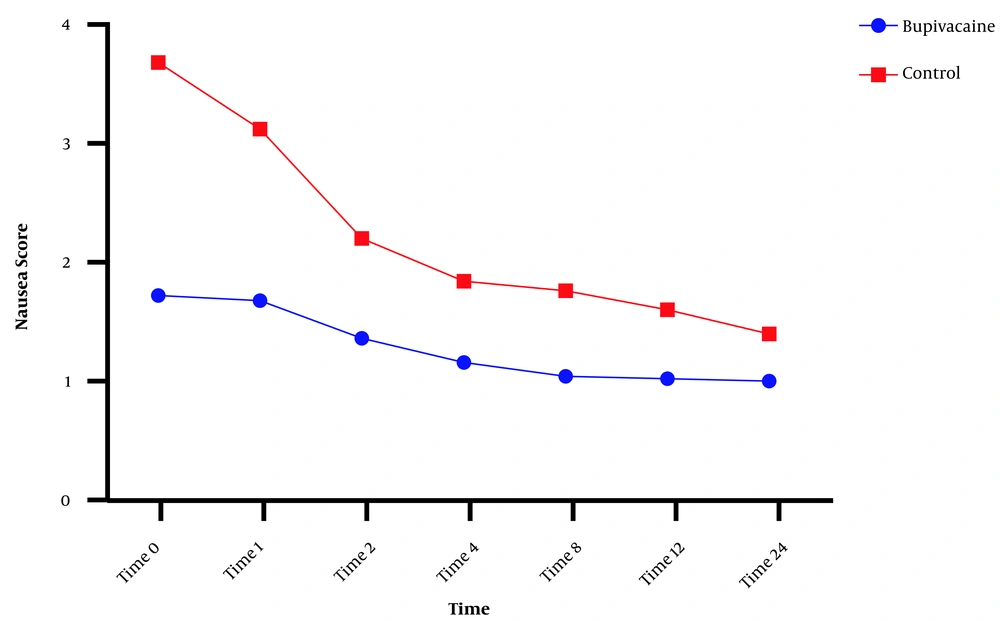
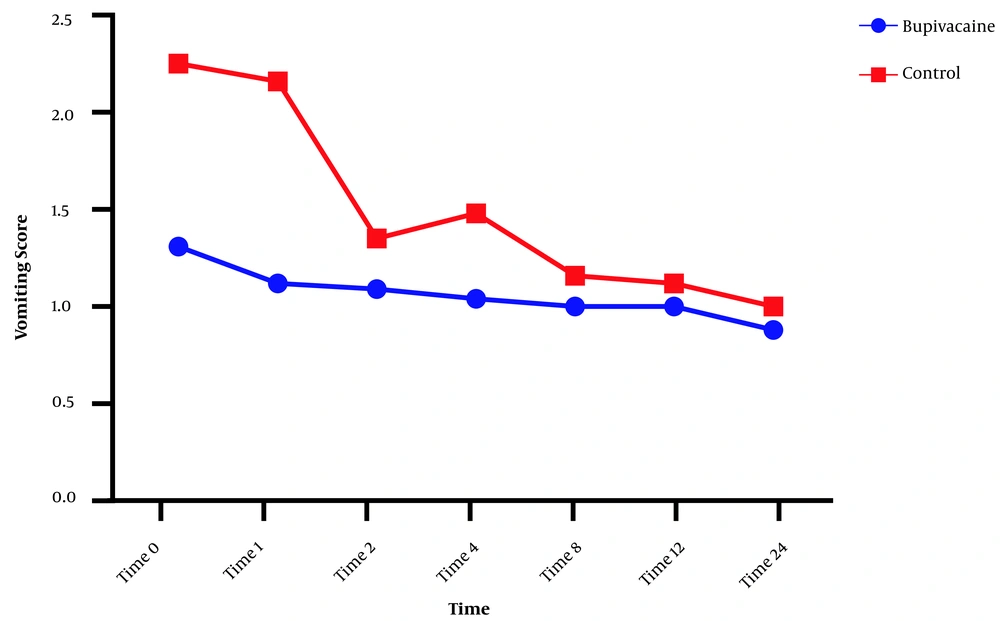
Postoperative nausea and vomiting in the two groups were compared, and significant differences were noted between the groups at all times regarding nausea, and significant differences in vomiting were found between the groups at 0, 1, 2, 4, and 8 but not at 12 and 24 hours after surgery (Figures 3 and 4).
Comparison of nausea between the bupivacaine and control groups. Data are expressed as mean ± SD. The statistical test used was t-test. *P < 0.05 is considered significance level. At all times, the mean incidence rate of nausea in the control group was significantly higher than in the bupivacaine group (P < 0.001).
Comparison of vomiting score between the bupivacaine and control groups. Data are expressed as mean ± SD. The statistical test used was t-test. *P<0.05 is considered as the significance level. The mean score for measuring the incidence of vomiting in the control group was significantly higher than in the bupivacaine group at 0, 1, 2, 4, and 8 h after surgery (P < 0.001).
Time to First Request for analgesic 390.00±3.82 mi in the bupivacaine group vs. 120.86 ± 3.34 in control group P = 0.001.
The average duration of pain loss was 390 min in the bupivacaine group versus 120 min in the control group (P = 0.001). The mean amount of total pethidine consumption was 5.23 ± 3.72mg in the bupivacaine group versus 38.28 ± 23.1 mg in the control group (P = 0.001). The mean total acetaminophen used was 241.87 ± 256.49 mg in the bupivacaine group versus 503.12 ± 412.16 mg in the control group (P = 0.005; Table 2).
| variables | Bupivacaine Group (n = 48) | Control Group (n = 48) | P |
|---|---|---|---|
| Duration of pain-free (min) | 390.00 ± 153.82 | 120.86 ± 123.34 | 0.001* |
| Total petedin consumption (mg) | 5.23 ± 3.72 | 38.28 ± 23.19 | 0.001* |
| Total acetaminophen used (mg) | 112.87 ± 256.49 | 503.12 ± 236.16 | < 0.001* |
Abbreviations: min, minute; mg, milligram.
aValues are expressed as mean ± SD.
bThe statistical test utilized was t-test. *P < 0.05 is regarded as significant.
The pain scores with the consumption of a pliable diet on return home were significantly lower on the first postoperative day in the bupivacaine group than in the placebo group (8.2 ± 1.4 vs 6.3 ± 1.7, respectively; P < 0.05).
4. Discussion
Control of pain and PONV after tonsillectomy is one of great significance for patients because most of these patients are children, and the pain in this group of patients can create an unfavorable mental background. This study aimed to assess the effects of a local injection of bupivacaine on postoperative pain and nausea vomiting after tonsillectomy, and the key result was that this way of administration of bupivacaine had a better analgesic effect and reduced PONV after tonsillectomy; thus, it might be reliably used for postoperative pain relief.
İhvan et al. found that bupivacaine caused significant pain relief at 2 and 6 hours after surgery compared to the control group, but no differences were shown at 24 hours after surgery (9). In our research, however, the pain difference between the two groups continued 24 hours after surgery. This discrepancy may be because of a lower dose of bupivacaine or due to the different areas of injection. While the local effect of bupivacaine continues up to about 6 to 9 hours, such a long effect in our study (about 24 hours) may be due to the fact that the neural block by bupivacaine prevents sending pain impulses to the central nervous system immediately after the operation, reduces excitability resisting treatment, and is responsible for long postoperative pain relief.
Ozmen et al. compared the long-term effects of 0.25% and 0.5% bupivacaine on pain after tonsillectomy. They determined that patients’ pain was reduced even up to one week after taking the drug (7). The results of their study are consistent with our findings.
The results of a study by Kadar and Obaid showed that the use of bupivacaine in tonsillar pills can reduce relief after surgery and provide the possibility of drinking and eating soon after operation (10).
The results of Somdas et al. showed a significant difference in pain relief at the injection side of bupivacaine in the first 8 hours after administration (11). The results of our study were consistent with the above findings.
Postoperative nausea and vomiting is an unpleasant experience that can lead to more serious complications, such as dehydration, electrolyte imbalance, the opening of surgical sutures, increased venous pressure, and so on (12, 13). It also increases intracranial and ocular pressure and hospital costs (and delays the discharge of outpatients and inpatients) (14, 15).
Various studies have examined the effects of various drugs such as intravenous dexamethasone, gabapentin, ketamine, morphine, lidocaine, bupivacaine, and ropivacaine as the main or complementary drugs to reduce pain, nausea, and vomiting. Among them, the effectiveness of lidocaine and bupivacaine was more popular due to their cheapness and availability (16).
In the present study, at all the measurement times, the mean PONV scores were greater in the control group compared to the bupivacaine group (P < 0.05).
Costas-Gastiaburo et al. compared the effect of 0.5% of bupivacaine injection with bupivacaine compound with adrenaline and normal saline on the reduction of morbidities after tonsillectomy. Their results showed the highest incidence of nausea and vomiting among patients treated with bupivacaine alone (71.4%) (17).
They explained that the impact was mostly due to the type of solution chosen. The results of their study did not agree with the present findings, which may be attributed to the differences in dose and combination of injected drugs.
Teunkens et al. compared bupivacaine and tramadol local administration on the pain and PONV after tonsillectomy. They concluded that there was less need to prescribe piritramide in the tramadol group. Furthermore, their study established no significant correlations between the incidence of post-operation nausea and vomiting and antiemetic need or associated complications (18).
Faruk Cicekci investigated the correlation between levobupivacaine and levobupivacaine + adrenaline and pediatric tonsillectomy outcomes. Their results indicated that in both study groups, with the quantity of analgesic consumption 24 hours post-operation being equal, the participants experienced the same post-operation pain levels and PONV rates. In addition, the two groups were consistent in terms of time until the first oral consumption, time taken for recovery, and the time until they requested their first analgesic dose (19). This was consistent with our results.
Ahmed El Daly, in a study with the effect of injection of local anesthetics on pain after tonsillectomy, they found that topical application of the tonsillar bed led to a significant reduction in postoperative throat pain (1).
Regarding side effects, no special complications were observed in using bupivacaine in the peritonsillar area. Bean-Lijewski found an obstruction in the upper airway in the two cases (20). They elucidated that this had been due to very deep injection and the blockage of the dendrites of the recurrent laryngeal nerve. This complication was not secondary to the type of injected drug (bupivacaine), and if any other drug was injected, this side effect could have occurred. The pain intensity and the amount of analgesic consumption of the two groups within 24 after the operation was compared. Duration of being pain-free, petedin need, and the time to start drinking liquids were significantly less in the bupivacaine group than the control group. This was consistent with our results.
4.1. Limitations and Recommendations
The first limitation of our study concerns the side effects associated with the peritonsillar injection of bupivacaine. For future studies, we recommend that other postoperative analgesics that are higher in quality and pose a lower risk of complications be used in tonsillectomy procedures. The second limitation was the lack of understanding of pain scores by some patients.
4.2. Conclusions
Preoperative local injection of 0.5% bupivacaine in tonsillectomy effectively reduced postoperative pain, opioid consumption, PONV in children. We recommend the prophylactic administration of bupivacaine before the beginning of tonsillectomy to achieve adequate postoperative analgesia and lesser PONV.