1. Background
Vitamin D is a steroid hormone which can be produced in our skin under the ultraviolet radiation. Vitamin D plays an important role in the hemostasis of calcium and phosphorus and mineralization of bones (1, 2).
An active form of vitamin D called 1,25-dihydroxyvitamin D3 acts as an immune system moderator to stimulate the innate immune system (3, 4). Deficiency in vitamin D level during pregnancy has been reported between 18% and 84% (5, 6), Several studies in adult patients have shown an association between the occurrence of sepsis and deficiency in vitamin D level (7). Also, some studies on neonates have shown an association between respiratory infections and sepsis and the presence of vitamin D deficiency (5).
Through a complex mechanism, vitamin D enhances immunity by inducing antimicrobial peptides in neutrophils, epithelial cells, and macrophages (8). Vitamin D modulates systemic inflammatory response and locally controls pathogens by helping to inhibit or kill some bacterial strains. Direct invasion of pathogenic bacteria is prevented by vitamin D through enhancing the clearance of these infecting organisms at sites such as respiratory system (9).
However, information about the association between vitamin D deficiency and the occurrence of sepsis in neonates is still inconclusive. Neonatal sepsis indicates bacteremia and its related clinical signs and symptoms within the first month of birth and is an important cause of neonatal mortality. Since the immune system in neonates is not fully matured (10), they are susceptible to developing infections (11-13). Sepsis occurs among 1 to 8 neonates out of 1000 live births. Worldwide, one million neonates die annually due to sepsis (14). Neonatal sepsis is divided into two types based on its timing: early-onset sepsis (EOS) happens within the first seven days of birth, and late-onset sepsis (LOS) happens between the 8th day and the 28th day of birth. Early-onset sepsis is usually caused by microorganisms acquired from the mother. The risks for developing EOS include prematurity, low birth weight, prolonged labor, prenatal asphyxia, pre-mature rupture of amniotic membrane, foul smelling amniotic fluid, or the presence of meconium (11). Another study showed neonates born to mothers with low vitamin D levels and neonates with low cord vitamin D levels were at risk for neonatal sepsis (15).
Neonatal sepsis may leave long-term negative health effects for the child; thus, identifying the associated risk factors and implementing prevention measures may reduce the occurrence of sepsis and its side effects, utilization of NICUs and the associated costs, and separation of mothers and their neonates.
2. Objectives
Due to the role of vitamin D in strengthening the immune system and the high mortality rate among neonates who develop sepsis, this research aimed to evaluate the possible association between serum vitamin D levels in pregnant mothers and their neonates and the incidence of EOS. Also, the association between maternal and neonatal serum vitamin D levels was evaluated.
3. Methods
This prospective case-control observational study included 50 term or late pre-term neonates (≥ 34 - 37 < weeks) who were diagnosed with early-onset neonatal sepsis and were consecutively admitted to NICU in Taleghani Hospital, Tehran, Iran from July to December 2019. The control group included 50 healthy neonates matched for gestational age range (< 37 or ≥ 37 week) and sex from our outpatient clinic. We matched neonates younger than 37 GA weeks in the case and control groups, and then we matched neonates with GA equal or more than 37 weeks from the case and control groups. The criteria for diagnosing EOS included (16) any or a combination of the following clinical signs: respiratory signs (distress, apnea, tachypnea, and hypoxemia), cardiovascular signs (tachycardia and bradycardia), hemodynamic signs (change in skin color, tissue hypoperfusion, and hypotension), neurological signs (irritability, lethargy, hypotonia, low activity, and convulsion), gastrointestinal system signs (poor feeding, abdominal distension (17), and not tolerating the feed), changes in body temperature (fever over 38 Celsius degrees or over 100.4F and hypothermia lower than 36 Celsius degrees or less than 96.8F), or metabolic signs (acidosis or hyperglycemia). The following tests were performed for the neonates diagnosed with EOS: complete blood count, C-reactive protein, blood type, serum vitamin D level, and blood culture. Chest X ray, and in some cases, cerebrospinal fluid analysis and culture were examined. Serum vitamin D levels for the case and control groups were measured. Serum vitamin D level and clinical history for all the mothers were collected.
With the type 1 error of maximum 0.05, the statistical power of 80%, the difference of at least 10 units between the two groups, and the standard deviation of 15 units, the sample size calculated for each group was 35 neonates. The sample size in this study was 50 neonates with EOS and 50 healthy control neonates. After proving the normality of the distribution of the studied variables by Kolmogorov-Smirnov test, to examine the relationship between qualitative variables between the groups, chi-square test or Fisher's exact test and for quantitative variables t-test for normally distributed data and Mann-Whitney test for non-normally distributed data were used. Pearson test evaluated the correlation between serum vitamin D levels in mothers and their neonates. SPSS (IBM version 25) was used for statistical analysis. A P-value of less than 0.05 was considered statistically significant. Patient privacy and medical research ethics were observed.
4. Results
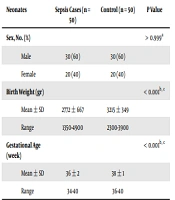
Demographics of 50 neonates diagnosed with EOS and 50 healthy neonates as control group are presented in Table 1. Each group included 30 (60%) boys and 20 (40%) girls. The average birth weight were 2772 and 3215 grams for the sepsis and control groups, respectively, and Student’s t-test showed that the control group had a higher birth weight compared to sepsis cases (P < 0.001) as detailed in Table 1. The mean gestational age for the sepsis cases was 36 ± 2 (range 34 - 40) weeks, and for the control group it was 38 ± 1 (range 36 - 40) weeks (P < 0.001).
aP value was calculated using chi-Square and Fisher’s Exact Tests.
b P value was calculated using t-test.
c P value is less than 0.05 and shows statistical significance.
Table 2 presents the data obtained from the complete blood count and C-reactive protein tests for the sepsis cases. The mean serum vitamin D level for sepsis cases was 49.75 ± 25.53 nmol/L (range 20.47 - 157.25) (or 19.93 ± 10.23 ng/mL, range 8.2 - 63), and for the control group it was 56.41 ± 18.17 nmol/L (range 13.73 - 89.86) (or 22.6 ± 7.28 ng/mL, range 5.5 - 36). Compared to the sepsis cases, the control group had higher serum vitamin D levels (P = 0.022). The list of neonates’ chief complaints is presented in Table 2 and includes: Respiratory symptoms such as grunting or tachypnea, feeding intolerance, mottling or hypoperfusion, and fever together with lethargy and feeding intolerance. In the sepsis group, blood culture for 45 (90%) neonates was negative. Chest X-ray for 14 (28%) of the sepsis cases showed opacity.
| Neonates | Sepsis Cases (n = 50) | Controls (n = 50) | P Value | ||
|---|---|---|---|---|---|
| Mean ± SD | Range | Mean ± SD | Range | ||
| Serum vitamin D (nmol/L) | 49.75 ± 25.53 | 20.47 - 157.25 | 56.41 ± 18.17 | 13.73 - 89.86 | 0.022b |
| Hemoglobin (mmol/L) | 9.77 ± 2.1 | 6.7 - 19.86 | |||
| White blood cell (per microliter) | 14778 ± 6598 | 5100 - 30000 | |||
| Platelets (per microliter) | 227840 ± 87779 | 44000 - 571000 | |||
| C-reactive protein (mg/L) | 17.2 ± 5.1 | 11 - 37 | |||
| Chief complaint, No. (%) | |||||
| Respiratory (grunting, tachypnea) | 32 (64) | ||||
| Feeding intolerance | 8 (16) | ||||
| Mottling, hypoperfusion | 3 (6) | ||||
| Fever, lethargy, feeding intolerance | 7 (14) | ||||
| Blood culture, No. (%) | |||||
| Negative | 45 (90) | ||||
| Positive | 5 (10) | ||||
| Chest X-ray, No. (%) | |||||
| Normal | 36 (72) | ||||
| Opacity | 14 (28) | ||||
a In g/dL unit, hemoglobin values were 15.74 ± 3.39 (range 10.8 - 32).
b P value shows statistical significance since it is lower than 0.05.
Table 3 presents the characteristics of mothers of the neonates in both groups. Serum vitamin D levels were lower in mothers of the neonates with sepsis compared to the control mothers (55.01 ± 22.11 versus 68.24 ± 21.22 nmol/L, respectively; P = 0.005). Also, there was a higher chance that mothers of the sepsis cases delivered through C-section (P = 0.001).
| Maternal Information | Sepsis Cases, (n = 44) | Control Group (n = 46) | P Value |
|---|---|---|---|
| C-section, No. (%) | 34 (77.3) | 19 (41.3) | 0.001b |
| Vaginal delivery, No. (%) | 10 (22.7) | 27 (58.7) | 0.00052b |
| Gravid, No. (%) | |||
| 1 | 16 (36.4) | 27 (58.7) | 0.034b |
| 2 | 22 (50.0) | 15 (32.6) | 0.0927 |
| 3 | 6 (13.6) | 4 (8.7) | 0.4533 |
| Maternal BMI (Kg/M2) | 0.087 | ||
| Mean ± SD | 28.41 ± 1.71 | 27.8 ± 1.61 | |
| Range | 24 - 32 | 25 - 32 | |
| Maternal age (y) | 0.193 | ||
| Mean ± SD | 31.07 ± 6.09 | 29.59 ± 4.56 | |
| Range | 19 - 51 | 22 - 45 | |
| PMH negative, No. (%) | 28 (63.6) | 32 (69.6) | 0.704 |
| IVF, No. (%) | 4 (9.1) | 3 (6.5) | 0.6527 |
| Gestational diabetes mellites, No. (%) | 9 (20.5) | 6 (13.0) | 0.3472 |
| Hypothyroidism, No. (%) | 3 (6.8) | 5 (10.9) | 0.4965 |
| Vitamin D in maternal serum (nmol/L) | 0.005b | ||
| Mean ± SD | 55.01 ± 22.11 | 68.24 ± 21.22 | |
| Range | 13.48 - 157.75 | 23.96 - 124.8 |
Abbreviations: BMI, body mass index; SD, standard deviation; IVF, in vitro fertilization; PMH, past medical history.
at-test showed that mothers in the control group had higher vitamin D levels in serum.
b P value shows there is statistical significance since it is lower than 0.05.
As Table 4 displays, evaluating the serum vitamin D levels in neonates with sepsis and their mothers failed to show any correlation between maternal and neonatal serum vitamin D levels (R = 0.241, P = 0.115). However, for the control group, neonatal and maternal serum vitamin D levels were positively correlated, and neonates’ serum vitamin D levels followed maternal serum vitamin D levels (R = 0.731, P < 0.001).
| Maternal Serum Vitamin D Level | R | P Value |
|---|---|---|
| Sepsis cases serum vitamin D level | 0.241 | 0.115 |
| Control group serum vitamin D level | 0.731 | < 0.001b |
a There was a correlation between serum vitamin D levels in healthy control neonates and their mothers.
bP value lower than 0.05 shows statistical significance.
5. Discussion
Our study showed that neonates experiencing EOS alongside their mothers had lower serum vitamin D levels compared to the control group. Another study has similarly reported that serum vitamin D level was lower in neonates with sepsis (18). Although the case-control study was designed for matched pairs within GA ranges, the sepsis group happened to have a bit lower gestational age, which may have resulted in lower birth weight compared to the control group. Despite the high importance of matching in case-control studies, this type of research bias may still happen in small sample sizes like ours, which may necessitate some compromise in the precision of matching criteria. For example, for GA we used a range rather than an exact number. Yet, in larger sample sizes, there is a higher chance of finding exact matching pairs for case-control studies.
However, unlike our results, an observational study showing similar results for vitamin D levels did not find any difference between neonates with sepsis and healthy control neonates in terms of birth weight (19). Also, contrary to a study by Say (20) who found no significant correlation between vitamin D level in the cord blood and the risk of neonatal sepsis in premature neonates, other studies by Agraval et al. and Dhandai et al. have similarly reported lower serum vitamin D levels in neonates with sepsis although they had studied late-onset sepsis (21, 22). In our study, among neonates with sepsis, there was a higher chance of being delivered through C-section compared to the control group. A study showed no difference between neonates with sepsis and healthy control neonates regarding their mode of delivery (19). Similar to our study, Ozdemir and Cag and Dhandai et al. found lower levels of vitamin D in the serum of mothers of neonates who developed sepsis (21, 23).
Our study showed that the serum vitamin D level in mothers was positively associated with the serum vitamin D level in their healthy neonates. This finding was supported by a study by Cetinkaya et al. (18).
Additionally, Dhandai et al. studying late-onset sepsis reported an association between serum vitamin D levels in mothers and their neonates even when the neonate developed sepsis. Yet, we only could find this association between healthy control neonates and their mothers. They also found that neonates’ low serum vitamin D level was associated with a higher incidence of sepsis, and as a result, showed that maternal serum vitamin D level affected the incidence of sepsis (21). However, our findings failed to show any association between maternal and neonatal vitamin D levels among sepsis cases perhaps due to the limited sample size. Another reason may be that we evaluated maternal vitamin D levels of the sepsis cases during the last trimester of pregnancy and not necessarily in the last days of pregnancy or after delivery. Thus, perhaps some mothers might have taken vitamin D supplements.
Other studies have not considered these effects in their reports (21).
A study by Saboute et al. showed that higher levels of vitamin D in maternal serum reduced the incidence of neonatal sepsis (24). Although our data failed to show any association between serum vitamin D levels in maternal and neonatal samples, sepsis cases had lower levels of serum vitamin D similar to the above study. Also, Saboute’s study showed a positive association between increased maternal age and higher prevalence of sepsis in neonates. Our data could not find these associations. Additionally, they found a positive association between a lower Apgar score and a higher prevalence of sepsis, but our study did not include that data.
Vitamin D Receptors are widely expressed in immune system cells and epithelial tissues (23). Vitamin D, especially its active metabolite 1,25 dihydroxyvitamin D3, plays an important role in calcium hemostasis and immune system activity (25). Demand for calcium increases during the last trimester of pregnancy, such that about 30 grams of calcium is removed from maternal skeleton by the hormonal effects to make the neonatal skeleton. Thus, receiving vitamin D and calcium is important during pregnancy for fetal hemostasis, bone growth, and mineralization (26, 27). There is still no agreement regarding the optimum level of vitamin D in pregnancy and in neonates, but 11 nanograms per milliliter (ng/mL) in the neonate’s serum prevents rickets, and serum level above 30 ng/mL is needed to prevent secondary hyperthyroidism and increase in calcium absorption. Thus, above 30 ng/mL is ideal and below 20 ng/mL shows deficiency (28, 29). Other studies evaluated if low vitamin D levels may cause intrauterine growth retardation, low birth weight, or EOS. Sufficient maternal vitamin D levels may boost maternal and neonatal immune systems (30-32). Supplemental vitamin D during pregnancy may help reduce the occurrence of EOS in term neonates (18, 33).
Our study limitations included the limited sample size that may highlight differences between groups’ averages. In our relatively small sample, we did not have the option to find the exact matching criteria for GA, and consequently, birth weight. Hence, we matched the neonates based on a GA range rather than an exact number. A larger sample size may make finding the exact matching pairs achievable to better identify the possible association between the serum vitamin D levels in neonates and their mothers for both case and control groups. Also, data stratification based on possible confounding variables, such as birth weight, GA, and route of delivery, in a larger study may better identify the strength of the association of serum vitamin D level with the prevalence of EOS. We were not able to match the case and controls for maternal vitamin D supplement consumption since that data was not recorded. Future research needs to address this factor and control for supplement consumption.
5.1. Conclusions
Development of EOS may be associated with vitamin D deficiency in maternal and neonatal serum. Neonates who developed EOS also had a lower birth weight and higher chance of being delivered through C-section. For healthy neonates, there was a positive correlation between serum vitamin D levels in mothers and their neonates. Future studies may need to evaluate the effects of taking vitamin D supplements by pregnant women who have vitamin D deficiency on the incidence of EOS.