1. Background
Children with diabetes mellitus frequently have emotional and behavioral problems (1, 2). An increased rate of behavioral problems such as anxiety problems (1) and depression (3) has been reported in a group of T1DM children compared to a matched control. Globally, governments imposed social restrictions during the COVID-19 pandemic. This restrictive policy, in turn, limited access to health facilities, which was worrisome for hospitalized patients' families and was likely a factor influencing the metabolic control in T1DM patients (4). Poor metabolic control is a risk factor for behavioral problems and an increased risk for complications (5, 6) and mortality (7). The impacts of social restrictions during the COVID-19 pandemic on behavioral problems and metabolic control in T1DM children are still controversial.
Type 1 diabetes mellitus patients require regular insulin injections and continuous blood sugar monitoring during life. The long-life treatment may cause negative emotion-induced maladaptive behaviors and lead to the behavioral and functional problems as well as the risk of suicide attempts (1, 8-10). However, poor metabolic control is indicative of poor compliance, which is a risk factor for behavioral disorders (11). The correlation between behavioral problems and metabolic control is still controversial (12, 13), especially during the COVID-19 pandemic. Therefore, it is necessary to analyze the behavioral problems among T1DM children with good and poor metabolic control during the COVID-19 pandemic.
2. Methods
This cross-sectional study was carried out from January to August 2021 in the endocrine outpatient clinic of General Hospital Dr. Soetomo, Surabaya, Indonasia. The study was approved by the ethics board of General Hospital Dr. Soetomo Surabaya under 0246/KEPK/VIII/2021, and was performed in accordance with the Helsinki Declaration.
A total of 27 patients diagnosed with T1DM based on the American Diabetes Association (14), aged 4 - 17 years, and routinely controlled at the Pediatric Endocrinology Outpatient Clinic were included in this study. Patients who had life-threatening conditions and were hospitalized in pediatric intensive care unit were already excluded. This study sample size was determined using the sample calculation formula for a cross-sectional study (15).
The patient's age, gender, and level of HbA1C were evaluated. Behavioral problems were assessed using an Indonesian version of the Pediatric Symptom Checklist 17 (PSC-17) questionnaire completed by the subject's parents. The PSC-17 questionnaire was sent via google-form, and the subject's parents were guided by the research team via phone or zoom meetings. Inform consent was sent by short messages to the subject's parents, and it was printed out, signed, and returned to the author by them.
PSC-17 is a 17-question short screening questionnaire that helps identify and assess changes in children's emotional and behavioral problems. The Indonesian version of this questionnaire has high reliability (α = 0.821) and validity (P < 0.05) (16). Each answer is scored (0 - 2) according to the following question to make a total score suspect behavioral problems if the sum of internalizing values ≥ 5, externalizing ≥ 7, attention values ≥ 7, and if the total score of PSC-17 was ≥ 15 (16, 17). In this study, T1DM patients were divided into two groups, including patients with good metabolic control group (HbA1C > 8) and poor metabolic control group (HbA1C ≤ 8) (16, 17). Glycosylated hemoglobin (HbA1C) reflects the average glucose level for the last 5 - 8 weeks and indicators of metabolic control in diabetic patients.
Collected data were analyzed using Statistical Product and Service Solution (SPSS) version 18.0. Descriptive data were presented as mean ± SD, while the number of cases and percentages were shown for categorical data. Categorial variables were calculated using Pearson or Spearman analysis based on a normality test with P-value ≤ 0.05 and 95% confidence interval (CI).
3. Results
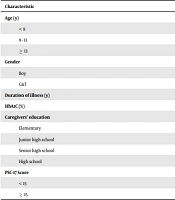
Our study subjects were 27 children with T1DM including 17 boys and 10 girls, aged 12.63 ± 3.39 (95% CI 11.29, 13.97) years. The baseline characteristics of T1DM patients are presented in Table 1. The average duration of illness was 5.41 ± 3.61 (95% CI 3.98, 6.84) years, and the average HbA1c in our study was 10.29 ± 2.39 (95% CI 9.34, 11.24) %. The average HbA1C level among Asian children was 9.1% (18).
| Characteristic | No. (%) | Mean ± SD |
|---|---|---|
| Age (y) | 27 (100) | 12.63 ± 3.39 |
| < 8 | 3 (11.1) | |
| 8 - 13 | 8 (29.6) | |
| ≥ 13 | 16 (59.3) | |
| Gender | ||
| Boy | 17 (63) | |
| Girl | 10 (37) | |
| Duration of illness (y) | 27 (100) | 5.41 ± 3.62 |
| HbA1C (%) | 27 (100) | 10.29 ± 2.39 |
| Caregivers' education | ||
| Elementary | 0 | |
| Junior high school | 5 (18.5) | |
| Senior high school | 16 (59.3) | |
| High school | 6 (22.2) | |
| PSC-17 Score | 27 (100) | 8.70 ( ± 4.75) |
| < 15 | 18 (66.7) | |
| ≥ 15 | 9 (33.3) |
Demographic Characteristics of Patients with T1DM
Nine out of 27 subjects had abnormal PSC-17 scores (≥ 15), and 18 out of 27 ones were within the normal score range (< 15). The average level of HbA1c in the abnormal score group of PSC-17 was 9.43 ± 3.52 (95% CI 0.68, 18.19) %, while the level of HbA1c in the normal score group of PSC-17 was 10.4 ± 2.31 (95% CI 9.43, 11.37)%. No association was detected between behavioral problems with the duration of illness (P = 0.91) and age (P = 0.37).
Table 2 shows the correlation between behavioral problems and metabolic control. Four out of 27 subjects (14.8%) had internalizing problems, and five out of 27 ones (18.5%) had externalizing problems, but none of them had attention problems. A comparison of behavior problems between the good and poor metabolic control groups showed no significant difference regarding the internalizing or externalizing problems (P = 1.00 and P = 0.14).
| Behavioral Problems (PSC-17 Score) | HbA1C Level | P-Value | |
|---|---|---|---|
| ≤ 8 (n = 8) | > 8 (n = 19) | ||
| Internalizing | 1.00 | ||
| ≥ 5 | 1.8 (12.5) | 3.19 (25.8) | |
| < 5 | 7.8 (87.5) | 16.19 (84.2) | |
| Externalizing | 0.14 | ||
| ≥ 7 | 3.8 (37.5) | 2.19 (10.5) | |
| < 7 | 5.8 (62.5) | 17.19 (89.5) | |
| Attention | - | ||
| ≥ 7 | 0.8 (0) | 0.19 (0) | |
| < 7 | 8.8 (100) | 19 (100) | |
Correlation Between Behavioral Problems and Metabolic Control
5. Discussion
In this study, no significant difference was found between the good and poor metabolic control groups in terms of behavior problems. During the COVID-19 pandemic, 33.3% of the children with T1DM had behavioral problems. This result was consistent with the findings from previous study in 2009, where the rate of behavioral problems was assessed in T1DM patients aged 17 - 19 years, and the rate of 33.3% was recorded for the participants regarding behavioral problems, which was three times higher than that recorded for the control group (9.7%) (13). Another study on Indonesian children with T1DM before the pandemic also showed that 45.8% of the children had behavior problems (2). A recent study found no differences between the diabetic group (n = 132) and the healthy group (n = 131) over three years in terms of psychosocial function such as anxiety, depressive symptoms, anger, or behavioral difficulties (3). Yet, the diabetic group showed a greater decline in social acceptance and a higher rise in disturbed eating behavior. However, the study about behavioral problems in children with T1DM during COVID-19 Pandemic is limited.
According to our study results, the most common behavioral problems among 27 participants were externalizing (18.5%) and internalizing problems (14.8%). Furthermore, the frequency of externalizing problems among participants with good metabolic control was higher than that of internalizing problems. It was shown that T1DM children with good metabolic control mostly had externalizing problems during the COVID-19 pandemic. This result was inconsistent with the finding from a previous study that had found externalizing behavioral problems in poorer glycemic control (19). Another study showed that unstable blood glucose was related to psycho-behavioral changes in T1DM children (20, 21). The presence of externalizing problems is associated with four factors: the aspects of the child development, sociocultural factors, parenting and caregiving experiences, and peer-group experiences (22). Children's peer experiences in school are important factors negatively contributing to the development of the externalizing problems. Since school activities were limited during the COVID-19 pandemic, children did not have peer experiences, nor did they have unlimited access to medical services, and people had to stay home; all these reasons seemed to be associated with the presence of externalizing problems in this study.
A longitudinal study by Butwicka et al. indicated that children with T1DM had a three times greater risk factor than healthy children regarding behavioral problems, especially in families with psychiatric disorders. It was also observed that T1DM children with behavioral problems made 1.7 times more suicide attempts than children with T1DM who had no comorbid (OR 1.7; 95% CI 1.4 - 2.0) (1). The study by Bernstein et al. demonstrated that the prevalence of mental disorders in children with T1DM were as follows: 31.9% with anxiety disorders, 19.1% with behavioral problems, and 14.2% with mood disorders in the adolescent population (23). Several other studies have identified a relationship between T1DM and various psychosocial conditions, including anxiety disorders, eating disorders, behavioral disorders, mood disorders, and depression (23). These behavioral problems were common in T1DM children and negatively contributed to the disease management and metabolic control.
In our study, no significant association was detected between the level of control metabolic and behavioral problems, and no relationship was observed between behavioral problems with the duration of illness and age. Another large population study based on Danish National Patient Register (NPR) found the highest risk of psychiatric comorbidity among T1DM children diagnosed at ages 10 to 14 years and with five years of diabetes (24, 25). This study also suggested that T1DM children with behavioral disorders had higher mean age than children with non-behavioral disorders. Several studies revealed that behavioral problems (19, 25, 26) and higher stress levels (26, 27) affected the course of the illness and the level of metabolic control. However, other studies failed to find a significant correlation between the level of metabolic control and behavioral problems (13, 28).
Globally, the incidence of T1DM is increasing, and it is anticipated that over 90,000 children are afflicted with T1DM annually (29). In 2018 in Indonesia, 1,220 children and adolescents with T1DM were identified (30). From 2000 to 2010, T1DM incidence in Indonesia increased sevenfold from 3.88 to 28.19 per 100 million population (31). Our study found that the mean HbA1C value was 10.29 ± 2.39% during the COVID-19 pandemic. The previous research by Dr. Soetomo showed that the mean HbA1c was 10.06 ± 2.49% during the first year of the COVID-19 pandemic, while it was 9.20 ± 1.86% before the lockdown (10). It was shown that metabolic control among children with T1DM was deteriorated during COVID-19. The deterioration of the metabolic control during the lockdown has been found associated with the several factors, such as the caregivers refusal to take their children to the hospitals due to the fear of the contagious virus, limited transportation, and limited health care. This reason negatively impacts metabolic control in patients with diabetes, especially T1DM, who are on insulin therapy. Our result in this regard was in line with the finding from previous study during the COVID-19 pandemic, suggesting that the impact on glycemic control among 52 patients with T1DM was 19 (36.5%) and had hyperglycemic episodes; among them, 4 (7.7%) landed into DKA (Diabetic Ketoacidosis). Eight out of 19 patients had hyperglycemia but had no insulin injections during the lockdown due to nonavailability (4). The mean HbA1C of pre-lockdown and lockdown phases were 8.8 ± 1.3% and 10 ± 1.5%, respectively, and the difference was statistically significant (P < 0.001) (4). However, another study in Italy revealed that metabolic control of adolescents with T1DM was not deteriorated during the restrictions due to COVID-19 pandemics, and was improved in those pursuing their physical activities during the quarantine (32).
Another study that investigated the effects of the COVID-19 pandemic on the treatment of 7000 T1DM children from Sweden's national database (SWEDIABKIDS) found that the proportion of patients with good metabolic control remained unchanged in 2019 and 2020 (33). This may have been attributed to the fact that Sweden, compared to other countries, had an active treatment of T1DM with low mean HbA1C on a national level, including telemedicine treatment as the alternative combination to regular visits when physical distancing was required during the pandemic (32). In our study, on the other hand, telemedicine was not a routine alternative treatment to preserve the quality of care for T1DM. Therefore, T1DM patients had relatively poorer metabolic control during the COVID-19 pandemic.
As for our study limitations, the behavioral problems was not explored during the pre-lockdown phase. Also, comparing the behavioral problems in the pre-lockdown phase and those in the lockdown phase was not an option at the time of our study. Despite the given limitations, the strength of our study lies in the fact that it was conducted during the critical time of COVID-19 pandemic. Our study results may have been valuable for future studies and caring for patients with T1DM. It was recommended that PSC-17 questionnaire should be used as a useful tool for investigating the behavior problems in children with T1DM, especially in children needing primary care.
4.1. Conclusions
There was no significant difference between the good and poor metabolic control groups regarding the behavior problems (P = 1.00, IP; P = 0.14, EP). The frequency of EP was higher among children with good metabolic control, and the frequency of IP was higher among children with poor metabolic control.