1. Background
25-Hydroxyvitamin D (25(OH) D) is an important fat-soluble steroid hormone that plays an important role in bone metabolism and neuromuscular functions (1). Moreover, 25 (OH) D has anti-proliferative, pro-apoptotic, and immunomodulatory properties (2, 3). In recent years, vitamin D deficiency has been noticed in all countries and age groups, including pregnant women and children, and particularly in preterm newborns (4-6).
Low levels of maternal vitamin D during pregnancy may be associated with an increased risk of preeclampsia, gestational diabetes mellitus (GDM), preterm labor, and small for gestational age (SGA) newborn birth. All of these pregnancy conditions may lead to the birth of infants being more prone to VDD and hypocalcemia. Vitamin D deficiency and insufficiency is observed in approximately 90.3% of preterm newborn infants, about 54.3% of whom suffer from VDD. The reason for this high prevalence is that neonates receive more than 70% of their vitamin D needs through the placenta during the second and third trimesters of the pregnancy. Therefore, premature infants are deprived of this source and may suffer from VDD. In premature infants, the lack of this vitamin, in addition to the consequences mentioned above, may lead to an increased risk of rickets of prematurity, respiratory tract infections, and chronic respiratory diseases, such as bronchopulmonary dysplasia, as well as seizures, and growth disorders (7-10).
In neonates, vitamin D3 levels lower than 20 ng/mL are defined as vitamin D deficiency. Serum 25 (OH) D level lower than 10 ng/mL is defined as severe vitamin D deficiency. Serum 25 (OH) D level between 20 and 30 ng/mL is defined as insufficient vitamin D. Vitamin D deficiency (VDD) may lead to metabolic bone disease (MBD) in preterm infant newborns; the prevalence of this disease is about 55% in preterm infants weighing less than 1000 grams at birth (11).
Although treatment of vitamin D deficiency in newborns is still challenging, some groups recommend a minimum 25 (OH) D target of 30 - 32 ng/mL for vitamin D levels in order to prevent the increased risk of possible vitamin D-related morbidities (12). On the other hand, considering reduced respiratory infection and RSV-associated (respiratory syncytial virus-associated) bronchiolitis in infants following pregnancy and infancy vitamin D supplementation in addition to the above-mentioned effects of VDD in newborns, it is recommended to maintain the serum concentration of Vitamin D at sufficient levels (13, 14). In a clinical trial study carried out on Iranian children with VDD, Rahafard et al. concluded that both oral and injectable vitamin D therapy had equal effectiveness in the treatment of this deficiency among children. The results of their study showed that in younger children, injectable form in the short term yielded a better response (15). In a clinical trial performed by Gupta et al., the effectiveness of oral versus intramuscular vitamin D was assessed in adult Indian patients with VDD, and their findings demonstrated that both oral and intramuscular routes were effective for the treatment of VDD. Also, 25-hydroxyvitamin D levels in the injectable cholecalciferol group showed a sustained increase from baseline compared to the oral form (16). In a study in Zanjan city, the average level of vitamin D in mothers’ serum was 19.4 ± 3.9 nmol/Liter, and the level of vitamin D in umbilical cord blood was 16.7 ± 2.9 nmol/Liter. Hypovitaminosis D was diagnosed in 86% of women and 75% of infants in winter as well as in 46% of mothers and 35% of infants in summer. A positive correlation was found between mother’s blood and umbilical cord (r = 0.55 and P < 0.001) (17).
Considering the importance of vitamin D in preterm newborns, and hence we did not find any study evaluating the effects of vitamin D medicinal products, especially the two forms of oral and injectable vitamin D available in Iran, on premature infants who were faced with vitamin D deficiency or insufficiency,
2. Objectives
We aimed to compare the serum levels of vitamin D fifteen days following treatment with oral versus injectable vitamin D.
3. Methods
3.1. Study Design
This randomized clinical trial study was conducted in Shahid Akbarabadi Hospital affiliated with Iran University of Medical Sciences in 2022, on all infants suffering from vitamin D deficiency or insufficiency. All steps of this research were done with the approval of the Ethics Committee and code IR.IUMS.FMD.REC.1397.787 of the Research Assistant of Iran University of Medical Sciences and written permission was obtained from the hospital manager to access the necessary data. The goals and outcomes of the study were explained to the parents of neonates participating in the study, and all the patients were treated with the full consent of their parents. Also, this research was registered on 26/02/2022 in Iran’s clinical trial site with the code identifier IRCT20160120026115N8, http://www.irct.ir/.
3.2. Sample Size
In this study, considering an effect size of 0.6, alpha of 0.05, and beta of 0.20, each group sample size was calculated at 45.
3.3. Inclusion and Exclusion Criteria
Premature infants less than 37 weeks of gestation admitted to the hospital NICU, whose serum vitamin D levels were less than 30 ng/mL on 14th day of life, were included in the study. Exclusion criteria were a history of receiving oral or injectable vitamin D prior to the study, infants affected with malabsorption or thyroid disorder, and infants with low glomerular filtration rate (GFR) for age.
3.4. Sampling and Randomization
Ninety preterm neonates affected with vitamin D deficiency or insufficiency admitted to NICU and visited daily by a neonatologist were selected to enter the study. The patients’ parents received explanations about the study and completed the written informed consent form if they were willing for their neonates to participate in the study. The simple randomization block method was used for random allocation in excel software.
3.5. Intervention
Demographic and clinical data of newborns were collected by a questionnaire. Then, randomly, the aforementioned patients were treated with oral drops of 1,000 units daily. Ultra Vitamin D (1,000 IU or 25μg D3 in 1 mL) manufactured by Tehran Vitabiotics company, Iran, was used for 15 days or a single dose of 15,000 units. Moreover, vitamin D3 (300,000 IU in 1 mL) manufactured by Osve company, Iran, was used intramuscularly at the beginning of the study. Patients were followed up on the 16th day after drug administration by measuring serum 25 (OH) D3, calcium, phosphorus, and alkaline phosphatase (ALP). In order to measure vitamin D serum levels using enzyme-linked immunosorbent assay (ELISA), blood samples were collected from infants, stored and transferred to the laboratory at 0 - 4°C (2).
3.6. Blinding
Those who measured the serum levels of the considered parameters and the statistical analyzer were blinded in the study. But patients and care providers that introduced the drug to newborns were not blinded because of the routes of treatment being oral or parenteral. Therefore, this study is an un-blinding randomized clinical trial.
3.7. Statistical Analysis
SPSS version 22 software was used for statistical analysis of data. The results for quantitative variables were expressed as mean and standard deviation (Mean ± SD) and categorical qualitative variables as percentage. Comparison between quantitative variables was made using t-test, and in case of non-normal distribution, using Mann-Whitney U test. Chi-square test and Fisher’s exact test were used to check the relationship between qualitative variables. Significance level of P-value was considered less than 0.05. No confounder was involved in the study.
4. Results
Among the 90 infants with a gestational age < 37 weeks who were included in the study, 47.78% were boys. There was no significant difference in terms of gender between the patients who entered the study. As shown in Table 1, it was found that the average gestational age and birth weight in the two groups of patients receiving two forms of oral and injectable drug were similar with no significant difference.
| Variables | Oral Group (Mean ± SD) | Injection Group (Mean ± SD) | P-Value |
|---|---|---|---|
| Gestational age (w) | 29.84 (3.09) | 29.06 (1.76) | 0.245 |
| Birth weight (g) | 1265.83 (283/42) | 1164.03 (191.17) | 0.118 |
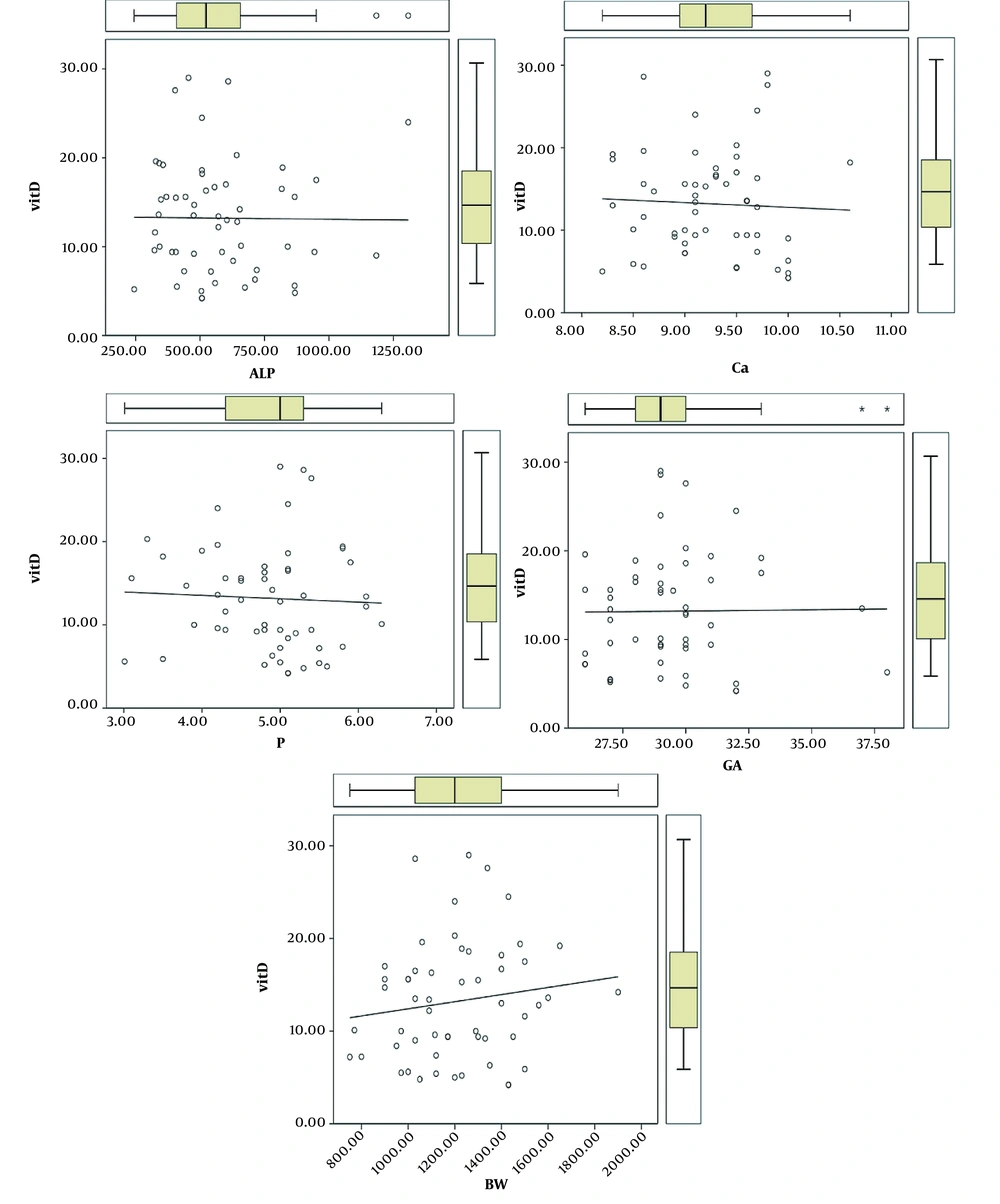
It was found that with an increase in gestational age and birth weight, the initial vitamin D serum levels of patients would move upwards. However, this correlation was not statistically significant. On the other hand, the initial vitamin D serum levels had no significant correlation with the serum calcium, phosphorus, and alkaline phosphatase levels of the patients (Figure 1).
In the group receiving intramuscular vitamin D, the serum level of vitamin D before treatment was equal to 13.56 ± 6.81 ng/mL. Also, the serum alkaline phosphatase level in these patients was 609.54 ± 242.52 mg/dL, phosphorus level 4.90 ± 0.81 mg/dL, and calcium level 9.11 ± 0.54 mg/dL. The level of vitamin D was 36.68 ± 10.48 ng/mL in serum of the patients after the intervention. Furthermore, the serum level of ALP in these patients was equal to 663.90 ± 294.08 IU/L, phosphorus 4.93 ± 0.45 mg/dL, and calcium 9.18 ± 0.49 mg/dL.
The serum vitamin D level before treatment was equal to 12.73 ± 5.87 ng/mL in the group of patients who received oral vitamin D as VDD treatment. Also, the mean level of ALP in these patients was 531.66 ± 177.56 IU/L, serum phosphorus level was 4.73 ± 0.67 mg/dL, and calcium level was 9.40 ± 0.49 mg/dL. The mean serum level of vitamin D was 45.16 ± 11.47 ng/mL in these patients after oral treatment. In addition, the mean level of ALP was 661 ± 191.42 IU/L, phosphorus 4.91 ± 0.49 mg/dL, and calcium 9.21 ± 0.46 mg/dL in these patients. According to Table 2, the serum levels of vitamin D, ALP, phosphorus, and calcium in the patients of both injectable and oral groups were not significantly different before the intervention (P-value > 0.05). Besides, there was no significant difference in serum levels of ALP, phosphorus, and calcium in the patients of two groups of treatment fifteen days after the intervention (P-value > 0.05). However, even though the serum vitamin D levels in the two groups were not significantly different before the intervention, the level of vitamin D was significantly higher in the oral group than in the injection group after the intervention (P-value = 0.006).
| Variables | Before Treatment (Mean ± SD) | After Treatment (Mean ± SD) | ||||
|---|---|---|---|---|---|---|
| Oral Group | Injection Group | P-Value | Oral Group | Injection Group | P-Value | |
| Serum vitamin D (ng/mL) | 12.73 (5.87) | 13.56 (6.81) | 0.637 | 45.16 (11.47) | 36.68 (10.48) | 0.006 |
| Serum ALP(IU/L) | 531.66 (177.56) | 609.54 (242.52) | 0.192 | 661.0 (191.42) | 663.90 (294.08) | 0.967 |
| Serum phosphorous(mg/dL) | 4.73 (0.67) | 4.90 (0.81) | 0.373 | 4.91 (0.49) | 4.93 (0.45) | 0.863 |
| Serum calcium(mg/dL) | 9.40 (0.49) | 9.11 (0.54) | 0.411 | 9.21(0.46) | 9.18 (0.49) | 0.816 |
5. Discussion
In the present study, we assessed the serum levels of vitamin D3 two weeks after oral and injectable drug administration and compared the rise of vitamin D levels in the two mentioned methods. In this study, authors found that although the mean serum level of vitamin D was higher in the group receiving oral vitamin D, the route of receiving the drug in infants did not affect the therapeutic effectiveness as an independent factor. The findings of the study demonstrated that both oral and injectable treatments raise vitamin D levels to sufficient ones. Although it rises more in the oral group, it remains in sufficient range. The level of vitamin D was 13.20 ± 6.37 ng/mL in premature infants suffering from VDD in our study. In Park et al.’s study on 278 cases of premature infants, with an average gestational age of 33 ± 2 weeks, they found that the level of vitamin D was 10.7 ± 6.4 ng/mL and 91% of the babies had VDD (18). Also, in a similar study by Dawudo and Nath, the average vitamin D level was 16.3 ng/mL in premature infants (19). These findings emphasize the importance of screening for vitamin D deficiency in premature babies. In the present study, the average gestational age and birth weight were similar in the two groups receiving the drug. Also, the serum levels of vitamin D, ALP, phosphorus, and calcium were not significantly different in the patients of the injection and oral groups before the intervention (P-value > 0.05). On the other hand, the serum levels of ALP, phosphorus, and calcium in the patients of the injection and oral groups were not significantly different fifteen days after the intervention (P-value > 0.05). In Park et al.’s study, as in our study, serum calcium and phosphorus levels in premature infants suffering from vitamin deficiency did not show significant changes. However, a significant increase was observed in ALP levels (18).
In this study, as gestational age and birth weight increased, patients’ initial vitamin D levels increased. However, this correlation was not statistically significant. Also, the initial vitamin D level did not significantly correlate with the serum calcium, phosphorus, and ALP levels of the patients (P-value < 0.05). In the study by Terek et al. in 2018, vitamin D level was not related to gestational age and birth weight (20). Burris et al. also did not observe any linear relationship between vitamin D level and gestational age (21). In this way, Park et al. reported an insignificant correlation between vitamin D levels with gestational age and birth weight (18). However, in this study, even though there was no significant difference between the serum vitamin D levels in the two groups before the intervention, the level of vitamin D was significantly higher in the oral group than the injection group after the intervention (P-value = 0.006). One of the reasons for the higher level of vitamin D after oral treatment compared to injection may be the type and speed of the absorption of the oral treatment. That is, studies show that when using an oral supplement compared to an injection, the increase in the serum level of 25 (OH)D is faster but more transient (22, 23). On the other hand, based on the one-way covariance analysis, the use of each treatment method independently did not change the treatment effect (P-value = 0.073), which means that the use of either the injectable or oral drug, comes with no difference in terms of effectiveness.
In some studies, the level of vitamin D was higher in the injection method. For example, in the study of Billoo et al. in 2009, although both injectable and oral forms of vitamin D supplementation in infants increased vitamin D levels close to the normal range, injectable vitamin D performed better in this regard (24). Also, in the study of Tellioglu et al. in 2012 on elderly people over 65 years old, although both oral and injectable methods had a good effect in raising the serum vitamin D level, the injectable type increased it slightly more (25). However, some studies indicate the effectiveness of the two treatment methods. In this regard, Wylon et al.’s study demonstrated that both injectable and oral supplementation had the same efficacy and comparably increase serum vitamin D levels (26). It seems that the difference in the findings of these studies is due to the difference in the time of evaluating the patients after treatment and the drug doses used for infants. Ultimately, the effectiveness of both treatment methods in case of continued oral use and correct injection use were similar. According to Wylon et al.’s study, the highest level of vitamin D after treatment in both therapeutic methods was on the 28th day after consumption, which were also quantitatively similar (26).
There were no side effects caused by receiving the drug in any of the injection and oral treatment groups. Therefore, considering the similar effectiveness of both treatment methods, it seems that the main factor in choosing a treatment method by physicians is the patient’s clinical condition. For example, in case of food intolerance or contraindication to oral intake or intestinal malabsorption, intramuscular injection is the preferred route of administration (27, 28). Small sample size and absence of long-term follow-up are the limitations of the study. It is suggested to evaluate the level of vitamin D in a larger group of premature infants treated with supplements intermittently and with longer intervals to obtain more reliable results.
5.1. Conclusions
The results of our study showed that in premature infants, the administration of vitamin D orally or by injection, both significantly increases the serum concentration of 25 (OH)D to sufficient levels. Therefore, both treatment alternatives can be used safely in practice. Depending on the drug compliance in patients or the presence of digestive and absorption disorders, the injection method can be used as the preferred method.