1. Background
Phenylketonuria (PKU) is an inherited genetic disorder characterized by a partial or total deficiency of the enzyme phenylalanine hydroxylase, which is responsible for converting phenylalanine (PHE) into tyrosine. Consequently, there is an accumulation of PHE in the child’s body (1-4).
Early initiation of treatment for this condition, following neonatal screening, is highly recommended. The cornerstone of treatment primarily involves implementing a PHE-restricted dietary regimen (5-7). The main objectives of this dietary approach are to support normal growth and development, maintain PHE levels within safe limits to prevent mental retardation in affected children, and ensure the necessary metabolic processes (8, 9).
The protein-restricted diet in individuals with PKU affects their ability to meet the necessary protein requirements for growth and development. The increasing prevalence of childhood overweight and obesity is a concern since it predicts the persistence of these malnutrition disorders later in life. Approximately 50% of infants who are overweight at six months old and 80% of children who are obese at five years old will remain overweight in adulthood (10). In addition to specific health risks such as type-2 diabetes mellitus, cardiovascular disease, dyslipidemia, hypertension, respiratory dysfunction, several cancers, and nonalcoholic fatty liver disease, obese boys may experience issues like a small penis, overactive bladder, poor body image, decreased quality of life, anxiety, and reduced physical activity (11, 12).
Concerning children with PKU, several reports suggest an elevated BMI in PKU patients compared to normally developing children, although the underlying causes remain a topic of debate (12-14). The carbohydrate-rich diet, which is limited in terms of food variety, may influence food preferences and choices, potentially contributing to adult obesity (15). Studies have indicated that children with PKU consume less protein and fat but higher amounts of carbohydrates. Additionally, it has been observed that children with PKU have a higher daily glycemic index than their healthy counterparts (16).
Previous studies have reported increased rates of malnutrition disorders in children with PKU. For instance, Holm et al. observed a trend toward obesity in children with PKU when comparing the overweight status of 124 children with PKU to that of normally developing children (17). Similarly, Allen et al. suggested that alterations in body composition could make children with PKU more prone to obesity (17-19). However, few studies have explored the association between overweight/obesity prevalence in children and PHE levels. Furthermore, the relationship between lipid profiles, nutritional status, and PHE levels has been rarely discussed. Hence, our study aims to address these gaps.
2. Objectives
Based on the information above, the present study builds upon the previously reported trend of overweight and obese individuals with PKU. Its objective is to determine the prevalence of obesity and overweight in children diagnosed with PKU and establish correlations between their nutritional status, PHE levels, and lipid profiles.
3. Methods
This cross-sectional study included 47 children diagnosed with PKU who were receiving care at the nutritional clinic of the National Nutrition Institute, Cairo, Egypt, over one year from July 2021 to June 2022. The anthropometric assessment involved measuring weight, height/length, and body mass index (BMI). A biochemical nutritional evaluation was also conducted, including fasting lipid profile and fasting blood sugar measurements. Inclusion criteria comprised children diagnosed with PKU whose parents or caregivers provided consent for participation. Exclusion criteria encompassed any other metabolic disorders and children with comorbid conditions.
3.1. Operational Definitions
(1) Overweight: In children up to 2 years of age, or between 2 - 5 years, overweight is defined as weight for length or height exceeding the mean by 2 standard deviations (SD) according to the World Health Organization (WHO) Child Standards for Growth. For children aged 5 - 19 years, overweight is defined as a BMI z score exceeding 1 SD of the WHO Child Standards for BMI based on WHO charts.
(2) Obesity: In children up to 2 years of age, or between 2 - 5 years, obesity is defined as weight for length or height exceeding the mean by 3 SD according to the WHO Child Standards for Growth. For children aged 5 - 19 years, obesity is defined as a BMI z score exceeding 2 SD of the WHO Child Standards for BMI based on WHO charts.
(3) Wasting: In children up to 2 years of age, or between 2 - 5 years, wasting is defined as weight for length or height falling below the mean by 2 SD according to the WHO Child Standards for Growth. For children aged 5 - 19 years, wasting is defined as a BMI z score falling below 2 SD of the WHO Child Standards for BMI based on WHO charts.
(4) Underweight: In children, underweight is defined as weight for age falling below the mean by 2 SD according to the WHO Child Standards for Growth.
(5) Stunting: Stunting is defined as length/height for age below the mean by 2 SD of the WHO growth charts.
(7) Reference ranges for pediatric phenylalanine levels and lipid profiles were utilized to establish abnormal levels (23).
3.2. Outcome Measures
The main goal was to assess the prevalence of overweight and obesity among children diagnosed with PKU.
The secondary objectives were to examine the pattern of lipid profiles and fasting sugar levels among the children and to establish correlations between PHE levels and nutritional status as additional secondary outcomes.
3.3. Ethical Concerns
The research protocol underwent review and approval by the Ethical Committee of the General Organization of Teaching Hospitals and Institutes (GOTHI), with an ethical approval number of IN000115. The study was conducted in adherence to the principles outlined in the Declaration of Helsinki, ensuring ethical standards in medical research involving human subjects. Informed consent was obtained from each parent or caregiver before enrolling in the study, and there were no penalties for those who declined participation.
3.4. Sample Size Estimation
The main objective of this study is to determine the prevalence of overweight and obesity in children with PKU. According to Williams et al., 86% of PKU patients were reported to be overweight and obese (19). Sample size calculation using Epi info software (version 7.2.5.0) indicated that a minimum sample size of 46 patients is required for this study, with an alpha level of 0.05, a confidence level of 95%, and an acceptable margin of error of 10% (19, 24).
3.5. Sampling Method
Patients were enrolled using a consecutive sampling method, whereby every eligible patient attending the nutritional clinic at the National Institute for Nutrition was included in the study until the required sample size was achieved.
3.6. Statistical Analysis
The data were summarized as mean ± SD, median and interquartile range, or frequencies and percentages as appropriate. Non-parametric tests, such as the Mann-Whitney U test, were used to compare numerical variables between study groups due to the non-normal distribution of the data. Correlation analysis was performed using the Spearman correlation coefficient (rs). Categorical data were compared using chi-square (χ2) test or Fisher’s exact test. A two-sided P-value ≤ 0.05 was considered statistically significant. Statistical analyses were conducted using IBM Statistical Package for the Social Sciences (SPSS), version 22 for Microsoft Windows, by IBM Corp, Armonk, NY, USA.
4. Results
Our study included children diagnosed with PKU who were receiving care at the nutritional clinic of the National Nutrition Institute. The mean age of the participants was 5.68 years, with a higher proportion of males (Table 1).
| Patient Data | Value |
|---|---|
| Gender (No. (%)) | |
| Males | 27 (57.4) |
| Females | 20 (42.6) |
| Age | |
| Min. - Max. | 0.1 - 12 |
| Mean ± SD | 5.68 ± 3.47 |
| Median (IQR) | 6 (6.2) |
Anthropometric assessments were conducted, revealing a significant number of malnourished children (n = 34), accounting for 72.3% of the study participants. Among the enrolled children, 55.3% (n = 26) were classified as overweight or obese (Table 2).
| Participants | No. (%) |
|---|---|
| Malnourished (n = 34) a | |
| Underweight and stunted | 2 (4.3) |
| Obese and stunted | 1 (2.1) |
| Overweight and stunted | 2 (4.3) |
| Obese | 21 (44.7) |
| Stunted | 1 (2.1) |
| Wasted | 5 (10.6) |
| Overweight | 2 (4.3) |
| Other children | 13 (27.7) |
| Total Overweight and obese children | 26 (55.3) |
a 72.3% of the study participants.
Phenylalanine levels were examined and correlated with anthropometric results as well as nutritional biochemical findings, including fasting lipid profile and blood sugar. A significant difference was observed in phenylalanine levels between malnourished cases and children with normal anthropometric assessment. The majority of malnourished patients (76.5%) had higher phenylalanine (P = 0.046) levels (mean ± SD = 10.23 ± 6.15) compared to other children (mean ± SD = 6.54 ± 4.9), with a statistically significant difference (P = 0.025) (Table 3).
a Statistically significant at P ≤ 0.05.
b Chi-square test.
c Mann-Whitney U test.
Regarding overweight and obese children, there was a tendency towards higher phenylalanine levels, although this difference was not statistically significant (P = 0.414). There was also no significant difference observed between males and females in terms of phenylalanine levels (Table 4).
| Characteristics | High Level of Phenylalanine | P Value | |
|---|---|---|---|
| No | Yes | ||
| Overweight/obese | 0.414 | ||
| No | 8 (38.1) | 13 (61.9) | |
| Yes | 7 (26.9) | 19 (73.1) | |
| Gender | 0.098 | ||
| Males | 6 (22.2) | 21 (77.8) | |
| Females | 9 (45) | 11 (55) | |
Regarding the correlation between PHE levels and fasting lipid profile and blood sugar levels, there was a weak, non-significant correlation between phenylalanine levels and cholesterol, triglycerides, LDL, HDL, and VLDL levels (rs = 0.142, 0.265, 0.13, -0.1, 0.123) (P = 0.341, 0.072, 0.384, 0.982, 0.409) respectively. However, a significant positive correlation was found between phenylalanine and fasting blood sugar levels (rs = 0.6, P = 0.01) (Table 5).
a Spearman correlation.
b Statistically significant at P ≤ 0.05.
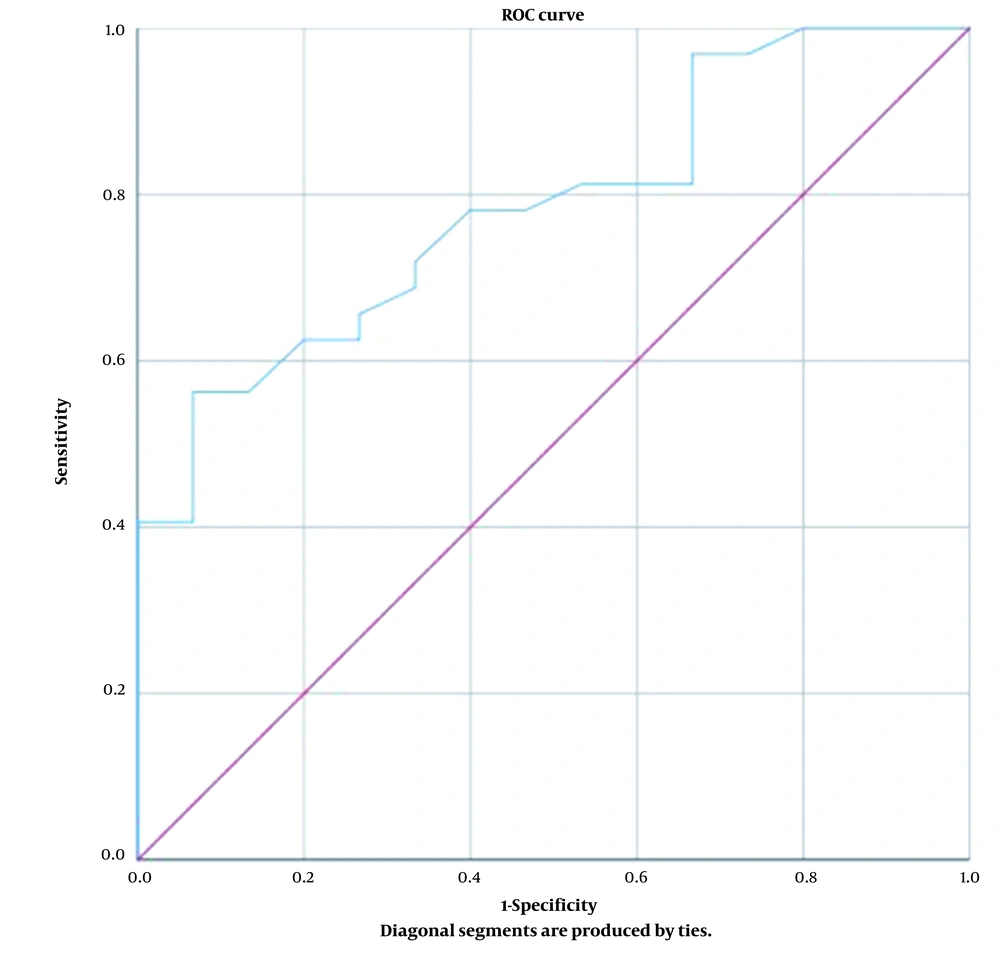
Higher fasting blood sugar levels were significantly associated with elevated PHE levels (Table 6). A fasting blood sugar cut-off point of 83.5 mg/dL accurately detected high phenylalanine levels. Additional information can be found in Table 7 and Figure 1.
| Fasting Blood Sugar | High Level of Phenylalanine | P Value b | |
|---|---|---|---|
| No | Yes | ||
| Hypoglycemia | 3 (20.0) | 0 (0.0) | 0.019 |
| Normal | 12 (80.0) | 29 (90.6) | |
| Hyperglycemia | 0 (0.0) | 3 (9.4) | |
a Values are expressed as No. (%).
b Montecarlo test; statistically significant at P ≤ 0.05.
| AUC | P a | Cut Off Point | Sensitivity | Specificity | 95% CI | |
|---|---|---|---|---|---|---|
| Value | 0.784 | 0.002 | 83.5 | 71.9% | 66.7% | 0.654 - 0.915 |
Abbreviation: AUC, area under the curve; CI, confidence interval.
a Statistically significant at P ≤ 0.05.
A logistic regression analysis was performed to predict high phenylalanine levels, considering age, gender, presence of malnutrition, and fasting blood sugar as covariates. A significant association was found between higher fasting blood sugar (mg/dL) and the prediction of high phenylalanine levels (P = 0.013). Detailed results can be found in Table 8.
Abbreviation: CI, confidence interval.
a Statistically significant at P ≤ 0.05.
5. Discussion
5.1. Main Findings of Our Study
The majority of the study participants (n = 34, 72.3% of enrolled children) were malnourished, with a higher incidence of overweight and obesity (n = 26, 55.3% of participants). Malnourished children had significantly higher PHE levels compared to normal children, while no significant difference was observed between overweight/obese children and the rest of the participants. Among the laboratory findings, fasting blood sugar significantly correlated with PHE levels.
5.2. Interpretation and Comparison of the Findings
Our study included children with PKU who were referred to the nutritional clinic of the National Nutrition Institute, and it was observed that there was a clear male predominance. This finding aligns with a previous study on newly diagnosed children with PKU, where 62.5% of the enrolled children were males (25). The presence of male predominance is not an uncommon finding and can be attributed to the higher prevalence of diseases in males and their shorter life expectancy compared to females, which is a universal demographic fact (26). Furthermore, no significant difference was found between males and females in terms of the prevalence of overweight and obese children in our study, which is consistent with previous findings (27).
Most of the children (72.3%) in our study were malnourished, which is consistent with previous research indicating lower height and weight Z-scores for children with PKU compared to normal children (28). These findings underscore the importance of nutritional surveillance in children with PKU to ensure adequate nutrient intake and support their growth based on relevant standards. Maintaining a long-term low phenylalanine diet can increase the risk of various types of malnutrition in these children. Compliance with a strict low PHE diet becomes more challenging as children grow older, primarily due to the wider availability of food choices. Therefore, adhering to a lifelong low PHE diet poses obstacles for both affected children and the medical team responsible for their care (29). A significant proportion of the patients enrolled in the study were classified as overweight and obese children. This finding aligns with a previous systematic review highlighting the common occurrence of overweight and obesity in children, possibly attributed to inappropriate food consumption and low physical activity levels (30). Several reports suggest a higher prevalence of overweight among PKU patients compared to the general population, although the exact reasons behind this association remain unclear (13, 14). It is important to note that a decrease in protein-based energy supplementation may result in increased consumption of dietary sources rich in carbohydrates, especially simple carbohydrates. As a result, the macronutrient proportions in the children’s diet may differ from those of normal children, potentially leading to higher calorie intake and an increased risk of being overweight or obese (31). A recent study discovered that energy intake and protein-derived energy did not differ significantly between PKU patients and healthy individuals. However, the proportion of energy obtained from dietary carbohydrates was higher in the PKU group (32). Additionally, this recent study identified reduced energy expenditure during moderate physical activity as another contributing factor to obesity in children with PKU.
It is essential to highlight that our study also included a significant number of children with other forms of malnutrition, such as underweight, wasted, and stunted children. This finding aligns with a previous observation made during the investigation of growth status in children with PKU. Researchers have identified limited access to PKU-appropriate food as the primary factor contributing to growth retardation in these children (33).
Our study observed higher phenylalanine levels in overweight and obese children, although the difference was not statistically significant. This finding is consistent with previous research suggesting that elevated PHE levels may serve as a predictive factor for overweight/obesity, potentially indicating poorer adherence to treatment (27). Additionally, we found weak non-significant correlations between PHE levels and low HDL, as well as higher levels of LDL and VLDL, which aligns with findings from the study above. Moreover, a significant positive correlation was observed between PHE levels and fasting blood sugar levels. This finding aligns with a previous study indicating that patients with PKU are at risk of insulin resistance and carbohydrate intolerance, likely associated with their higher caloric intake from carbohydrates (34).
A previous study identified an association between plasma PHE levels and inhibition of cholesterogenesis, while the low cholesterol intake from special diets for PKU children may also contribute to lower serum cholesterol levels. This study found an inverse correlation between cholesterol and phenylalanine levels that might be due to both the inhibition of cholesterogenesis and the low cholesterol content in the special protein -restricted diet in children with PKU (35). In children, a connection has been observed between hyperphenylalaninemia (HPA) and hypocholesterolemia (19). In this context, our study found a weak non-significant positive correlation between cholesterol levels and phenylalanine levels. It is important to mention that adherence to a special low-protein diet for PKU, which is typically low in animal proteins, can lead to lower cholesterol levels in children with PKU. Therefore, higher cholesterol levels in these patients might indicate non-compliance with the special diet. However, it is important to note that cholesterol levels cannot solely reflect compliance with a low-protein diet. In other words, high consumption of simple sugars has also been associated with increased cholesterol levels (36). An important point that can be emphasized from the lack of a significant correlation between cholesterol and phenylalanine levels is the need to identify possible hidden sources of phenylalanine. It is crucial to recognize that animal-based protein sources are not the sole contributors to PHE. Therefore, we should also consider other sources of proteins providing PHE, such as plants, including vegetables and fruits, with higher simple carbohydrate content and possible resultant higher cholesterol level (37). Additionally, plant sources are known to provide proteins (38). As a result, it is not advisable to randomly plan a child’s diet solely based on a plant-based approach without considering the protein content, glycemic indices, and carbohydrate loads of the foods.
5.3. Recommendations
Malnutrition assessment is essential for children with PKU. Early lipid screening, including obesity and lipid profile evaluation, is crucial to prevent adverse health outcomes in these vulnerable children, considering the long-term impact of childhood obesity into adulthood. Monitoring their dietary plans is important to avoid complications such as impaired glucose tolerance. Increased carbohydrate intake may contribute to overweight/obesity in these vulnerable children. Patients with PKU should receive long-term follow-ups, even if the PHE-restricted diet does not appear to be a risk factor for obesity development, given the increasing incidence of obesity in the general population. Personalized nutritional counseling and comprehensive monitoring of nutritional status should be part of an interdisciplinary approach. Additionally, maintaining regular physical activity is vital for these children. Further research with robust methodology is needed to fully address this topic and guide the clinical practice of healthcare professionals.
5.4. Limitations
The small sample size in our study is a potential limitation, which can be attributed to the relatively low prevalence of PKU. A larger sample size would likely provide a more representative data.
5.5. Conclusions
Malnutrition, particularly overweight and obesity, appears to be a significant concern in children with PKU. Fasting blood sugar levels may serve as nutritional biomarkers for children with PKU, given the observed linear correlation between fasting blood sugar and PHE levels.