1. Background
Migraine is the most prevalent type of primary headache among children and can cause significant debilitation during childhood and adolescence. Its incidence in childhood varies with age, ranging from 3% in younger children to 20% in adolescents (1). In children who are affected, migraines diminish the quality of life in aspects such as physical well-being, social and economic activities, and school performance (2). Typically, migraine episodes are moderate to severe in intensity, lasting from 2 to 72 hours if untreated. The pain is often localized, throbbing, and exacerbated by physical activity. Migraines may be accompanied by symptoms like nausea, vomiting, and sensitivity to light and sound. The duration of migraine attacks in children tends to be shorter compared to adults, and they frequently occur on both sides of the head. There are various types of migraines, with the most common being migraine with aura (classic migraine) and migraine without aura (common migraine) (3)
Constipation is a common condition in children, accounting for 3 - 5% of pediatrician referrals and 25% of referrals to pediatric gastroenterologists (4). A meta-analysis indicates that constipation prevalence ranges from 0.5% to 32.2%, with an overall prevalence of 9.5% (5). Previous studies have linked migraines to various health issues, including mental disorders, sleep disturbances, cardiovascular diseases, and digestive problems like peptic diseases and inflammatory bowel diseases (6-17). Functional disorders of the digestive system, in particular, have been strongly associated with migraine headaches. Research suggests that harmful stimuli from the digestive system are transmitted to the brain stem's nucleus of the solitary tract in close proximity to the caudal trigeminal nucleus. The caudal trigeminal nucleus also communicates with the nucleus of the solitary tract. With repeated digestive system stimulation, these signals may inadvertently reach the caudal trigeminal nucleus, potentially leading to neuroinflammation. Moreover, immune products, especially histamines released under the digestive tract's mucosa, can enter systemic circulation, making migraine patients more susceptible to attacks (18). Additional studies highlight the significance of serotonin pathways and psychological factors in the development of both headaches and constipation. During migraine attacks, a reduction in serotonergic activity has been observed, which can cause cranial vessel vasodilation and sensitization of the trigeminal nerve's meningeal afferents. Serotonin is also crucial for regulating various physiological functions in the digestive system, such as motility, secretion, and visceral sensitivity. Furthermore, previous findings have shown increased blood levels of 5-hydroxytryptamine in cases of diarrhea and decreased levels of constipation (19-24).
Despite various treatments available for migraines in children, there remains a need for additional clinical research on both preventive treatments and acute migraine attack management. Few clinical trials have been conducted in pediatric populations, often yielding inconsistent results (25). Investigating diseases associated with migraines is crucial as it may aid in discovering more effective treatments and understanding potential pathophysiological mechanisms.
2. Objectives
Given the high incidence of both migraine and functional constipation in children, this study was undertaken to explore the impact of constipation treatment on headache improvement in children who suffer concurrently from migraine and functional constipation.
3. Methods
This clinical trial focused on children aged 4 to 15 years with migraines who were referred to the pediatric neurology clinic at Besat Hospital in Hamadan, Iran, during 2019 - 2020. The study included those eligible for migraine prophylaxis.
Initially, a constipation diagnosis questionnaire based on the Rome IV diagnostic criteria was completed for the patients, and 32 patients with concurrent functional constipation were enrolled in the study. These children were then randomly divided into intervention and control groups.
The intervention group, consisting of 16 patients, received treatment for functional constipation in addition to propranolol (manufactured by Razak Company) at a dosage of 1 mg/kg, with a maximum daily dose of 80 mg. The control group, also comprising 16 patients, was treated solely with propranolol (manufactured by Razak Company).
The functional constipation treatment included dietary changes and the administration of Pidrolax powder without electrolytes (produced by Sepidaj Company) at a dosage of 0.7 g/kg twice daily, with the maximum dosage ranging from 13.8 to 40 g/day. For children with rectal impaction, a 5 mg Bisacodyl suppository was used daily for 3 - 5 days. Throughout the study, patients were not permitted to use any additional treatments for constipation.
The diagnosis of rectal impaction was determined through abdominal examination, digital rectal examination, and, when necessary, plain abdominal radiography. A successful response to constipation treatment was defined as achieving 1 to 3 painless bowel movements per week in the last month of treatment and a reduction in the frequency of fecal incontinence to fewer than 2 episodes per month.
Before initiating treatment, a questionnaire was completed to gather patient information and details about headache characteristics, such as the frequency of headaches in the past month and the duration of headache attacks. At the end of the first month, the questionnaire on headache characteristics was revisited, along with an additional questionnaire to evaluate the response to constipation treatment. Additionally, a 5-point Likert scale checklist was used to assess parental satisfaction with the treatment. The inclusion criteria for the study were as follows: (1) Age between 4 to 15 years; (2) diagnosis of migraine headache by a pediatric neurologist based on the ICHD III β criteria; (3) diagnosis of functional constipation by a pediatric gastroenterologist according to the Rome IV criteria; (4) eligibility for migraine prophylaxis with propranolol, indicated by frequent headaches occurring more than once a week or more than 4 times a month, unsatisfactory response to symptomatic treatments, or presence of debilitating headaches leading to school absenteeism, dysfunction at home, or disruption of the child's social activities.
The exclusion criteria were: (1) The presence of contraindications to receiving propranolol, such as a history of asthma, liver disease, diabetes mellitus, bradycardia, heart block, or severe blood pressure drop; (2) lack of satisfaction or cooperation in participating in the study.
In this study, the chi-square test (or Fisher's exact test) was utilized to compare qualitative variables, while the Mann-Whitney test was applied to compare quantitative variables. Data analysis was conducted using SPSS statistical software version 20, with a significance level set at less than 0.05. The Ethics Committee of Hamadan University of Medical Sciences approved this study, assigning the ethics code from the Deputy of Research and Technology, IR.UMSHA.REC.1398.951, and the IRCT number IRCT20120215009014N361.
Prior to commencing the study, written informed consent was obtained from all participants or their parents.
4. Results
In this clinical trial, 32 children with migraine headaches and functional constipation who met the inclusion criteria were enrolled into 2 equal groups.
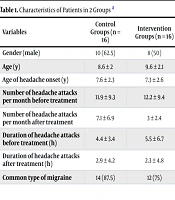
There were no significant differences between the 2 groups in terms of gender distribution, average age, and other baseline characteristics (as shown in Tables 1). and 2).
| Variables | Control Groups (n = 16) | Intervention Groups (n = 16) | P-Value |
|---|---|---|---|
| Gender (male) | 10 (62.5) | 8 (50) | 0.722 |
| Age (y) | 8.6 ± 2 | 9.6 ± 2.1 | 0.160 |
| Age of headache onset (y) | 7.6 ± 2.3 | 7.3 ± 2.6 | 0.530 |
| Number of headache attacks per month before treatment | 11.9 ± 9.3 | 12.2 ± 9.4 | 0.731 |
| Number of headache attacks per month after treatment | 7.1 ± 6.9 | 3 ± 2.4 | 0.016 |
| Duration of headache attacks before treatment (h) | 4.4 ± 3.4 | 5.5 ± 6.7 | 0.277 |
| Duration of headache attacks after treatment (h) | 2.9 ± 4.2 | 2.3 ± 4.8 | 0.615 |
| Common type of migraine | 14 (87.5) | 12 (75) | 0.654 |
Characteristics of Patients in 2 Groups a
| Variables | Intervention Group | Control Group | P-Value |
|---|---|---|---|
| Stress | 5 (31.25) | 3 (18.75) | 0.560 |
| Playing and studying with a mobile phone | 4 (25) | 8 (50) | |
| Insomnia | 1 (6.25) | 2 (12.5) | |
| Tiredness | 2 (12.5) | 2 (12.5) | |
| Overworking | 1 (6.25) | - | |
| Nutrition | 3 (18.75) | 1 (6.25) | |
| Total | 16 (100) | 16 (100) |
Trigger Factors of Migraine Headaches in 2 Groups a
Before the intervention, children in both groups experienced an average of 12 headache attacks per month, with each attack lasting between 4 to 5.5 hours; these figures were not significantly different. Following treatment, the intervention group exhibited a significant reduction in the number of headache attacks compared to the control group (P = 0.016). Although the average duration of each headache attack in the intervention group decreased more than in the control group, this difference was not statistically significant (Table 1).
Regarding parental satisfaction with the treatment, while it was higher in the intervention group compared to the control group, this difference was not significant (Table 3). The treatment of constipation was successful in all children.
5. Discussion
Functional constipation and migraine headaches are both prevalent in children. Frequent headache attacks and their associated complications diminish personal interactions, the quality of life, and the daily activities of children suffering from migraines.
The findings of this study indicate that treating constipation and migraines concurrently in children reduces the frequency of headache attacks. However, this combined treatment does not significantly impact the duration of each headache attack or parents' satisfaction with the treatment. These results may be attributed to the study's small sample size and the short duration of the intervention.
Currently, issues in migraine pharmaceutical treatments, such as drug intolerance, inadequate or insufficient response to treatment, and high treatment costs, mean that many patients do not receive satisfactory prophylactic care.
Previous research has demonstrated a link between headaches and upper digestive tract complaints in children (26, 27), but there has been limited investigation into the relationship between headaches and lower digestive tract complaints, such as constipation.
In a cross-sectional study conducted in 2014 by Inaloo et al. in Shiraz, which aimed to explore the relationship between headache and functional constipation, 326 children aged 4 to 12 years were examined. The study found that children with constipation were 4.2 times more likely to experience headaches (12). In a 2018 cross-sectional study by Martami et al., aimed at investigating the relationship between migraine and tension-type headaches and gastrointestinal tract diseases in the lower and upper regions, 1574 overweight and obese patients from the Obesity Research Center of Sina Hospital in Tehran were studied. This study revealed a significant association between migraine and constipation, even after adjusting for other confounding factors. Specifically, patients with migraines were found to have a 3.96 times higher likelihood of experiencing constipation (16). Furthermore, Aamodt et al.'s research indicated that individuals with headaches are nearly twice as likely to suffer from constipation (6). The 2019 study by Rezaeiashtiani et al., involving 50 adult patients with both migraine and constipation, found that the intensity of migraine headaches in the intervention group was significantly lower compared to the control group post-treatment. However, no significant difference was observed between the two groups in terms of parental satisfaction with the treatment and the duration of headache attacks after treatment, aligning with the findings of our study. Additionally, in Rezaeiashtiani et al.'s study, the severity of constipation post-treatment was significantly reduced in the intervention group compared to the control group (13). In our study, all children in the intervention group experienced successful constipation treatment, whereas no change in constipation severity was observed in the control group.
In a 2015 study by Park et al. in Korea, out of 96 patients with primary headaches, 24 were found to have constipation. Post-treatment, improvements in both headache and constipation were observed. The authors suggest that constipation plays a significant role in causing headaches and that headaches and constipation may share a common pathophysiology (14). Previous research has indicated that serotonin (5-hydroxytryptamine) is involved in both migraine and constipation, increasing in the digestive tract mucosa of constipation patients while decreasing in those with migraines (19). Additionally, constipation is linked to stress, dehydration, reduced fluid intake, and lack of appetite, factors that may also be connected to headaches. The Valsalva maneuver, often used by individuals with constipation, could also contribute to migraines (28). Furthermore, some studies have found that antioxidant levels are lower in migraine patients, and constipation may impede the absorption of antioxidants (29).
5.1. Conclusions
The study's findings indicate that treating constipation in children with both migraine and functional constipation leads to a reduction in the number of headache attacks. However, to generalize these results, a study with a larger sample size is needed. It is advisable to screen children with migraines for constipation and provide treatment if constipation is present.