1. Background
Type 1 diabetes (T1D) is a prevalent endocrine disorder among children, associated with various health complications. Its increasing incidence worldwide over recent decades has highlighted it as a significant health issue (1, 2). Although genetic factors are pivotal in T1D development, accumulating evidence suggests environmental factors also contribute to the disease's rising prevalence (3, 4). Endocrine-disrupting chemicals (EDCs), identified as potential environmental triggers, are of particular concern due to their role in diabetes pathogenesis (5, 6). Endocrine-disrupting chemicals can disrupt the human endocrine system by mimicking, blocking, or altering hormonal functions (6, 7).
Organochlorine pesticides (OCPs), a notable group of EDCs, have been extensively utilized in agriculture and public health. Organochlorine pesticides persist in the environment and accumulate in human tissues through food consumption and inhalation, leading to various adverse health effects, including impacts on the reproductive and endocrine systems, the nervous system, and associations with certain cancers (8, 9).
While the link between type 2 diabetes (T2D) and OCPs has been documented (10, 11), the connection between T1D and OCPs remains underexplored, suggesting they might be potential risk factors for T1D development. The mechanisms by which OCP exposure could lead to T1D are not entirely understood, but possible pathways include beta-cell damage due to oxidative stress and inflammation or alterations in the gut microbiome, resulting in immune dysregulation and T1D onset (12-14).
Animal studies on pesticide effects have shown inconsistent results, indicating both immune system suppression and stimulation (12, 15, 16). Human studies investigating the association between OCPs and T1D are limited and have yielded conflicting outcomes (17-20). Further research is recommended to clarify the relationship and underlying mechanisms between T1D and pesticide exposure (12).
2. Objectives
In light of the global increase in diabetes incidence, especially in developing countries, and the variation in pesticide exposure across regions, this study aims to investigate the association between organochlorine pesticide levels and T1D among children and adolescents in our area. Our research will shed light on the potential interaction between environmental factors, such as pesticide exposure, and the growing prevalence of this chronic disease, contributing to the development of focused preventive measures and policies.
3. Methods
This case-control study was conducted among 147 new cases of T1D and 147 healthy controls in Isfahan City from 2021 - 2022. Newly diagnosed T1D patients were recruited from various private pediatric endocrinologists' clinics and the diabetes clinic of Imam Hossein Children's Hospital, affiliated with Isfahan University of Medical Sciences. Control subjects, matched by residential area and without any history of T1D, were selected. Children with any endocrine disorder or metabolic disease were excluded from the study. Approval was granted by the Ethics Committee of Isfahan University of Medical Sciences under research project number 196082 and the Ethics Code IR.MUI.REC.1396.1.082. Written informed consent was obtained from the subjects or their parents.
3.1. Collection of Information
Baseline characteristics, including sociodemographic details (parental occupation, parental education), diet, breastfeeding duration, and family history of diabetes, were gathered using a validated questionnaire.
3.2. Anthropometric Measurements
A trained nurse measured body weight and height using validated instruments. Body mass index (BMI) was calculated by dividing weight (kg) by height squared (m2). The World Health Organization's specific BMI curves were used to classify participants' weight status, controlling for age and gender differences (Underweight as BMI < 5th percentile, normal weight as 5 - 85th percentile, overweight as 85 - 95th percentile, and obesity as > 95th percentile) (21).
3.3. Dietary Intake
The questionnaire also assessed participants' eating habits, examining 15 food groups, including fried hamburgers, fried meats, grilled meats, fried chicken, grilled chicken, seafood, grilled fish, fried fish, fast food, sausages, puffs, chips, mayonnaise, canned foods, and soft drinks. Subjects reported their weekly consumption frequency of these foods. Responses on the Likert scale were categorized as follows: 1 = "never," 2 = "once every two weeks," 3 = "once a week," and 4 = "several times a week."
Principal component analysis (PCA) on the food groups led to the identification of five dietary patterns among participants: An unhealthy snack diet (salty snacks), a Western pattern, a high-protein diet, a seafood diet, and canned foods. These patterns were then divided into tertiles (the first tertile representing low consumption level, the second tertile as medium consumption, and the third tertile as high consumption level).
3.4. Laboratory Measurements
Spot urine samples were collected from both the study and control groups to assess the levels of OCPs. The urine samples from all participants were stored at − 20°C until the extraction of OCPs. We analyzed 14 OCPs, including αBHC, β-BHC, heptachlor, aldrin, heptachlor epoxide, α-endosulfan, p,p’-DDE, dieldrin, endrin, β-endosulfan, p,p’-DDD, endrin-aldehyde, endosulfan sulfate, and 4,4’ DDT, in each sample.
The majority of the OCPs, such as α-BHC (94.9% below the limit of detection [LOD]), p,p'-DDE (98.3% below LOD), dieldrin (100% below LOD), endrin (100% below LOD), β-endosulfan (98.3% below LOD), endrin-aldehyde (99.7% below LOD), endosulfan sulfate (99.3% below LOD), and 4,4'-DDT (99.7% below LOD), were predominantly non-detectable and thus excluded from further analysis.
For heptachlor epoxide (67.7% below LOD) and p,p’-DDD (77.6% below LOD), a significant proportion of observations were below the LOD. Consequently, concentrations below the LOD were replaced with random values from a uniform distribution between zero and the respective LOD (22).
Other OCPs, such as β-HCH (94.6% detectable), heptachlor (89.5% detectable), aldrin (92.5% detectable), and α-endosulfan (74.8% detectable), showed a majority of participants with concentrations above the LOD. The urine concentrations of OCP metabolites below the LOD were substituted with LOD/2 for the statistical evaluation.
Of the OCPs measured, only six OCPs were selected for analysis. The urine concentrations of these six OCPs were adjusted for urinary creatinine and divided into tertiles: The first tertile representing low concentrations, the second tertile moderate concentrations, and the third tertile high concentrations of each metabolite.
3.5. Statistical Analysis
The analysis was conducted using STATA 10 software (StataCorp, College Station, Texas, USA). The urinary concentrations of OCPs are reported as median (25th - 75th percentile) and geometric mean. The initial analysis of variables related to T1D utilized independent t-tests and chi-square tests. The Mann–Whitney test compared urinary OCP levels between diabetic cases and non-diabetic controls. Multiple logistic regression assessed the associations between urinary OCP levels and T1D, adjusting for age, sex, breastfeeding duration, BMI, family history of diabetes, and five dietary patterns.
4. Results
The general characteristics of the participants in both the case and control groups are summarized in Table 1. The average age was 8.5 (± 3.7) years, with no significant difference between the diabetic cases and non-diabetic controls. Approximately 48.3% of participants were female. Significant differences were observed between the groups regarding BMI (P = 0.033) and breastfeeding duration (P = 0.016).
| Characteristics and Category | All Subjects (n = 294) | Controls (n = 147) | Cases (n = 147) | P-Value b |
|---|---|---|---|---|
| Age, y | 8.5 (3.7) | 8.6 (3.7) | 8.4 (3.7) | 0.668 |
| BMI, kg/m2 | 17 (4.3) | 17.6 (4.6) | 16.4 (4.1) | 0.033 |
| BMI categories | 0.363 | |||
| Underweight | 51 (20.7) | 20 (16.4) | 31 (25) | |
| Normal weight | 132 (53.7) | 67 (54.9) | 65 (52.4) | |
| Overweight | 24 (9.8) | 13 (10.7) | 11 (8.9) | |
| Obese | 39 (15.9) | 22 (18) | 17 (13.7) | |
| Gender | 0.641 | |||
| Girls | 142 (48.3) | 69 (46.9) | 73 (49.7) | |
| Boys | 152 (51.7) | 78 (53.1) | 74 (50.3) | |
| Father’s education | 0.575 | |||
| Illiterate/elementary school | 49 (16.7) | 22 (15) | 27 (18.5) | |
| Secondary school/high school | 195 (66.6) | 102 (69.4) | 93 (63.7) | |
| University | 49 (16.7) | 23 (15.6) | 26 (17.8) | |
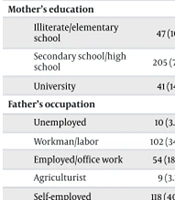
| Mother’s education | 0.977 | |||
| Illiterate/elementary school | 47 (16) | 24 (16.3) | 23 (15.8) | |
| Secondary school/high school | 205 (70) | 103 (70.1) | 102 (69.9) | |
| University | 41 (14) | 20 (13.6) | 21 (14.4) | |
| Father’s occupation | 0.817 | |||
| Unemployed | 10 (3.4) | 4 (2.7) | 6 (4.1) | |
| Workman/labor | 102 (34.8) | 49 (33.3) | 53 (36.3) | |
| Employed/office work | 54 (18.4) | 28 (19) | 26 (17.8) | |
| Agriculturist | 9 (3.1) | 6 (4.1) | 3 (2.1) | |
| Self-employed | 118 (40.3) | 60 (40.8) | 58 (39.7) | |
| Mother’s occupation | 0.689 | |||
| Housewife | 262 (89.4) | 128 (87.7) | 134 (91.2) | |
| Workman/labor | 4 (1.4) | 3 (2.1) | 1 (0.7) | |
| Employed/office work | 14 (4.8) | 8 (5.5) | 6 (4.1) | |
| Others | 13 (4.4) | 7 (4.8) | 6 (4.1) | |
| Breastfeeding duration, month | 0.016 | |||
| < 6 | 23 (8) | 5 (3.5) | 18 (12.5) | |
| 6 to 12 | 13 (4.5) | 8 (5.6) | 5 (3.5) | |
| > 12 | 250 (87.4) | 129 (90.8) | 121 (84) | |
| Family history of diabetes | 0.332 | |||
| Yes | 34 (13.9) | 19 (16.1) | 15 (11.8) | |
| No | 211 (86.1) | 99 (83.9) | 112 (88.2) |
a Values are expressed as mean (SD).
b P-values are based on the t-test, chi-square test, or Fisher’s exact test.
The frequency of food group consumption among children and adolescents with and without T1D is detailed in Table 2.
| Food Group | Cases (n = 147) | Controls (n = 147) | P-Value b |
|---|---|---|---|
| Salty snacks | 0.319 | ||
| Tertile 1 | 44 (35.8) | 32 (30.2) | |
| Tertile 2 | 36 (29.3) | 41 (38.7) | |
| Tertile 3 | 43 (35) | 33 (31.1) | |
| Western dietary patterns | 0.480 | ||
| Tertile 1 | 37 (30.1) | 39 (36.8) | |
| Tertile 2 | 45 (36.6) | 32 (30.2) | |
| Tertile 3 | 41 (33.3) | 35 (33) | |
| Meat foods | 0.165 | ||
| Tertile 1 | 46 (37.4) | 30 (28.3) | |
| Tertile 2 | 35 (28.5) | 42 (39.6) | |
| Tertile 3 | 42 (34.1) | 34 (32.1) | |
| Seafood | 0.515 | ||
| Tertile 1 | 37 (30.1) | 39 (36.8) | |
| Tertile 2 | 42 (34.1) | 35 (33) | |
| Tertile 3 | 44 (35.8) | 32 (30.2) | |
| Canned foods | 0.728 | ||
| Tertile 1 | 38 (30.9) | 38 (35.8) | |
| Tertile 2 | 42 (34.1) | 34 (32.1) | |
| Tertile 3 | 43 (35) | 34 (32.1) |
a Values are expressed as No. (%).
b P-value based on chi-square test
Table 3 displays the urinary concentrations of the six OCPs for both case and control groups. The urinary concentration of p,p'-DDD was significantly higher in cases than in controls (P < 0.001).
| Pesticides (μg/g Creatinine) | GM | Total (n = 294) | Cases (n = 147) | Controls (n = 147) | P-Value b |
|---|---|---|---|---|---|
| β-BHC | 9.1 | 11.8 (4.0 - 27.7) | 11.3 (4.1 - 27.7) | 14.2 (3.9 - 28.8) | 0.611 |
| Heptachlor | 1.94 | 2.4 (0.82 - 5.4) | 2.3 (0.8 - 5.4) | 2.4 (0.8 - 5.8) | 0.982 |
| Aldrin | 24.6 | 36.2 (11.4 - 102) | 32.5 (12.6 - 112.4) | 38 (10.6 - 102) | 0.918 |
| Heptachloro epoxy | 0.18 | 0.17 (0.06 - 0.52) | 0.2 (0.05 - 0.6) | 0.2 (0.06 - 0.45) | 0.949 |
| α-endosulfan | 4.62 | 10.1 (0.32 - 46.1) | 10.9 (0.4 - 48) | 6.4 (0.3 - 43.6) | 0.975 |
| p,p'-DDD | 0.127 | 0.12 (0.05 - 0.29) | 0.09 (0.04 - 0.2) | 0.15 (0.07 - 0.3) | < 0.001 |
Abbreviations: GM: geometric mean; p,p’-dichlorodiphenyldichloroethylene (p,p’-DDE).
a Values are represented as median (25th - 75th percentile).
b P-values referred to the Mann-Whitney U test.
Table 4 presents logistic regression models estimating the association between urinary OCP levels and T1D. After adjusting for age, sex, breastfeeding duration, BMI, family history of diabetes, and five dietary patterns, a significantly positive association was found between the highest levels of p,p'-DDD, and T1D (OR = 4.9; 95% CI: 2 - 12.3).
| Metabolites | Crude Models | Adjusted Model a | Adjusted Model b | |||
|---|---|---|---|---|---|---|
| OR (95% CI) | P-Value | OR (95% CI) | P-Value | OR (95% CI) | P-Value | |
| β-BHC | ||||||
| Tertile 1 | 1 | 1 | 1 | |||
| Tertile 2 | 1.2 (0.7-2.1) | 0.519 | 2.1 (1.01-4.4) | 0.045 | 2.9 (1.2 - 6.8) | 0.015 |
| Tertile 3 | 1.1 (0.6-1.9) | 0.719 | 1.4 (0.7-2.7) | 0.403 | 1.7 (0.7-4.0) | 0.214 |
| Heptachlor | ||||||
| Tertile 1 | 1 | 1 | 1 | |||
| Tertile 2 | 1.0 (0.6 - 1.8) | 0.944 | 1.2 (0.6 - 2.4) | 0.695 | 1.3 (0.6 - 2.9) | 0.519 |
| Tertile 3 | 0.9 (0.5 - 1.6) | 0.721 | 0.7 (0.4 - 1.5) | 0.427 | 0.6 (0.2 - 1.3) | 0.192 |
| Aldrin | ||||||
| Tertile 1 | 1 | 1 | 1 | |||
| Tertile 2 | 0.9 (0.6 - 1.7) | 0.944 | 1.2 (0.6 - 2.4) | 0.639 | 1.7 (0.7 - 4.0) | 0.209 |
| Tertile 3 | 1.1 (0.6 - 1.9) | 0.830 | 1.2 (0.6 - 2.4) | 0.658 | 1.8 (0.8 - 4.2) | 0.165 |
| α-endosulfan | ||||||
| Tertile 1 | 1 | 1 | 1 | |||
| Tertile 2 | 0.7 (0.4 - 1.3) | 0.284 | 0.8 (0.4 - 1.6) | 0.553 | 0.6 (0.3 - 1.4) | 0.238 |
| Tertile 3 | 0.98 (0.6 - 1.7) | 0.940 | 1.3 (0.6 - 2.6) | 0.539 | 1.3 (0.6 - 3.0) | 0.502 |
| Heptachloro epoxy | ||||||
| Tertile 1 | 1 | 1 | 1 | |||
| Tertile 2 | 1.3 (0.7 - 2.3) | 0.353 | 1.9 (0.9 - 3.9) | 0.086 | 1.5 (0.6 - 3.4) | 0.353 |
| Tertile 3 | 0.9 (0.5 - 1.6) | 0.723 | 1.1 (0.6-2.3) | 0.734 | 1.0 (0.5 - 2.3) | 0.934 |
| p,p'-DDD | ||||||
| Tertile 1 | 1 | 1 | 1 | |||
| Tertile 2 | 2.5 (1.4 - 4.4) | 0.002 | 3.5 (1.7 - 7.5) | 0.001 | 5.6 (2.2 - 14.4) | < 0.001 |
| Tertile 3 | 2.8 (1.6 - 5.0) | < 0.001 | 4.1 (1.9 - 8.6) | < 0.001 | 4.9 (2 - 12.3) | 0.001 |
a Adjusted for age, sex, breastfeeding duration, body mass index, and family history of diabetes.
b Adjusted for age, sex, breastfeeding duration, body mass index, family history of diabetes, and 5 dietary patterns.
Furthermore, participants with the middle tertile of urinary β-BHC had a higher odds ratio (OR) of T1D than those in the lowest tertile (OR = 2.9; 95% CI: 1.2 - 6.8). No association was observed between other OCP concentrations and T1D.
5. Discussion
This study compared urinary OCP levels in T1D patients and healthy subjects and evaluated their association with the condition. We observed higher urinary concentrations of p,p'-DDD in diabetic children compared to controls, along with significant positive associations between the highest levels of p,p'-DDD, and T1D. Moreover, we noted a higher OR of T1D in participants with a medium level of urinary β-BHC compared to those with the lowest level.
Recent evidence underscores the significant role of environmental factors, particularly EDCs, in the pathogenesis of T1D, suggesting them as key contributors to the rising global incidence of T1D (4-6).
Despite mounting evidence of an association between OCP exposure and T1D, existing literature faces several challenges. Most studies are cross-sectional or case-control, with inconsistencies in the types of OCPs measured and measurement methods across studies. As an initial study in our region, we conducted the current research.
Our literature review uncovered studies exploring the impact of prenatal and early-life exposure to OCPs on T1D development (17, 18).
In a case-control study conducted in Sweden involving 150 children diagnosed with T1D and 150 age and birth-day-matched controls, researchers assessed the levels of p,p’-DDE in mothers' stored serum during pregnancy. The concentrations of p,p’-DDE were similar between the T1D cases and controls (9.2 vs. 9.6 ng/mL, respectively) and showed a decrease over time. The findings did not support the hypothesis that prenatal exposure to persistent organic pollutants (POPs) increases the risk of developing T1D (17).
In the FINDIA and DIABIMMUNE birth cohort studies, researchers investigated the association between early-life exposure to environmental pollutants and the risk of developing diabetes-predictive autoantibodies in children genetically susceptible to T1D. They measured levels of various POPs and per and polyfluorinated substances in cord blood and plasma samples from infants at 12 and 48 months of age. No significant link was found between exposure to these environmental chemicals and the development of clinical T1D, suggesting that fetal or early childhood exposure does not significantly increase the risk of β-cell autoimmunity and T1D (18).
A study in Egypt found that children newly diagnosed with T1D had higher serum levels of eight out of nine evaluated organochlorine and organophosphorus pesticides compared to healthy controls (19). Lindane was the most frequently detected organochlorine pesticide among T1D patients (70.7%), followed by o,p’-DDD (21.3%), p,p’-DDE (21.3%), and endrin (10.7%), with malathion being the most commonly found organophosphorus compound (65.3% of cases). This suggested an increased risk of T1D in children exposed to these compounds (19).
A recent study in the USA, using data and biological samples from the SEARCH-CC, explored the role of POPs in the onset of T1D among a well-characterized group of young individuals. Additionally, an experimental model was used to study the effects of POPs on β-cells. Elevated plasma levels of p,p’-DDE, trans-nonachlor, and PCB-153 were associated with an increased risk of T1D in patients with normal insulin sensitivity but not in those with insulin resistance (IR). Experimental findings showed that PCB-153 and p,p’-DDE significantly reduced insulin levels within cells and insulin secretion in pancreatic β-cells (20).
The interpretation of these findings suggests that young patients with T1D who also exhibit IR possess a greater amount of adipose tissue or exhibit higher BMI and waist circumference compared to those with normal insulin sensitivity. This increased adipose tissue capacity allows T1D patients with IR to store higher levels of POPs, thereby reducing their circulating levels (20).
In our study, among the OCPs analyzed, only the level of urinary p,p'-DDD was found to be elevated in diabetic patients, and a significant association between this metabolite and T1D was identified. Additionally, an increased level of β-BHC exposure may be associated with T1D.
Given the limited number of studies in this area and regional variations in pesticide use, as well as genetic differences among participants, our results could serve as foundational information for future research evaluating exposure sources and their effects.
Yang et al., in a study within the USA, explored the potential relationship between serum concentrations of OCPs and 25-hydroxyvitamin D (25(OH)D) levels in the general U.S. population, using data from the National Health and Nutrition Examination Survey (NHANES) 2003 - 2004. They found significant inverse associations between serum 25(OH)D levels and the OCPs detectable in over 80% of participants, specifically p,p’-DDT, p,p’-DDE, and β-hexachlorocyclohexane. Notably, p,p’-DDT showed consistent inverse associations across different subgroups, categorized by age, race, and chronic disease presence (23).
Considering the role of vitamin D deficiency in the pathogenesis of T1D (24, 25), it is suggested that OCP exposure may increase the risk of T1D among children with vitamin D deficiency. However, further research is needed in this area.
The limitations of our study include its case-control design, small sample size, the assessment of urinary OCP metabolites from a single urine sample, and the simple classification of pesticides. A strength of this study lies in the careful matching of the control group to the patient group in terms of age, sex, location, and novelty.
In conclusion, our study supports the potential role of p,p'-DDD, and β-BHC in the development of T1D. Considering the significant public health implications of this association, further research is essential to elucidate the mechanisms behind this relationship and to explore potential preventive strategies. For future studies, employing larger designs, increasing sample sizes, providing more precise pesticide classifications, and conducting a comprehensive analysis of various variables are recommended.