1. Introduction
Brugada syndrome is a hereditary channelopathy, which is autosomal dominant. The incidence of this syndrome in adults is five in 10,000 (1). In most patients, mutations in the sodium channel gene SCNSA are present (1, 2). This syndrome is characterized by ST-segment elevation in the right precordial leads, right bundle branch block (RBBB) pattern and ventricular tachyarrhythmia in patients with normal structural heart (1). This syndrome can cause sudden death, especially in males during their forties. Also, it is responsible for 20% of cases with sudden death in patients with normal heart and 4% of all sudden death cases in adults (1). However, this syndrome can either cause sudden death in the first year of life or cause no symptoms for life. Brugada syndrome has three types, based on the shape of the T wave, ST segment configuration and changes in the last section of the ST elevation (3). Sudden death occurs due to ventricular arrhythmia such as ventricular tachycardia or ventricular fibrillation. Fever is the most common provocative factor for arrhythmia in these patients (4). The clinical features of this syndrome include dysrhythmia, ventricular arrhythmia, syncope, seizures, respiratory problems, sudden death, cardiac arrest and sudden death at a young age. Unlike conditions such as long QT syndrome (LQTS) and CPVT, arrhythmia attacks occur during rest and sleep in this syndrome, which is similar to LQTS-3 (5). Patients can be treated with quinidine until they receive implantable cardioverter defibrillator (ICD). Quinidine can reduce VF/VT attacks in these patients. A noticeable proportion of the patients do not have any apparent symptom in their electrocardiogram; the so-called low risk patients; thus a drug challenge is required to unmask the electrocardiogram (ECG). Frequent and regular follow-up is recommended for low risk patients (6). The intermediate risk patients are those who do not have any symptom but their ECG is indicative of Brugada syndrome. In this case report, we presented a case with atypical symptoms of Brugada syndrome.
2. Case Presentation
A 2.5-year-old girl, known case of cerebral palsy (CP), was admitted to the intensive care unit because of aspiration pneumonia and acute respiratory distress. The patient was diagnosed as quadriplegic CP at the age of six months. The patient was admitted due to aspiration pneumonia seven times before this admission. She was the first child of the family, was born via normal delivery and had a birth weight of 8.2 kg. Her current weight was 9 kg. The body temperature at the time of admission was 38.2°C and 37.9°C in the second day of hospitalization. Average heart rate on admission was 128 beats per minute (bpm) and 120 bpm on the second day. In physical examination, S1 and S2 heart sounds were normal and no murmur was present in the auscultation. All extremities were flaccid and hypotonic. Neurological development was delayed and also speech recognition and patient movements were disrupted and delayed. In neurology examination, distal tendon reflex was absent and plantar reflex was upward. None of the laboratory findings were indicative of metabolic disorder. She had a history of surgery at the age of six months due to gastro esophageal reflux and was under treatment with pantoprazole. She had no family history of sudden death, arrhythmia or syncope. However, her parents were cousins. Upper GI series was negative. Chest X-ray showed pneumonia of the right upper lobe, and because of recurrent pneumonia, spiral computed tomography scan of the lungs was requested to rule out congenital structural anomalies. Urine analysis was normal and blood analysis suggested iron deficiency anemia. Laboratory analysis of the kidney and liver functions were normal, and the patient did not have any electrolyte disturbances except hypocalcaemia (Ca = 6.4). Serum albumin was within a normal range. The SCN5A gene mutation was excluded in the genetic analysis. Empiric antibiotic treatment was begun.
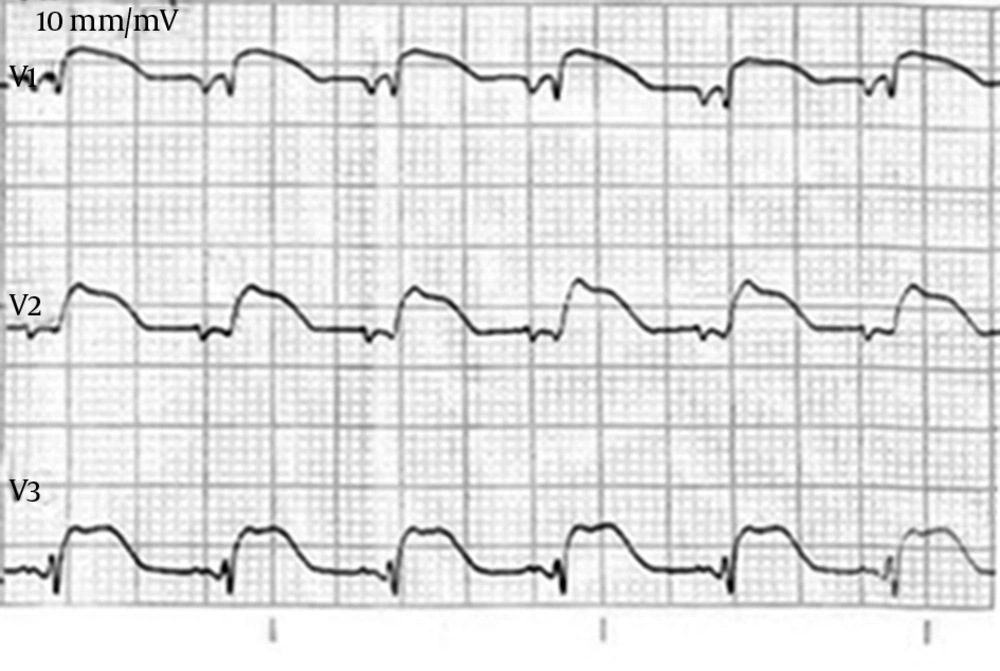
In the ECG, sinus heart rhythm was seen (PR interval = 160 ms, ORS duration = 80 ms and QTC = 420 ms), and in V1 - V3 leads caved ST-segment elevation > 2 mm, J point elevation with down sloping, and RBBB pattern was observed. The ECG findings were compatible with type 1 Brugada syndrome (Figure 1). Echocardiographic findings were normal. On the second day, the patient developed polymorphic ventricular tachycardia and the resuscitation was unsuccessful.
3. Discussion
The exact prevalence of Brugada syndrome in children is not known. As it is a rare heart condition, lack of knowledge about its symptoms and unawareness of the clinical manifestations and ECG changes is a barrier to early diagnosis of this syndrome. Prevalent Brugada syndrome is 8 - 10 times more in males; however the possibility of having a mutated gene does not differ by gender. This disease is the most common cause of sudden death in young men under 50 years of age without any other heart disease in parts of Asia, particularly south East Asia (7). The whole family history of our case for Brugada syndrome or sudden death was negative. According to a study in 2007 on 30 children with Brugada syndrome, fever was the most common cause of arrhythmia attacks (4), which is more compatible to our case. It should be noted that hyperkalemia is known as a provocative factor for arrhythmia in Brugada syndrome but our case had hypocalcaemia. However, we made the diagnosis through ECG changes in this case.
Brugada syndrome has three types; Type I Brugada pattern consists of a right bundle branch block with down-sloping ST segment elevations greater than 2 mm and inverted T waves in leads V1 through V3. Type 2 and 3 Brugada patterns also have a right bundle branch block-like pattern, yet the ST segment has a saddleback appearance and T wave inversion is not as marked. Some times ECG findings are equivocal patterns. This challenge in Type 2 or 3 Brugada is visible specially, even in the setting of symptoms, is not sufficient to make the diagnosis. Several maneuvers, such as moving the ECG leads higher in the chest or drug challenge are useful for changing the ECG and diagnosis. Intravenous sodium channel blockers such as flecainide or procainamide are often used for achieving classic pattern ECG, type 1.
Of course, ventricular tachycardia can occur during the drug challenge, thus this must be done in a controlled, monitored setting.
Based on the ECG findings in our case, our patient had Brugada syndrome type-1 and as she had no obvious clinical signs, but had clear changes in ECG, she fell under the intermediate group. Usually patients with Brugada syndrome have normal physical examinations. Most cases of Brugada syndrome have been associated with alterations in the SCN5A gene. Mutations in other genes have been shown to cause a variant of Brugada syndrome, including the genes coding for alpha1 and beta2b subunits of the L-type calcium channel (CACNA1C and CACNB2). Another mutation in BRS is HEY2 and Glycerol-3-phosphate dehydrogenase 1-like gene (GPD1L) has been shown. Mutations in genes GPD1-Land SCN1B have been identified in a few familial cases. The genetic study in our case revealed SCN5A mutations that are the most common mutations identified in Brugada syndrome. Generally, Brugada syndrome is hereditary and is transmitted as an autosomal dominant disease; however, almost 50% of cases are sporadic. Although no relationship has been found between gene mutations and fever (8), a relationship between gene mutation and severity and delayed conduction disorder have been identified (9).
If a child is a known case of Brugada syndrome, treatment of fever should be done promptly, as fever is the most important cause of VT episodes. This occurs because of rapid inactivation of sodium channels aggravated by fever. In other words, gene mutations make the sodium channels sensitive to fever (3, 10).
In our case, the PR interval was normal and heart block was absent, and also ECG showed ST elevation of > 2 mm in V1 - V2 leads and incomplete right bundle branch block. It has been found that in 85% of patients, PR interval and QRS duration are in the upper limits of the normal range (11). One study showed that prolongation of the conduction time is an emergency finding as well as a common manifestation in children (12). Thus, children, who have normal heart structure and present VT episodes following fever, should be assessed for channelopathy depolarization.
Implantable cardioverter defibrillator is the first-line therapy for patients with BrS with a history of malignant arrhythmia VT/VF or syncope.
The most appropriate treatment, especially for high-risk Brugada patients is ICD implantation. Therefore, it is important to identify patients at high risk for ICD implantation. About 8% of heart attacks occur in primarily asymptomatic patients (3). However, high risk patients include children with a history of seizures, syncope, resuscitated sudden death, persistent ECG pattern of Brugada syndrome, drug-triggered ECG changes or history of sudden death in the patient’s family. Due to negative physical and social effects, ICD indication in low annual rate of arrhythmic events in asymptomatic patients versus in patients with VF and in patients with syncope needs careful judgment.
Pharmacotherapy with quinidine is an alternative to ICD implantation or during the pre-ICD phase (13). In some cases, hydroquinidine has been used for children with Brugada syndrome type I with the 28-month-follow up being eventless (4). In some articles, it has been suggested not to use quinidine in the prevention of VF. Because of this controversy, its use in children is limited (14).
Despite successful utilization of quinidine, use of antiarrhythmic class IA drugs in Brugada syndrome is contraindicated. Another drug that is being investigated is Tedisamil, which blocks (Ito) stronger and more effective than quinidine (15, 16). Some other drugs that may be effective in the treatment of Brugada syndrome include beta-adrenergic such as isoproterenol and phosphodiesterase inhibitor III such as cilostazol. Cilostazol increases heart rate by effecting Ica and reducing Ito and thereby can be used in the treatment of Brugada syndrome (1, 17).
Genetic background and electrophysiological and clinical characteristics in the last two decades has revealed parts of BrS. evolution and the role of genetic mutations and polymorphisms while other confuse factors, such as fever and gender, need further research. In patients with frequent VF, catheter ablation may be a treatment option and will help analyze the pathophysiology of BrS.
3.1. Conclusion
It seems appropriate to perform an ECG on all children with fever and pneumonia with positive family history of sudden death. Moreover, Brugada syndrome should be suspected in all children with fever, in whom ventricular arrhythmias have occurred. Careful evaluation of fever in these patients and prompt treatment with antipyretics should be fast and accurate.