1. Background
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder, which is the most widespread cause of dementia and its incidence will continue to increase rapidly as the population ages (1). It is characterized clinically by the deposition of beta-amyloid (Aβ) plaques and tau proteins in the brain that leads to progressive cognitive function loss in AD (2). In addition to the risk factors for AD mentioned above, there are others like obesity (3), cardiovascular disease, high cholesterol, blood pressure, and sedentary (4). Of those, inactivity is one of the most important risk factors for AD (4).
Memory which allows animals to encode, retain and retrieve information declines in the early stages of AD and often precedes the biological hallmarks (5). The brain’s hippocampus is a key player in these processes (6). It is shown that the hippocampus-dependent learning and memory, a component of cognitive function, declines in the AD model of animals (7).
No effective treatment has yet been successfully found to treat all pathological aspects of AD (8). Therefore, special attention has been applied to the non-pharmacological prevention strategy with minimal cost and adverse effects (9, 10), and especially physical aerobic training (11, 12). Recent studies suggested that resistance training could improve several aspects of cognition (13, 14) and prevent the age-related cognitive decline of memory among elderly people (15). For example, studies indicated that weeks of progressive resistance training improves the hippocampus-dependent memory in the rats (16) and aged people (17). Even a single bout of resistance training could improve contextual memory in the Wistar rats (18). In patients with AD, Garuffi et al. revealed that resistance training improves the performance of activities of daily living like agility, flexibility, strength, and balance (19). Also, in a diabetic rat model, resistance training ameliorated cognitive deficits (20). Recently, Pena et al. demonstrated that resistance training in the AD mice model could affect hippocampal factors and attenuates memory deficits (14). So, to our knowledge, little is known about the effects of resistance training as a non-pharmacological preventing or ameliorating the memory loss on AD samples, and also it is yet unclear which type of training is preferable in this field.
2. Objectives
The aim of this study was to determine the effects of resistance training in comparison with aerobic training on spatial learning and memory in a rat model of AD using MWM.
3. Methods
3.1. Animals
Male Wistar rats (4 months and 250 ± 10 g) were obtained from the Animal House of Tabriz University of Medical Sciences. They were housed six per standard polycarbonate cage with standard sawdust as bedding (temperature 21 ± 2°C, 12/12 light-dark cycle). Rats were fed a standard pellet diet which had ad libitum access to food and water. Animals were divided into six groups (n = 6): Control; aerobic training (treadmill); resistance training (resistance); AD rats without training (Aβ + normal saline); AD rats with aerobic training (Aβ + treadmill); and AD rats with resistance training (Aβ + resistance). This research project has been reviewed by the Research Ethics Committee of Sport Sciences Research Institute and was approved according to compliance with Ethical Standards in Research of the Ministry of Science, Research and Technology, with the code IR.SSRI.REC.1399.822.
3.2. Induction of Alzheimer's Rat Model
Amyloid-β25-35 (Sigma-Aldrich, Beijing, China) was suspended at a concentration of 1 μg/μL. To obtain the aggregated form of Aβ25-35, the peptide solution was placed in an incubator at 37°C for 72 h. Aβ25-35 (10 nmol/10 μL) by intracerebroventricular (i.c.v.) injection was used in the brain ventricles rats with the coordinates AP = -1.2 mm, ML = ± 2 mm, DV = -4 mm. Alzheimer’s disease rats started training after one week of Aβ25-35 injection.
3.3. Aerobic Training Protocol
To familiarize the rats with the training, rats ran on the treadmill (10 m/min, 10 minutes, 3 days/weeks). According to a recent study (13, 21), the training protocol included three steps as presented in Table 1.
| Steps | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1- Warm-up | 10 minutes warm up at a speed of 10 m/min | |||||||
| 2- Main body | First week | Second week | Third week | forth week | Fifth week | Sixth week | Seventh week | Eighth week |
| Speed (m/min) | 12 | 15 | 17 | 20 | 20 | 20 | 20 | 20 |
| Time (min) | 10 | 10 | 15 | 20 | 25 | 30 | 35 | 40 |
| 3- Cool-down | 10 minutes cool down at a speed of 10 m/min | |||||||
Aerobic Training Protocol on Rodent Treadmill
3.4. Resistance Training Protocol
Before any resistance training, animals were submitted to three familiarization sessions (three trials per day) on an 80° inclined vertical ladder apparatus (110 × 18 cm) with 2-cm grid steps, as well as, with a housing chamber at the top of the ladder (16). In the first, second, and third familiarization trials, rats were placed on the top, middle, and bottom of the ladder, respectively. The resistance training protocol (10 repetitive dynamic movements to reach the chamber with the 30 s to 60 s rest) was modified from previous studies (3, 22) (Table 2). At the beginning of the training, 30% of the body weight was attached to the rats' tails, which gradually increased by 10% per week.
| Weeks | First Week | Second Week | Third Week | Forth Week | Fifth Week | Sixth Week | Seventh Week | Eighth Week |
|---|---|---|---|---|---|---|---|---|
| Load a | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 |
Resistance Training Protocol
3.5. Morris Water Maze Test (MWM)
Rats performed Morris water maze test (MWM) 24 hours after training protocols. The maze contained a black circular pool (diameter: 210 cm) filled with water (temperature: ~ 23°C, depth: 40 cm) located in a room with visual cues on the walls. A black platform (10 cm in diameter) was underwater 2 cm from the surface. The pool was abstractly separated into four quadrants with four starting points (N, S, W, or E) (23, 24).
3.6. Spatial Learning
In this task, rats received 4 trials/day over five days. A trial began when the rat was randomly placed in the water at one of the four starting points, with its head facing the wall. The rat had the 60 s to discover the platform; if an animal did not succeed to find the platform within the allocated time, it was guided to the platform and left on it for 30 s. The order of starting points in every trial was different. After each trial, animals were dried and returned to their cages for the 30 s. The time to find the platform in each trial (escape latencies) and the mean latency for every training day were measured (23, 24). To record the escape latency, a video camera was installed above the pool and connected to a computer.
3.7. Spatial Memory
To access spatial memory, a single probe trial was conducted 24 h after the last acquisition day, with the platform removed. During this test, animals were placed in the opposite position of the platform quadrant in the pool. Here, the time spent in the platform quadrant (time in the target quadrant) was measured (23, 24).
3.8. Statistical Analysis
Data are expressed as the mean ± SEM and analyzed using SPSS software (version 22). Behavioral performance was analyzed using a two-way repeated-measures analysis of variance (ANOVA) followed by Tukey’s post hoc tests for multiple comparisons.
4. Results
4.1. Spatial Learning Tasks
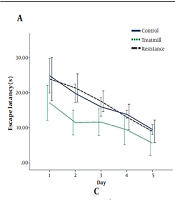
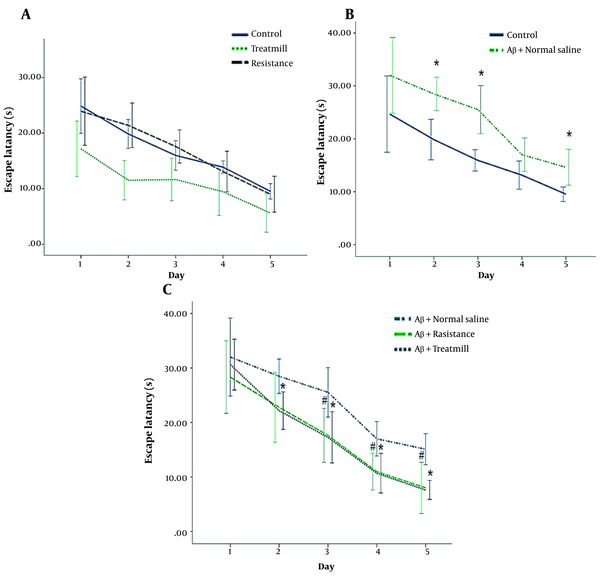
The results showed that the mean escape latency was significantly different between the Control and Aβ + normal saline groups (P < 0.05; Figure 1B). This test showed that on the second, third, and fifth days, the AD rats performed worse in finding the hidden platform compared to the control group (P < 0.05). However, there was no significant difference in the mean escape latency between the control, treadmill, and resistance groups (P < 0.05; Figure 1A). In addition, the mean escape latency was significantly better on the second, third, fourth, and fifth days in the Aβ + Resistance group and on the third, fourth, and fifth days in the Aβ + treadmill group compared to the Aβ + normal saline group (P < 0.05; Figure 1C). Notably, the post hoc analysis had not indicated any significant differences in escape latency values between the two kinds of training (P < 0.05). In other words, both treadmill and resistance trainings improved spatial learning performance in the rat model of AD.
Mean escape latency of rats in five days at different groups in the MWM spatial learning task. A, mean escape latency in control, treadmill and resistance groups; B, mean escape latency in Aβ + normal saline and the control groups. Aster (*) indicates significant differences between the Aβ + normal saline and the control groups (P).
4.2. Spatial Memory Task
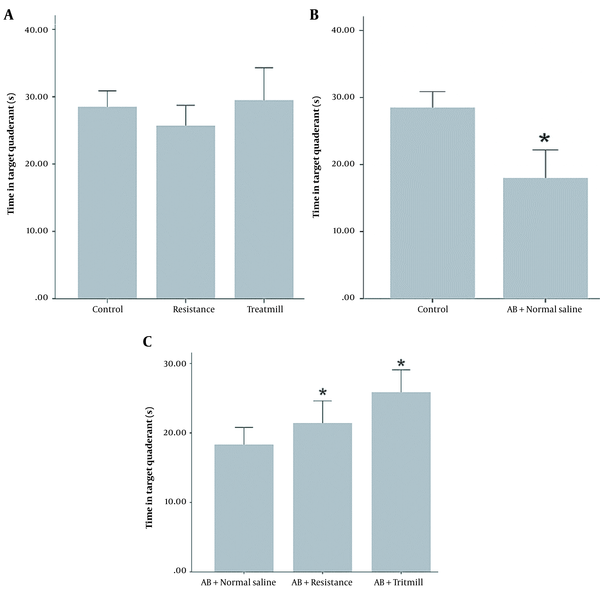
The results indicated that in the probe test, the time in the target quadrant significantly decreased in the AD rats compared to the control group (P < 0.05; Figure 2B). However, there was no significant difference in the time in the target quadrant between the control, treadmill, and resistance groups (P < 0.05; Figure 2A). Also, Aβ + resistance and Aβ + treadmill group significantly spent more time in the target quadrant compared to the Aβ + normal saline group (P < 0.05; Figure 2C). On the other hand, the post hoc analysis did not show a significant preference in the time target quadrant for the resistance training compared to the treadmill group in improving the spatial memory of AD rats. In other words, both treadmill and resistance trainings improved spatial memory in the AD rat model without any superiority over each other.
Time in target quadrant at different groups in the MWM spatial memory task. A, time in target quadrant in control, treadmill and resistance group; B, time in target quadrant in Aβ + normal saline group compared with the control; C, time in target quadrant in Aβ + treadmill and Aβ + resistance group compared with the Aβ + normal saline group in the MWM reference memory task. Aster (*) indicate significant differences between the two groups (* P < 0.05).
5. Discussion
According to studies, the factors that protect the heart, will protect the brain and reduce dementia-related diseases, and it seems that physical activity is one of those (4, 25). It is also stated physical inactivity are primary risk factors for dementia and AD (4). In recent decades, many studies as our study have well established that aerobic training has a positive effect on cognitive function and inhibits the process of memory loss in AD (26, 27). But about the effect of resistance training on cognitive function especially in AD cases there are fewer studies. In this study, we evaluated the effects of aerobic and resistance training on spatial learning and memory in the rat model of AD by using the MWM. Our results showed that both types of training could improve spatial learning and memory and inhibit memory loss in AD rats without any significant differences between them.
Our results were in agreement with the previous study that demonstrated 5-month voluntary running in the wheel-running results in a decrease in extracellular Aβ plaques in the frontal cortex and enhances the rate of learning and memory of TgCRND8 animals in the MWM, with significant reductions in escape latencies over the first 3 (of 6) trial days (28). Similar to previous research it has been shown that voluntary wheel running for 10 weeks also reduced all the neuropathological hallmarks of AD (neuronal loss, phosphorylated tau protein, Aβ burden, Thioflavin-S-positive plaques, astrogliosis, and Aβ oligomers) and improved spatial memory performance and hippocampus volume in the transgenic mouse model of AD (26). Consistently, other research showed that voluntary wheel running could improve learning and memory and increase neurogenesis in aged mice (29). Also revealed that treadmill training improves short-term and spatial memories by enhancing neurogenesis and suppressing apoptosis in the hippocampal dentate gyrus of old-aged rats (30). Recently reported that endurance treadmill training prevents and reverses compromised spatial learning and long-term memory and upregulates mitochondrial oxygen consumption in AD Wistar rats (27). Similar to our finding, another study showed that 12 weeks of treadmill running could protect and reverse the cognitive deficit progression based on MWM via decreasing the levels of Aβ plaque and hyper-phosphorylated tau protein and upregulating mitochondrial function and hippocampal neurogenesis markers in triple transgenic (3xTgAD) AD mice (12). On the other hand, it is established that hippocampal glycogen and its metabolite lactate are crucial for hippocampus-dependent memory function (31). Aerobic exercise plays an important role in regulating glucose metabolism in the hippocampus (32). So, aerobic training is a very useful strategy for reducing the risk or delaying the onset of memory and learning loss in AD and elderly people.
Many researchers have shown that resistance training could enhance functional abilities and decrease the risk of falls and functional limitations in the elderly (33). Although studied far less than aerobic training, resistance training has been shown to affect cognitive performance in the aging samples (34). Similar to previous, Cassilhas et al. showed a significant effect of resistance training on cognitive and physical functions of the elderly (35). Recently shown that even a simple resistance training session, using only body mass for resistance, may be an effective method for preventing the age-related cognitive decline of inhibitory control and working memory among elderly people (52 - 81 years) (15). Most recently in 2022, Serra et al. also revealed that 12-week resistance training improves the learning and spatial memory, as well as, neurotransmission-related and hippocampal metabolomic profiles in aged Wistar rats (17). Although the cognitive-related benefits of resistance training are well-known in the elderly population, very little is identified in the AD group. In agreement with our study, 9-week weighted ladder climbing as a resistance training reduced the Aβ burden in the hippocampus concurrent with increased concentrations of IGF-1 in 3xTgAD Mice (14). Similarly, Ashofteh et al. in 2022 found that resistance training could up-regulate the neurotrophins profile in the hippocampus of AD rats (36). These results were in agreement with our findings that showed resistance training could improve learning and spatial memory in the AD rat model.
In consistence with our results, only one study revealed positive effect of both trainings on cognitive function with no significantly differences between them (37), which showed that either training are beneficial in cognitive performance. Findings suggest that Brain-derived neurotrophic factor (BDNF)/Tropomyosin receptor kinase B (TrkB) and β-Calcium/Calmodulin-dependent kinase type II (CaMKII) mediates the effects of aerobic training on cognitive function (13), in a process in which energy metabolism (AMP-activated protein kinase (AMPK)) probably play an important role (38). But, resistance training via increasing the level of synaptic proteins, (synapsin I, synaptophysin), insulin-like growth factor 1 (IGF-1)/IGF-1R, and protein kinase B (Akt) pathway improves the spatial learning and memory (13). Therefore, the molecular pathway of their positive effect on cognition performance is different and requires further studies.
5.1. Conclusions
Our results demonstrate that spatial memory and learning deficits in AD rat model were reversed by both 8 weeks aerobic and resistance trainings, without any significant superiority over each other. Therefore, both trainings seem to be a helpful non-pharmacological approach to delay memory loss in AD.