1. Context
The outbreak of the novel coronavirus 2019 (COVID-19) was recognized by the World Health Organization (WHO) in February 2020 and announced as a global public health emergency (1). According to the report released by the WHO, over 18 million confirmed cases with approximately 700,000 deaths occurred worldwide by August 2020, imposing a significant burden on the available capacity and infrastructures of healthcare systems (2-4). COVID-19 is an extremely transmittable disease, which could easily spread through respiratory droplets (5). Furthermore, the cohorting of COVID-19 patients and hospital-wide colonization based on the recommendations of infection prevention and control (IPC) professional societies increased the likelihood of acquiring nosocomial SARS-CoV-2 among healthcare workers (HCWs) and non-COVID-19 patients (6, 7). In addition to its global impact, COVID-19 poses the risk of nosocomial infections. A study in a pediatric dialysis unit of a university hospital in Münster, Germany indicated nosocomial infections in 50 COVID-19 cases, including HCWs, patients, and patient companions. The majority of reported COVID-19 cases have occurred due to close contact with HCWs and the inadequate use of personal protective equipment (PPE) (8). Similar healthcare-associated infection cases have also been reported in China, Canada, and South Korea (9-11).
Nosocomial infections or hospital-acquired infections (HAIs) are not present upon admission and often occur in the course of receiving healthcare services within 48 - 72 hours after admission to healthcare centers (12). According to the WHO, the incidence rate of HAIs is 3.5 - 12% in developed countries and 7.5 - 19.1% in low- and middle-income countries (13). Before the COVID-19 outbreak, an epidemiological study conducted in 14 countries indicated the mean prevalence of HAIs to be 8.7% (14). In addition, studies conducted in the United States have reported urinary tract infections (UTIs) to be the most common type of nosocomial infections, accounting for 36% of reported infections, followed by surgical site infections (SSIs; 20%), bloodstream infections (BSIs; 11%), and pneumonia/nosocomial pneumonia (2.1%) (15). Almost a similar trend was observed in a study conducted in the intensive care unit (ICU) of a university hospital in Ahvaz (Iran), indicating the prevalence of nosocomial infections to be 10%. In terms of infection type, the highest rate has been attributed to UTIs (41%) followed by respiratory infections (28%), SSIs (20.5%), and BSIs (10.5%), respectively (16).
In order to prevent nosocomial infections in healthcare settings, infection control measures are recommended, including the proper use of PPE, reformation of wards to comply with environmental health and safety standards, and training on precautionary measures (eg, frequent hand washing and physical distancing) (17). The current literature confirms that allocating a significant portion of resources to preventing the spread of COVID-19 might indirectly decrease attention to conventional HAI preventive measures (18). Furthermore, an effective response to COVID-19 might lead to a considerable PPE supply deficiency in hospital settings, which is essential to every HAI control program (19). These factors, along with a lower healthcare staff-to-patient ratio, more complexity of patients’ clinical conditions, and increased length of hospital stay, may increase the risk of cross-contamination among patients. Although the current literature suggests the significant impact of the COVID-19 pandemic on nosocomial infections, evidence is still scarce in this regard (20, 21).
2. Objectives
The present study aimed to systematically review the current literature in order to estimate the prevalence of HAI during the COVID-19 pandemic and provide evidence-based data to guide clinicians in adopting effective preventive strategies.
3. Methods
3.1. Databases and Search Terms
A comprehensive search was conducted via electronic databases such as EMBASE, Google Scholar, Scopus, PubMed, and Web of Science from the onset of the COVID-19 pandemic until the end of June 2021. The search terms included ((COVID-19 [title/abstract] OR COVID 19 [title/abstract] OR COVID-19 Virus Disease [title/abstract] OR (COVID 19 Virus Disease [title/abstract] OR COVID-19 Virus Diseases [title/abstract] OR COVID-19 Virus Infection [title/abstract] OR COVID 19 Virus Infection [title/abstract] OR COVID-19 Virus Infections [title/abstract] OR 2019-nCoV Infection [title/abstract] OR 2019 nCoV Infection [title/abstract] OR 2019-nCoV Infections [title/abstract] OR Coronavirus Disease-19 [title/abstract] OR Coronavirus Disease 19 [title/abstract] OR 2019 Novel Coronavirus Disease [title/abstract] OR 2019 Novel Coronavirus Infection [title/abstract] OR 2019-nCoV Disease [title/abstract] OR 2019 nCoV Disease [title/abstract] OR 2019-nCoV Diseases [title/abstract] OR Coronavirus Disease 2019 [title/abstract] OR SARS Coronavirus 2 Infection [title/abstract] OR SARS-CoV-2 Infection [title/abstract] OR SARS CoV 2 Infection [title/abstract] OR SARS-CoV-2 Infections [title/abstract] OR COVID-19 Pandemic [title/abstract] OR COVID 19 Pandemic [title/abstract] OR COVID-19 Pandemics [title/abstract]) AND ("infection cross" [title] OR "cross infections" [title] OR "healthcare associated infections" [title] OR "healthcare associated infection" [title] OR "health care associated infection" [title] OR "health care associated infections" [title] OR "hospital infection" [title] OR "infections hospital" [title] OR "nosocomial infection" [title] OR "nosocomial infections" [title] OR "hospital infections" [title])).
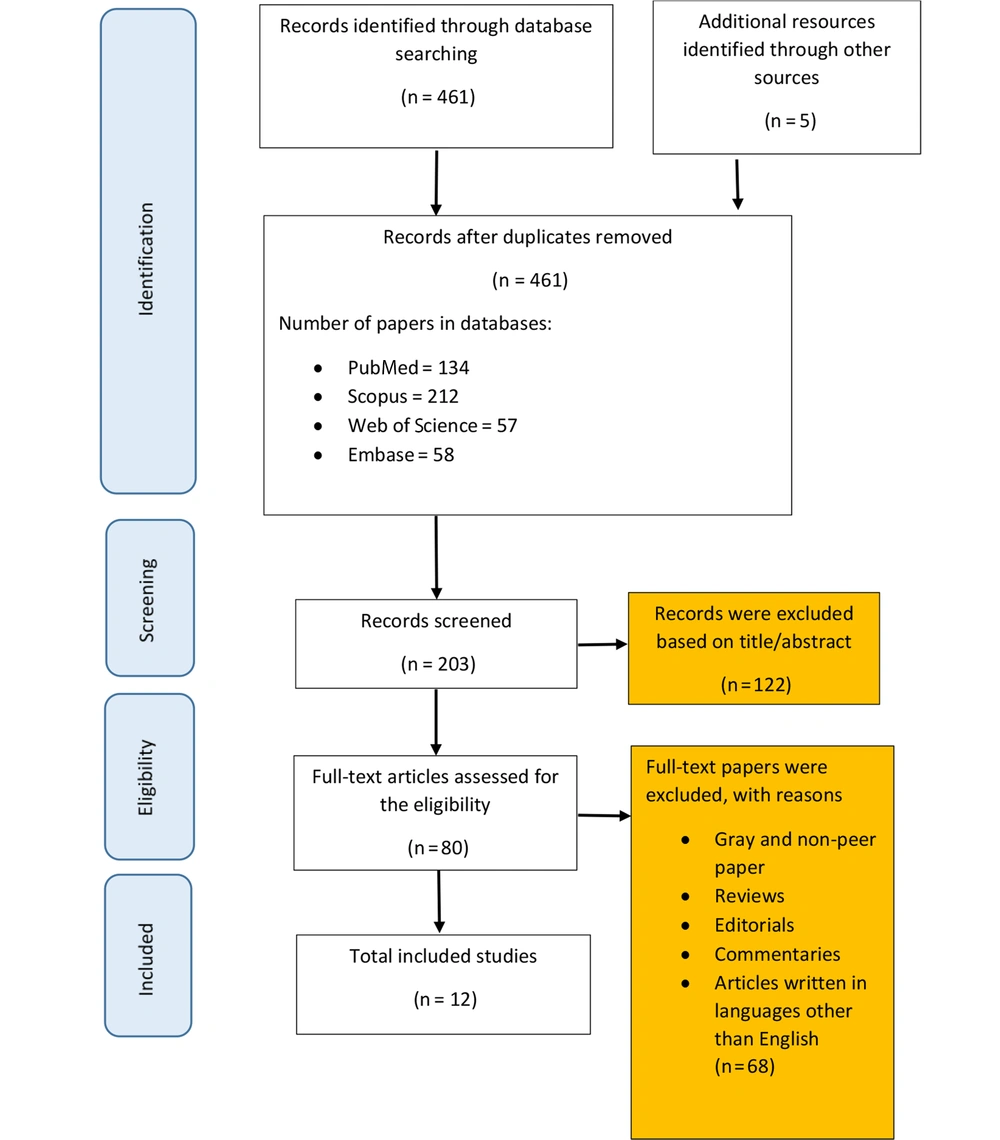
In the first step of searching the electronic databases, 466 records were identified. After removing duplicates, 203 articles remained for full-text review. In order to provide an updated estimation of the prevalence of HIAs during the COVID-19 pandemic, studies containing quantitative data on related measures were selected for further analysis. In addition, the reference lists of the selected articles and conference abstracts were screened to ensure that relevant data would be incorporated into the review process.
3.2. Inclusion Criteria
Studies were included if they reported quantitative data on the prevalence of HIAs and their determinants during the COVID-19 pandemic to find a set of articles based on the research keywords. Various types of observational studies were also considered eligible for further analysis, including cross-sectional studies, prospective studies, case studies, and cohorts. Furthermore, articles with available full texts and those published in English language from the beginning of the COVID-19 pandemic until June 2021 selected.
3.3. Exclusion Criteria
Excluded articles were interventional studies, review studies, reports, letters to the editor, books, case-control studies, and commentaries. In addition, studies with invalid methods and insufficient data and those focusing on diagnostic approaches, treatment methods, and medication were disregarded. Non-English papers published before the beginning of the COVID-19 pandemic or after June 2021 were excluded as well.
3.4. Article Selection Process
At the first step of the search process, 466 articles were identified. After removing duplicates, 203 records remained, 134 of which were published in PubMed, 212 were published in Scopus, 57 were published in Web of Science, and 58 articles were retrieved from EMBASE. After screening the titles and abstracts of the remaining records, 80 relevant publications were selected, which resulted in 12 articles after considering the inclusion and exclusion criteria. Figure 1 depicts the article selection process.
3.5. Data Extraction
A data extraction form was used to extract data by two independent investigators, including data on the author/authors’ name(s), study title, year of publication, study setting, sample size, age and gender of the subjects, country, the total prevalence of nosocomial infections, the prevalence of infections based on the infection type and modes of transmission, and the contributing factors (Appendix A in the Supplementary File).
3.6. Quality Assessment
The Newcastle-Ottawa scale (NOS) was used to evaluate the quality of the selected articles in terms of the robustness of the validity results. The NOS has a star system, by which a study is evaluated in terms of three main areas, including the ascertainment of exposure/outcome (three items), selection of study groups (four items), and their comparability (two items). In the quality appraisal of the selected articles, one point would be assigned to each of the items if the related area of the NOS was reported in the article. The highest achievable quality level was nine, and the studies with a total score of ≥ 7 were considered high-quality, while those with a score of < 4 were considered low-quality (22).
3.7. Statistical Analysis
Data analysis was performed by comprehensive meta-analysis in the Stata software version 14. To estimate the pooled prevalence of nosocomial infections in the reviewed studies, the random effects model was used. Statistical heterogeneity was also quantified using the I2 test. Due to the variability of the estimates based on different study settings and sociodemographic characteristics of the sample populations, subgroup analysis was also performed. In addition, Egger’s test was applied to assess publication bias.
4. Results
4.1. Prevalence of HIAs
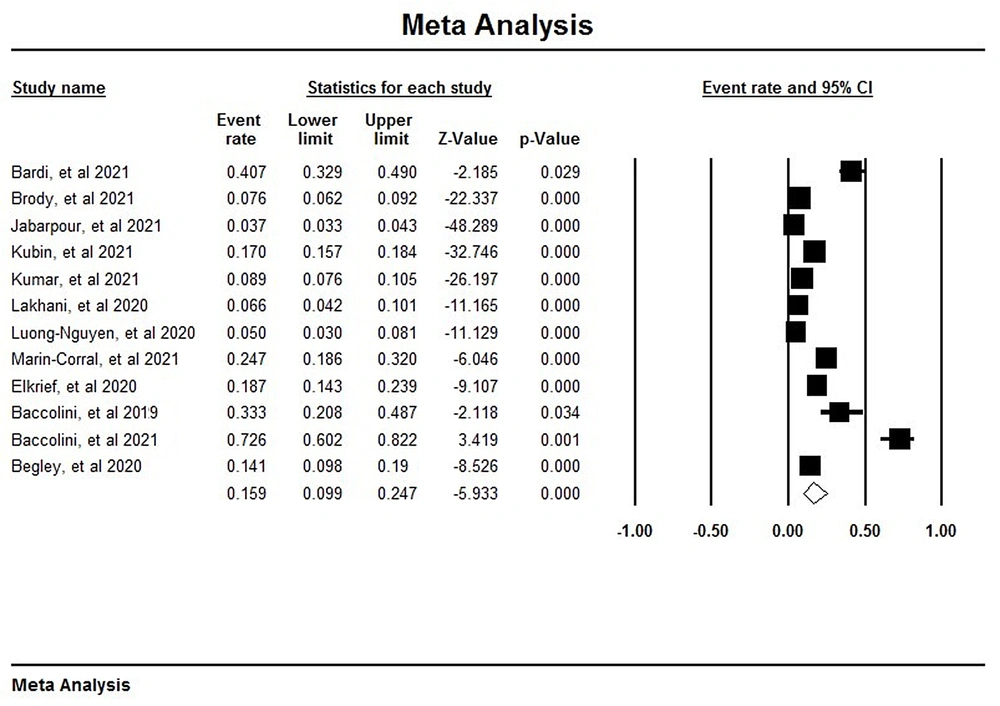
After analyzing 12 studies, it was observed that out of 13,294 participants, 718 cases acquired various types of nosocomial infections. Accordingly, the prevalence of HAIs during the COVID-19 pandemic was estimated at 15% (95% CI: 9 - 24) (Figure 2) (10, 19, 23-32).
4.2. Prevalence of HAIs Based on Country
According to the obtained results, the highest prevalence rate of nosocomial infections was reported in Italy (54%; 95% CI: 18 - 86), while the lowest prevalence rate was reported in France (5%; 95% CI: 3 - 8) and Iran (4%; 95% CI: 3 - 6), respectively (Table 1).
| Groups | Effect Size And 95% Interval | Test Of Null (2-Tail) | ||||
|---|---|---|---|---|---|---|
| Number Studies | Point Estimate | Lower Limit | Upper Limit | Z-Value | P-Value | |
| Australia | 1 | 0.14 | 0.10 | 0.20 | -8.53 | 0.00 |
| Canada | 1 | 0.19 | 0.14 | 0.24 | -9.11 | 0.00 |
| France | 1 | 0.05 | 0.03 | 0.08 | -11.13 | 0.00 |
| Iran | 1 | 0.04 | 0.03 | 0.06 | -48.29 | 0.00 |
| Italy | 2 | 0.54 | 0.18 | 0.86 | 0.18 | 0.86 |
| Spain | 3 | 0.20 | 0.07 | 0.46 | -2.21 | 0.03 |
| United States of America | 3 | 0.11 | 0.06 | 0.18 | -6.74 | 0.00 |
3.3. Prevalence of HAIs Based on the Infection and Microorganism Type
According to the information in Table 2, the highest infection rate in patients with HAIs belonged to COVID-19 infection (63%; 95% CI: 43 - 75), followed by bloodstream infections (39%; 95% CI: 56 - 24). On the other hand, the lowest rate of HAIs belonged to UTIs (16%; 95% CI: 9 - 26). Among the common organisms that caused HAIs, CoNS had the highest prevalence (63%; 95% CI: 43 - 75), followed by Enterococcus.
| Groups | Effect Size And 95% Interval | Test Of Null (2-Tail) | |||
|---|---|---|---|---|---|
| Point Estimate | Lower Limit | Upper Limit | Z-Value | P-Value | |
| Infections | |||||
| Bacteraemia | 0.37 | 0.31 | 0.45 | -3.33 | 0.00 |
| Bloodstream infection | 0.39 | 0.24 | 0.56 | -1.24 | 0.21 |
| COVID-19 | 0.63 | 0.43 | 0.75 | 3.26 | 0.00 |
| Pneumonia | 0.35 | 0.20 | 0.53 | -1.61 | 0.11 |
| Respiratory tract infection | 0.35 | 0.24 | 0.48 | -2.22 | 0.03 |
| Surgical site infection | 0.25 | 0.20 | 0.31 | -7.27 | 0.00 |
| Urinary tract infection | 0.16 | 0.09 | 0.26 | -5.31 | 0.00 |
| Other | 0.06 | 0.00 | 0.51 | -1.92 | 0.05 |
| Microorganism | |||||
| Acinetobacter spp. | 0.07 | 0.01 | 0.48 | -2.01 | 0.04 |
| Candida spp. | 0.05 | 0.03 | 0.09 | -8.92 | 0.00 |
| Coagulase-negative staphylococci | 0.28 | 0.18 | 0.41 | -3.19 | 0.00 |
| CoNS | 0.63 | 0.43 | 0.75 | 3.72 | 0.00 |
| E. coli | 0.16 | 0.11 | 0.24 | -7.13 | 0.00 |
| Enterobacter spp. | 0.06 | 0.04 | 0.11 | -9.02 | 0.00 |
| Enterococcus spp. | 0.42 | 0.30 | 0.55 | -1.19 | 0.24 |
| Klebsiella pneumoniae | 0.11 | 0.03 | 0.33 | -2.98 | 0.00 |
| Pseudomonas aeruginosa | 0.18 | 0.11 | 0.30 | -4.45 | 0.00 |
| Staphylococcus aureus | 0.08 | 0.02 | 0.34 | -2.74 | 0.01 |
| Other | 0.25 | 0.14 | 0.41 | -2.92 | 0.00 |
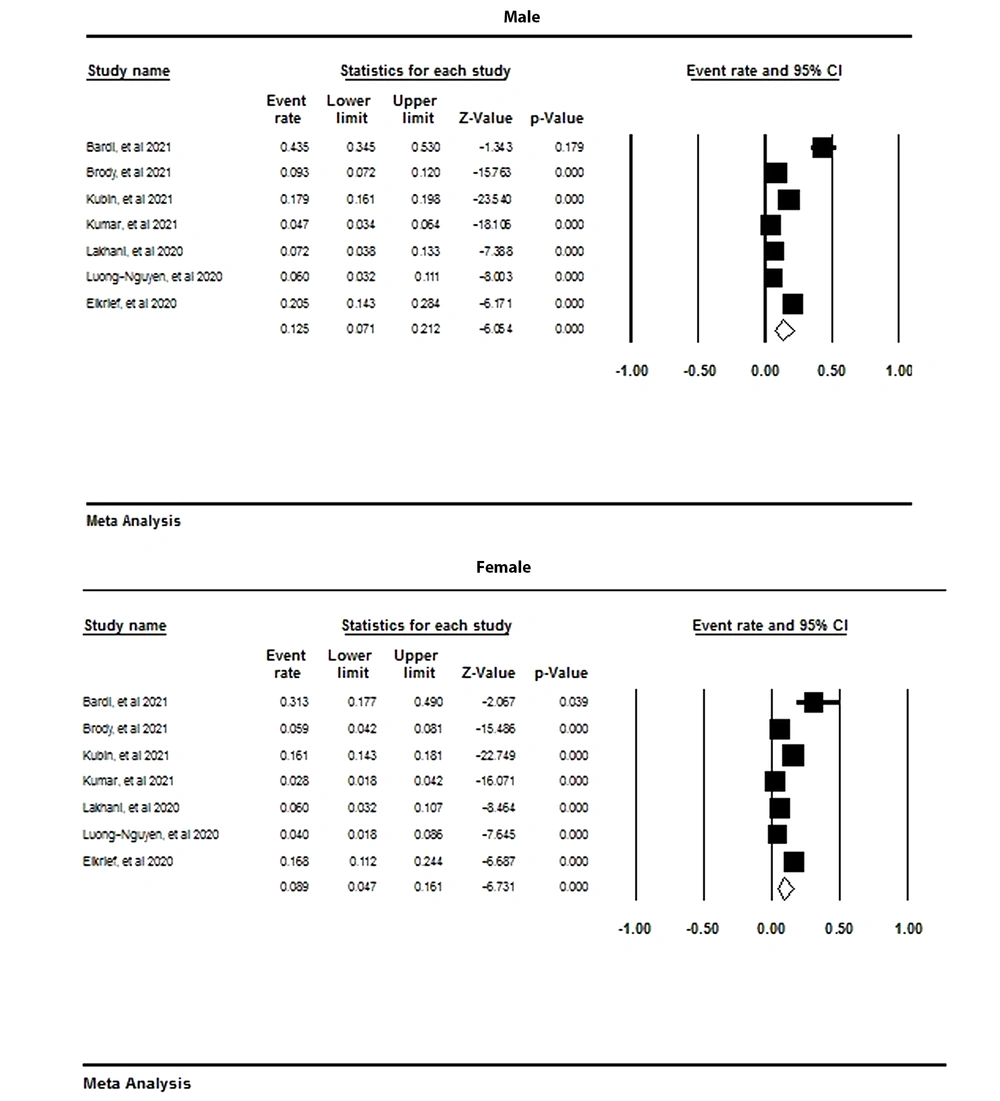
4.4. Meta-analysis Based on Gender
As is depicted in Figure 3, the prevalence rate of nosocomial infections in men was reported to be 12% (95% CI: 7 - 21), while it was estimated at 8% in women (95% CI: 16 - 4).
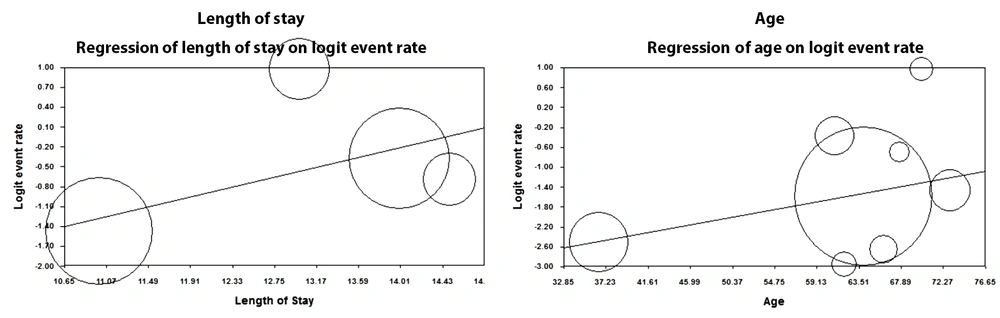
4.5. Meta-regression Based on Age and Length of Hospital Stay
According to the reviewed studies, the prevalence of nosocomial infections increased with age. In fact, one unit of increase in the age of individuals increased the prevalence of nosocomial infections by 0.03 (P < 0.05). Furthermore, a significant, direct correlation was reported between the length of hospital stay and nosocomial infections as one unit of increase in the length of hospital stay resulted in the increased rate of nosocomial infections by 0.35 (P < 0.05) (Figure 4).
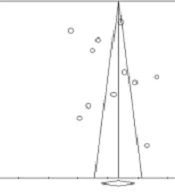
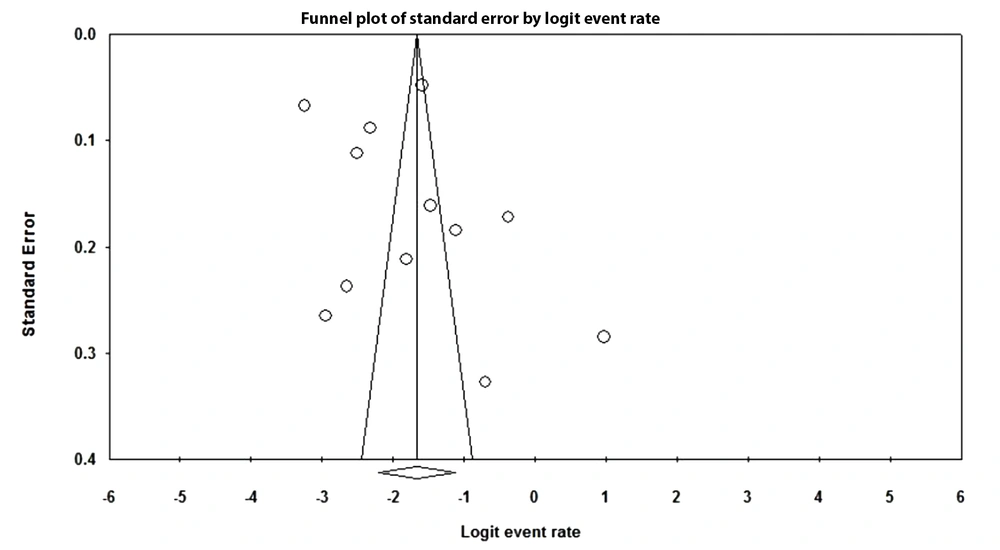
4.6. Publication bias
As is depicted in Figure 5, no evidence of publication bias or other study effects was observed. Moreover, the results of Egger’s regression test showed a higher value than 0.1 (two-tailed P-value = 0.43), which indicated no bias.
5. Discussion
5.1. Overview
To the best of our knowledge, this is the first systematic review and meta-analysis conducted globally on the prevalence of nosocomial infections, as well as identifying the risk factors for HAIs, with an emphasis on the demographic characteristics of the sample population and the length of hospital stay. The present study aimed to evaluate the prevalence of HAIs based on the type of infections and the causative pathogens. According to the results of the reviewed studies, the pooled prevalence of nosocomial infections during the COVID-19 pandemic was 15%, while the prevalence of this infection before the COVID-19 pandemic was estimated at 3.5 - 12% in developed countries and 5.7 - 19.1% in low- and middle-income countries (13). Similarly, the official data published in several studies indicate that 12 - 15% of nosocomial infections are hospital-acquired SARS-CoV-2 infections (9, 12, 22).
In a research conducted by Wang et al., patient-to-patient transmission was considered to be the major cause of infection in 12.3% of hospitalized patients infected by the 2019 novel coronavirus pneumonia (9). Furthermore, Rhee et al. reported that 15% of COVID-19 cases were probably hospital-acquired (33). In a systematic review and meta-analysis performed by Zhou et al., the prevalence rate of coronavirus 2 infections acquired in hospitals was 44%, and most of the cases were reported among medical personnel, particularly nurses and physicians. Meanwhile, only 2% of the infected cases were reported in inpatients and visitors (5).
A research conducted in the United States during the outbreak of COVID-19 also indicated that almost 65% of HCWs with symptomatic COVID-19 infection delivered care services to uninfected patients and might have transmitted the infection to patients or other staff (34). Therefore, the widespread and rapid outbreak of hospital-acquired COVID-19 in hospital settings during the pandemic highlights the importance of the accurate and timely diagnosis of all infected individuals and taking strict isolation precautions to minimize the spread of the infection in healthcare institutions. Despite an increasing trend in the rate of HAIs in recent years, a significant percentage of these infections could be avoidable through a strict surveillance system, adherence to infection control standards, and taking preventive measures (35).
5.2. Prevalence of Nosocomial Infections Based on Country
According to the current review, the prevalence of HIAs during the COVID-19 pandemic varied across different countries. The countries that reported the largest number of SARS-CoV-2 infection cases (eg, China and Italy) have been unable to manage the crisis effectively due to a lack of essential PPE, the inaccurate execution of physical distancing, and a shortage of medical staff. On the other hand, the countries that have been successful in achieving the required knowledge, skills, and resources to prepare for the management of the current pandemic have facilitated the timely diagnosis of COVID-19 cases, implemented strict isolation, and taken infection control and preventive measures, which have led to the lower prevalence of HIAs (12).
5.3. Prevalence of Nosocomial Infections Based on Infections
According to the results of the present study, bloodstream infections (39%) ranked second among nosocomial infections after hospital-acquired COVID-19. The current literature confirms this finding, indicating bloodstream, surgical site, and respiratory infections to be the most common types of HIAs (36). For instance, a review study conducted by Ghashghaee et al. reported bloodstream infections to be the most common type of HAIs, followed by SSIs and pneumonia (37). Another research by Birgani et al. revealed that 10% of inpatients in ICUs had symptoms of nosocomial infections, among which UTIs, respiratory infections, and SSIs were most common (16).
According to a rapid review study and meta-analysis conducted by Langford et al., pneumonia was the most common infection (38). In the mentioned study, the incidence rate of respiratory infections (eg, pneumonia and hospital-acquired COVID-19 infection) increased dramatically in low- and middle-income countries due to poor ventilation systems (39). Furthermore, an analysis of patients with confirmed COVID-19 revealed that respiratory tract infection was the most prevalent type of nosocomial infections, especially in ICUs. Other than bacteria and fungi, viruses also play a key role in transmitting nosocomial infections as they could rapidly spread through respiratory droplets and hand contact (40-43). In order to effectively prevent HAIs, it is essential to design proper organizational structures and properly comply with infection control and preventive guidelines (43, 44).
5.4. Conclusions
This review study indicated a strong association between the length of hospital stay and the prevalence of nosocomial infections, which is in line with the previous studies in this regard (45-47). According to Zhan and Miller, prolonged ICU admission (more than one week) could cause severe complications, which significantly increase the risk of HAIs (48). In terms of age- and gender-related risk factors, several studies have shown that male patients are at an increased risk of nosocomial infections. The findings in this regard also demonstrate that older age contributes to the higher probability of HAIs. In fact, most of elderly hospitalized patients have comorbidities and a poor immune response, which increase their susceptibility to COVID-19 infection (49). Therefore, special attention should be paid to the elderly in developing and implementing preventive and control strategies for nosocomial infections.
Hospital-acquired COVID-19 is an important public health issue, which requires a comprehensive review of its root causes in order to effectively implement corrective strategies and prevent similar cases. Since previous studies have shown nosocomial infections to be an important consequence of the COVID-19 pandemic (particularly in elderly and high-risk populations with prolonged hospital stay), the early detection of infected individuals could be a key step toward improving the quality of response toward the current pandemic.
Findings also highlight the importance of protecting patients against the transmission of COVID-19 from healthcare staff. Using proper ventilation systems in hospital wards, maintaining a safe physical distance, adherence to isolation standards, and provision of adequate PPE are among the effective measures suggested in hospital settings so as to successfully control the consequences of the current pandemic.
5.5. Limitations of the Study
Although this is the first systematic review and meta-analysis to investigate the prevalence of HAIs and their determinants during the COVID-19 pandemic, it has a number of limitations. First, there are a limited number of studies investigating nosocomial infections during the COVID-19 pandemic, which restricted the findings, particularly in terms of the infection and microorganism type in different countries. Second, limiting the review to English articles papers the absence of grey literature might have restricted the comprehensiveness of the findings.