Due to population growth, there is an urgent need for food control around the world. The prevalence of diseases, caused by the consumption of contaminated food, has always been one of the major problems in different communities, as elimination of these diseases, especially in countries with a poor health status, is costly (
11).
Confectionery products (especially pastry cream) as nutritious foods are susceptible to the growth and multiplication of different microorganisms and transmission of microbial agents, causing food poisoning in consumers. The present findings and similar studies conducted in different parts of Iran and the world confirm the high risk of microbial contamination in such products, besides contamination transmission to consumers (
12-
14).
Overall, sources of contamination in pastry, especially pastry cream and sweet rolls, are as follows: Primary contamination of milk; use of local and non-pasteurized cream; non-compliance with the freezing cycle during storage; contamination of equipment, such as knives and cutting tools; improper manipulation by individuals working at confectionery workshops; poor personal hygiene of workers; and bacteria transmission from the staff hands and noses during processing and transportation phases (
15,
16). Regarding the type of constituents and preparation and design methods, pastries are more likely to be contaminated by pathogenic agents, such as Enterobacteriaceae (
17).
The results of the present study showed that 47 out of 350 pastry cream samples contained
E. coli, which is consistent with the results of previous studies conducted in other cities of Iran on the contamination of pastry cream with
E. coli. In this regard, Nikniaz et al. in a study from Tabriz, Iran, reported the rate of pastry cream contamination with coliform bacteria and
E. coli to be 38.8% and 48.8%, respectively (
3). In another study by Soltan Dallal et al. in Tehran, Iran, it was shown that the rate of
E. coli contamination was 4.2% in pastry cream (
18).
E. coli is one of the pathogens resistant to the majority of antibiotics, causing health problems in different countries (
19). Few studies in Iran have investigated the antibiotic resistance of
E. coli in food products. The research carried out by Saenz et al. indicated the highest antibiotic resistance of
E. coli to nalidixic acid (53%) (
20). This finding is not consistent with the findings of our study, as the rate of resistance was estimated at 12.27% in our study, which can be explained by geographical differences of these studies.
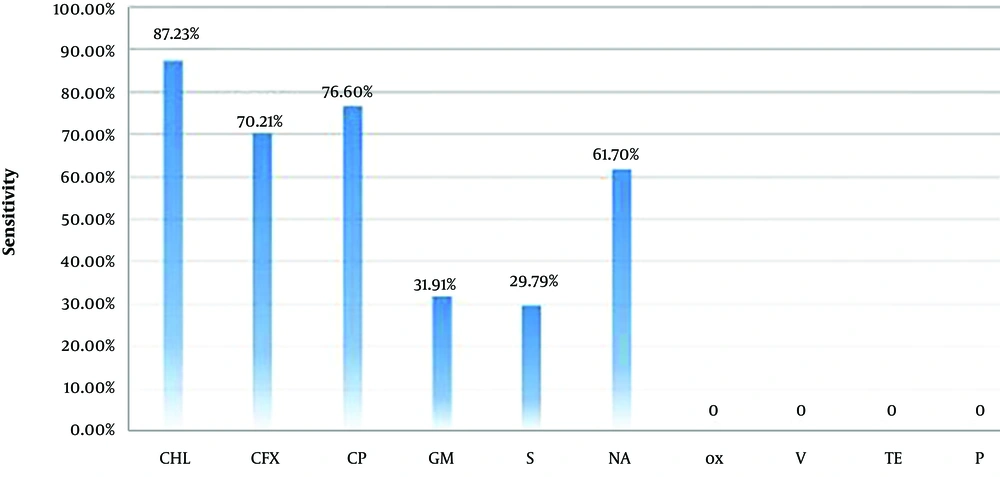
Moreover, in a study by Molaabaszadeh et al. on
E. coli isolates from traditional ice cream in Khoy, Iran, the highest and lowest resistance rates were 82.98% and 12.76% to oxacillin and ciprofloxacin, respectively. Similarly, in the present study, the highest resistance rate was 100% to oxacillin, and the lowest resistance was 89% to ciprofloxacin. In addition, Molaabaszadeh et al. reported antibiotic sensitivities of 40.43% and 61.7% to tetracycline and gentamicin, respectively, while resistance to these antibiotics was 55.31% and 25.54%, respectively (
21).
On the contrary, in the present study, the rates of sensitivity to tetracycline and gentamicin were 0% and 31.91%, respectively, while the rates of resistance to tetracycline and gentamicin were 100% and 19.15%, respectively. In another study by Mahdavi et al. on
E. coli isolated from traditional cheese in Maragheh, Iran, the rates of sensitivity to chloramphenicol and streptomycin were estimated at 84.35% and 31.2%, respectively (
22). These results are in agreement with the findings of the current study, as the corresponding rates were 23% and 29.8% in our study, respectively.
Additionally, in the study by Molaabaszadeh et al. on
E. coli isolates from urinary tract infections in Tabriz, Iran, the rates of sensitivity to gentamicin and nalidixic acid were estimated at 37.98% and 52.1%, respectively (
21). These results are similar to the findings of the present research. Moreover, in a study by Bonyadian et al. on
E. coli isolates from raw milk and pasteurized cheese in Shahrekord, Iran, the resistance rates of
E. coli to streptomycin and ciprofloxacin were reported to be 25.4% and 25.4%, respectively (
23). The corresponding rates in the present study were reported to be 10.63% and 14.89%, respectively. Considering multiple reports on the transmission of resistant
E. coli from dairy products to humans (
24) and similarity of antibiotic resistance of
E. coli in animal products and human infections, antibiotics should not be overprescribed (
25).
4.1. Conclusions
According to the present findings and similar research in Iran, E. coli is extremely resistant to antibiotics. The major sources of this type of food contamination include microbial contamination of raw materials, such as cream, poor personal hygiene of workers, and microbial contamination of utensils used for pastry production. Therefore, all these factors can influence the final product and cause contamination. To avoid the contamination of pastry cream with bacteria, some techniques, including cold chain management, employee personal hygiene, use of healthy raw materials, and proper thermal processes, should be adopted.