1. Background
Escherichia coli O157:H7 and Staphylococcus aureus are important foodborne pathogens (1). To control bacterial contamination, new approaches are required to identify the next generation of disinfecting agents (2). In comparison with organic reagents, inorganic antibacterial agents such as zinc oxide have received more attention in food applications because of their superior durability, heat resistance and biocompatibility (3, 4). To date, several studies have shown that zinc oxide nanoparticles (ZnO NPs) have toxicity toward extensive spectrums of Gram-negative and Gram-positive bacteria (5). Several studies have shown that various physicochemical properties, including surface reactivity, particle size, surface charge, agglomeration state, crystal structure, shape, and solubility can affect ZnO NPs toxicity (6).
Culture conditions can alter the physicochemical and biological properties of NPs. Miao et al. showed that acidification of the exposure medium increased the solubility of ZnO NPs (7). Reed et al. reported that solution temperature had an effect on dissolution of ZnO NPs in cell culture media. They showed that the solubility of ZnO NPs in Dulbecco’s modified Eagle’s medium (DMEM) and Roswell Park Memorial Institute medium (RPMI) reduced at 37°C in comparison with that in 20°C (8). Li et al. studied the influence of medium components on toxicity of ZnO NPs and found a significant difference in toxicity of ZnO NPs in various aqueous media (9). Li et al. showed that tannic acid decreased the ZnO NPs bioavailability and thereby decreased their toxicity to bacteria (10). Considering the correlation between culture media characteristics and antibacterial effect of ZnO NPs, the authors synthesized ZnO NPs with green and cost-effective methods and investigated their antibacterial activity. In an effort to further elucidate the antibacterial activity of ZnO NPs and study the importance of each variable, a series of experiments were carried out with variations in pH and temperature.
2. Objectives
The purpose of this study was to determine the interrelationship between reaction conditions and antibacterial activities of ZnO NPs. Therefore, the influences of pH and temperature as two principle growth parameters on the antibacterial activity of the ZnO NPs were studied at different concentrations of NPs. The relationship between cultural conditions and the antimicrobial behavior of ZnO NPs may be a new strategy to achieve the maximum antimicrobial activity.
3. Materials and Methods
3.1. Preparation of Zinc Oxide Nanoparticles
First, 0.02 moles of zinc acetate dehydrate (Merck, Germany) was dissolved in 50 mL of deionized water. Then, 0.40 moles of solid sodium hydroxide (Merck, Germany) was added slowly into the solution under magnetic stirring at room temperature, forming a transparent Zn (OH)42- solution. Thereafter, 2 mL of ionic liquid, 1-butyl-3-methylimidazolium bis (trifluoromethylsulfonyl) imide, [bmim][NTf2], was added to 3 mL of the above solution. The suspension was put into a domestic microwave oven (2.45 GHz, 850 W) in air. Of the output power of the microwave, 30% was used to irradiate the suspension for five minutes in a cycling mode (on for 10 seconds, off for five seconds). The white precipitate was collected by centrifugation (15000 rpm for 15 minutes), washed with deionized water and ethanol, and dried in vacuum oven at 40°C for 10 hours (11). Phase and purity of the products were analyzed using D8 Advanced diffractometer with Cu Kα radiation (λ = 0.15406 nm). The transmission electron microscopy was recorded by the LEO system (model 912 AB), operating at 120 kV for the samples.
3.2. Preparation of Zinc Oxide Nanofluid
The ZnO NPs were dispersed in glycerol (Merck, Germany) with the aid of a magnetic stirrer. To enhance the stability of the suspension, ammonium citrate (Merck, Germany) was used as a dispersant. Ammonium citrate prevents the growth and aggregation of ZnO NPs via electrostatic stabilization, resulting in a stable ZnO nanofluid. The weight ratio of the dispersant to the NPs was maintained at 1:1. The suspensions were stable at least for several months ie, no sedimentation of the particles in the samples was observed during this period (11). Particle size analyzer (Cordouan-Vasco 3) was used to determine the particle size distribution.
3.3. Bacterial Culture Conditions and Antibacterial Test
E. coli O157:H7 NTCC: 12900 and S. aureus ATCC: 25923 were used as standard Gram-negative and Gram-positive strains for antibacterial studies. Two-fold serial dilutions of ZnO NPs (0.09-1.5 mg mL-1) were made in tryptic soy broth (TSB) medium (HiMedia, India); 100 µL of each bacterial suspension with final concentration of 105 colony forming unit (CFU) mL−1 was transferred to 400 µL of ZnO NPs-containing culture medium. Then, 150 µL of each inoculum suspensions was placed in triplicate in a 96-well microtiter plate and incubated at 37°C on a reciprocal shaker (120 rpm). The growth of the cultures was monitored every two hours by measuring the optical density (OD) at 630 nm. The lowest concentration of the ZnO NPs that inhibited the growth after 24 hours of incubation was obtained as the minimum inhibitory concentration (MIC) of the ZnO NPs against the test bacteria. All the experiments were performed in triplicates.
3.4. Influence of pH, Temperature, and Concentration on Antibacterial Activity of Zinc Oxide Nanoparticles
The experiments were conducted at pH and temperature ranges in which bacterial cells can grow normally. Therefore, the antibacterial tests were performed at a pH range of 4-10 for E. coli O157:H7 and 5 - 10 for S. aureus and at different incubation temperatures (25 - 42°C). As mentioned above, 100 µL of the 24-hour-old bacterial suspensions were added to 400 µL of the culture medium with different concentrations of ZnO NPs (0.375 - 1.5 mg mL-1 for E. coli O157:H7; 0.09 - 0.375 mg mL-1 for S. aureus) at mentioned pH levels. After dispensing 150 µL of each inoculum per well, the 96-well microplate was incubated at different temperatures for 24 hours. These cultures were made in three wells. The bacterial growth was determined using the optical density of the samples every two hours.
It is clear that some harsh reaction conditions such as high acidic and alkaline pH reduce the growth of bacterial cells. Therefore, to exclude the inhibitory effect of reaction conditions on bacterial growth, the bacterial cultures without ZnO nanofluid at the same pH and temperature of the treated bacterial cultures were used as positive controls. Therefore, every sample of treated bacterial culture had its own positive control. The growth percentage of every positive control was considered 100% and the percentage of growth inhibition of every treated bacterial sample was calculated in comparison with its own positive control. Dispersant solution control was also established to exclude its antibacterial influence on the results of the experiment. To avoid the potential optical interference of the growing cultures caused by light-scattering properties of the NPs, the same liquid medium without bacteria, containing the same concentration of NPs, was incubated under the same pH and temperature (as blank control).
3.5. Data Analysis
The growth of the treated groups was compared to those of positive control groups to exclude the inhibitory effect of the reaction conditions on bacterial growth. Hence, the observed antibacterial activity referred to the antibacterial activity of ZnO NPs and it was not attributed to the experimental conditions such as acidic pH and temperature. The percentage of growth inhibition for both microorganisms in comparison with positive controls was determined via Equation 1:
Percentage of growth inhibition = 100 – [(growth at the presence of ZnO NPs)/(growth of positive control) × 100
The statistical significance of the parameter was determined at P value < 0.05.
4. Results
4.1. Characterization of Zinc Oxide Nanoparticles
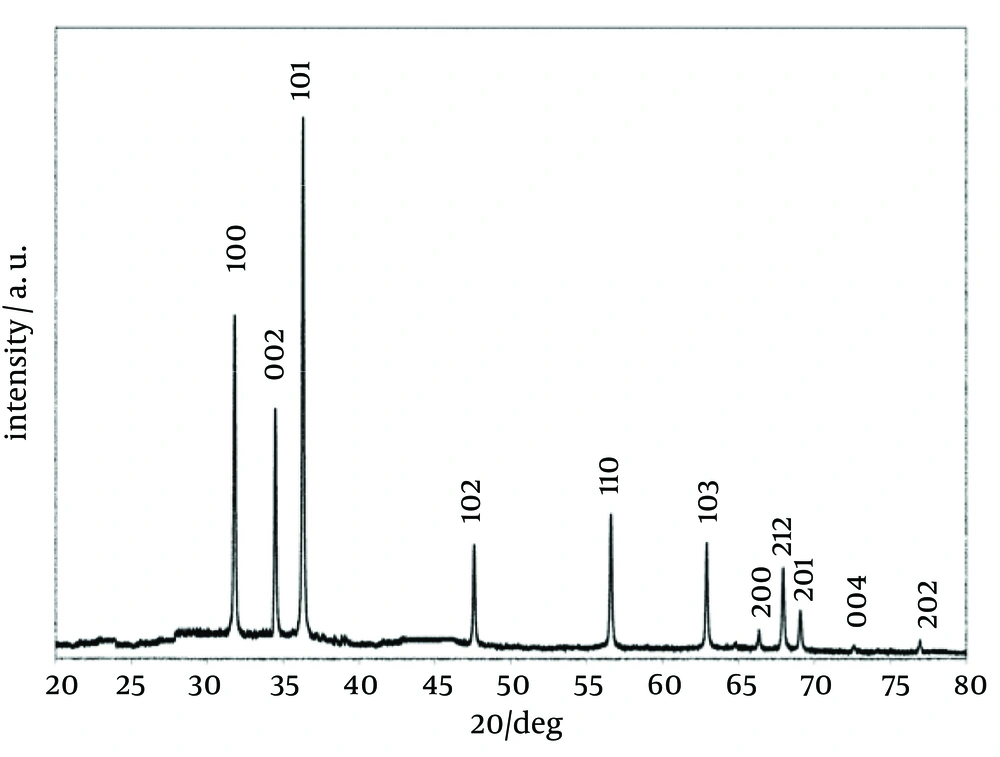
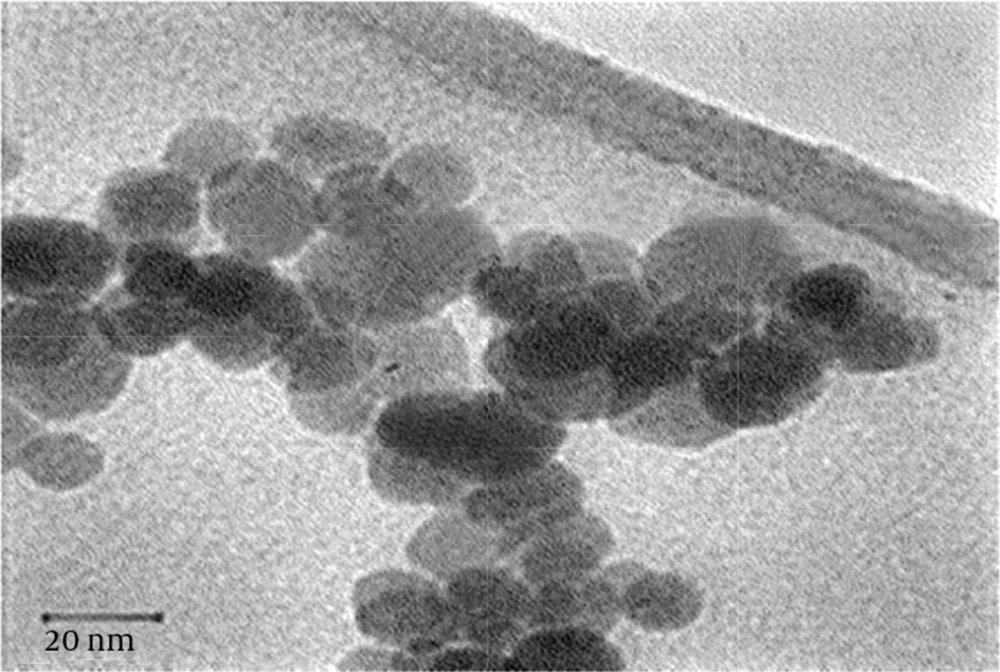
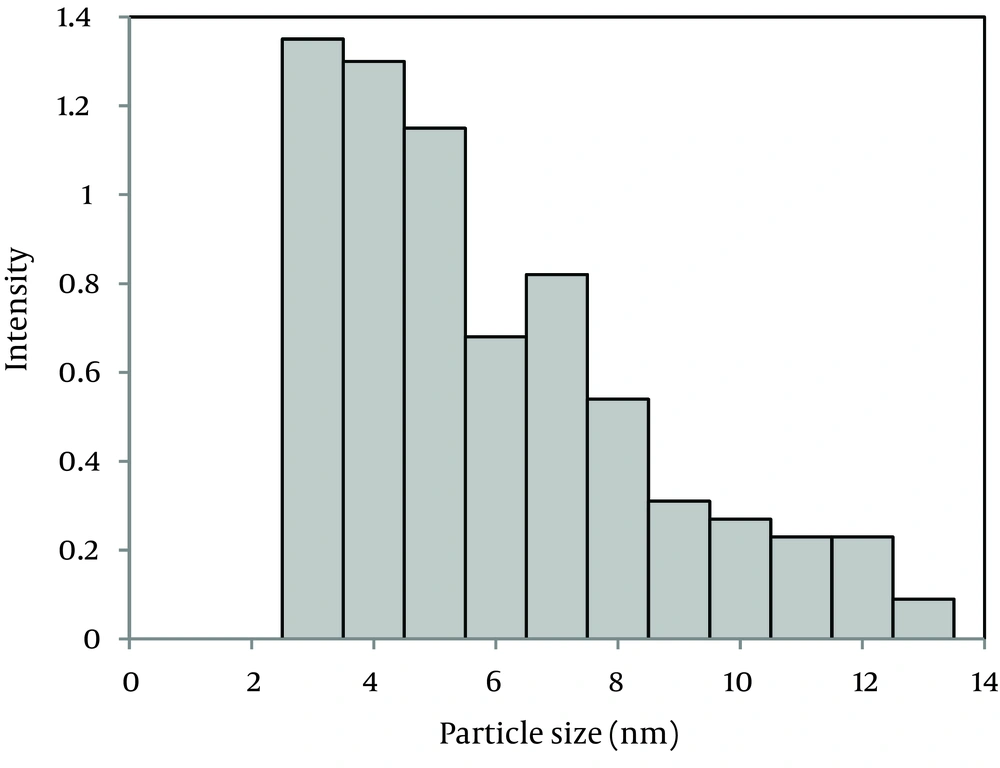
The X-ray diffraction (XRD) pattern of the nanoparticles is shown in Figure 1. All the peaks of (1 0 0), (0 0 2), (1 0 1), (1 0 2), (1 1 0), (1 0 3), (2 0 0), (1 1 2), (2 0 1), (0 0 4), and (2 0 2) reflections can be indexed to the known hexagonal wurtzite structure of ZnO with lattice constants of a = b = 3.242 Å and c = 5.205 Å. The peaks match well with those in the Joint Committee on Powder Diffraction Standards (JCPDS) card (No. 89 - 1397). The strong intensity and narrow width of ZnO diffraction peaks indicated that the resulting products were of high crystalline. Figure 2 is a transmission electron microscopy (TEM) image of the ZnO NPs. The TEM image shows uniform spherical ZnO nanoparticles with weak agglomeration. The average particle size of the ZnO NPs is 4.45 ± 0.37 nm according to the particle size distribution (PSD) (Figure 3).
4.2. Antimicrobial Activity of Zinc Oxide Nanoparticles Against Microorganisms
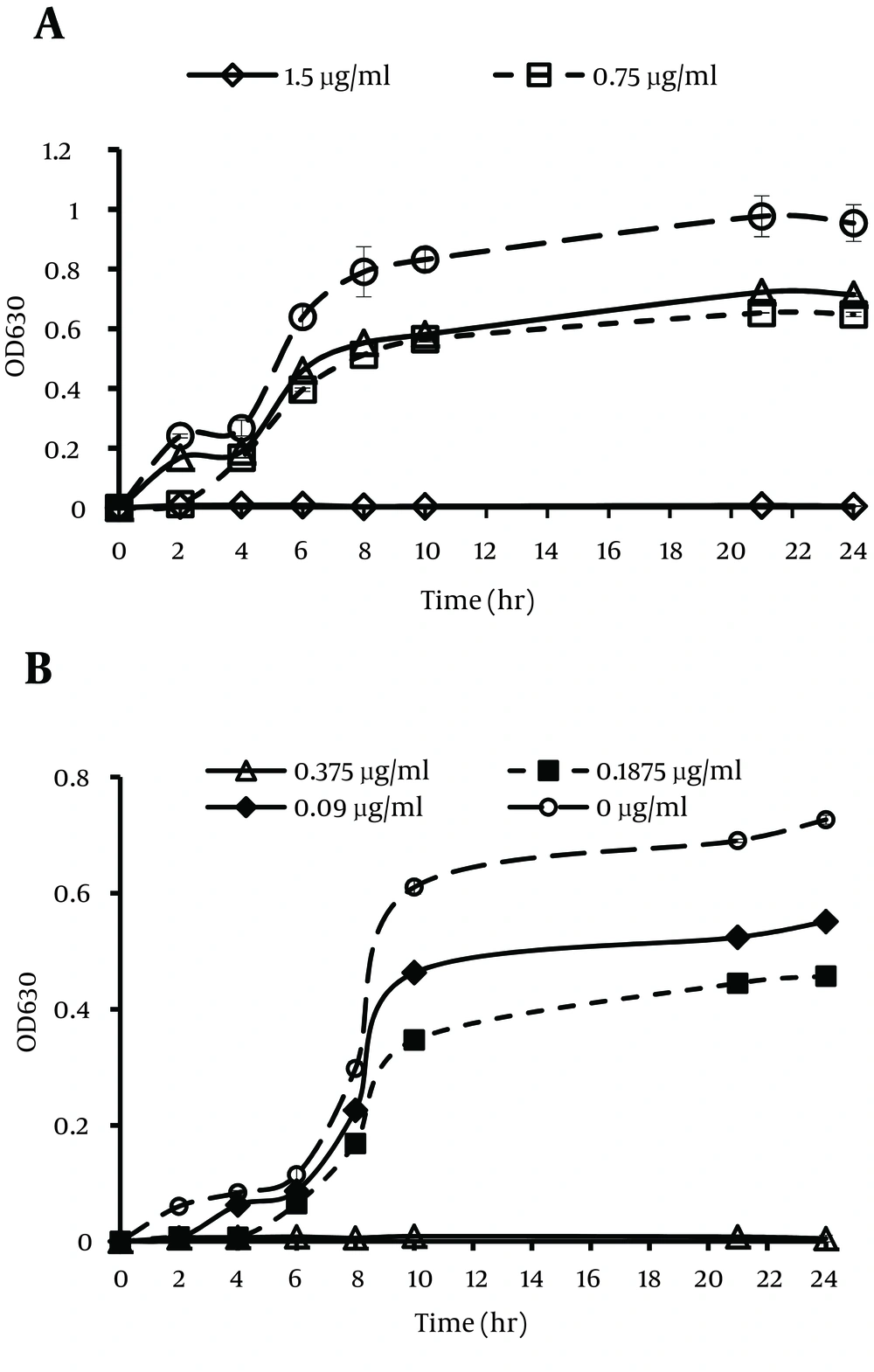
The inhibition of bacterial growth by ZnO NPs against E. coli O157:H7 and S. aureus are shown in Figure 4. The results of the antibacterial activity test of the dispersant showed a little effect on bacterial growth inhibition. Although the antibacterial activity of the dispersant was not significant, it was excluded from the total antibacterial activity of ZnO nanofluid to only have the antibacterial influence of ZnO NPs on the growth of bacterial cells. The bacterial growth of E. coli O157:H7 and S. aureus decreased as the concentration of ZnO NPs increased. The MIC values of ZnO NPs against E. coli O157:H7 and S. aureus were 1.500 and 0.375 mg mL-1, respectively. Different values of MIC against E. coli O157: H7 and S. aureus may be related to their different cell wall structures (12).
4.3. Effect of Process Parameters on the Antibacterial Activity of Zinc Oxide Nanoparticles
The response surface design using statistical software (Minitab 16) was used to pick factors that influence on the antibacterial activity of ZnO NPs (13). Analysis of the results confirmed that the antibacterial activity of ZnO NPs against both types of bacteria was dependent on the concentration of NPs, incubation temperature, and pH (P value < 0.05).
4.4. Influence of pH on Antibacterial Activity of Zinc Oxide Nanoparticles
The results showed that the overall influence of pH on the antibacterial activity of ZnO NPs against both bacteria was similar. As the pH increased from acidic to neutral, at concentrations below the MICs, the percentage of the growth inhibition of both microorganisms decreased. It suggests a pH-dependent mechanism for ZnO NPs antimicrobial properties (Table 1 and 2).
| Incubation Temperature, ZnO NP Concentration, µg.mL-1 | Percentage of Escherichia coli O157:H7 Growth Inhibition in Different pH | ||||||
|---|---|---|---|---|---|---|---|
| 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| 25ºC | |||||||
| 1.5 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 0.75 | 63 ± 2.68 | 61 ± 0.7 | 35 ± 0.7 | 27 ± 0.42 | 33 ± 0.7 | 35 ± 1.02 | 35 ± 0.63 |
| 0.375 | 48 ± 0.84 | 45 ± 0.71 | 27 ± 0.01 | 20 ± 0.21 | 25 ± 0.42 | 25 ± 0.42 | 25 ± 0.21 |
| 37 ºC | |||||||
| 1.5 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 0.75 | 79 ± 0.7 | 77 ± 0.98 | 52 ± 0.56 | 32 ± 0.91 | 41 ± 0.07 | 46 ± 0.56 | 49 ± 0.49 |
| 0.375 | 62 ± 2.21 | 59 ± 0.01 | 34 ± 0.55 | 25 ± 0.28 | 27 ± 0.14 | 29 ± 0.01 | 29 ± 0.91 |
| 42 ºC | |||||||
| 1.5 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 0.75 | 87 ± 0.28 | 84 ± 1.97 | 69 ± 0.77 | 40 ± 0.49 | 52 ± 0.07 | 57 ± 0.35 | 57 ± 0.14 |
| 0.375 | 78 ± 0.7 | 75 ± 0.49 | 51 ± 2.47 | 33 ± 0.01 | 38 ± 0.28 | 40 ± 0.35 | 45 ± 0.42 |
a Abbreviation: ZnO NP, zinc oxide nanoparticle.
| Incubation Temperature, ZnO NP Concentration, µg.mL-1 | Percentage of Staphylococcus aureus Growth Inhibition in Different pH | |||||
|---|---|---|---|---|---|---|
| 5 | 6 | 7 | 8 | 9 | 10 | |
| 25ºC | ||||||
| 0.375 | 100 | 100 | 100 | 100 | 100 | 100 |
| 0.187 | 52 ± .15 | 48 ± 1.2 | 23 ± 0.21 | 25 ± 0.91 | 26 ± 0.07 | 26 ± 0.35 |
| 0.09 | 37 ± 0.77 | 25 ± 0.21 | 19 ± 1.13 | 21 ± 0.7 | 22 ± 0.7 | 23 ± 0.28 |
| 37 ºC | ||||||
| 0.375 | 100 | 100 | 100 | 100 | 100 | 100 |
| 0.187 | 73 ± 0.14 | 69 ± 0.14 | 37 ± 1.41 | 39 ± 0.28 | 41 ± 0.35 | 41 ± 0.07 |
| 0.09 | 58 ± 0.01 | 46 ± 0.56 | 24 ± 0.07 | 27 ± 0.42 | 31 ± 0.77 | 33 ± 0.56 |
| 42 ºC | ||||||
| 0.375 | 100 | 100 | 100 | 100 | 100 | 100 |
| 0.187 | 89 ± 0.63 | 81 ± 0.14 | 48 ± 0.01 | 51 ± 0.56 | 53 ± 0.63 | 56 ± 0.21 |
| 0.09 | 74 ± 0.28 | 65 ± 1.48 | 27 ± 0.91 | 35 ± 0.7 | 39 ± 0.21 | 43 ± 0.14 |
a Abbreviation: ZnO NP, zinc oxide nanoparticle.
4.5. Influence of Temperature on the Antibacterial Activity of Zinc Oxide Nanoparticles
The influence of temperature on the antibacterial activity of ZnO NPs against S. aureus was similar to that of E. coli O157:H7. Heat treatment increased the antimicrobial activity of ZnO NPs against E. coli O157:H7 at the concentration levels below MICs (Table 1). Improved inhibitory effects of ZnO NPs toward S. aureus were also observed at higher temperatures (Table 2). A stronger inhibitory influence of ZnO NPs at acidic pH was also observed for all the temperatures (25-42°C).
5. Discussion
Based on the results, the inhibitory effects of ZnO NPs on the bacterial growth were influenced by the culture medium pH. The percentage of growth inhibition intensified, as the pH decreased from 7 to acidic pH. This finding was similar to the results obtained by Moreau et al. who reported that dissolution of nanoparticles increased under acidic conditions as well as in the presence of biological components such as amino acids and peptides (14). One of the possible paradigms of NP toxicity is the release of toxic ions from ZnO NPs under aqueous conditions (15). As a result, acidic pH increases the dissolution of ZnO NPs, which leads to higher toxic properties. Another systematic examination by Miao et al. suggested that the maximum total Zn+2 concentration increased as pH decreased from 9 to 7 (7). However, our results showed that shifting from neutral to alkaline pH has a minimal positive effect on the antibacterial activity of ZnO nanofluid towards both types of bacteria.
Huang et al. showed that in general, bactericides have a greatly improved efficiency when balancing with alkaline materials that dissolve the outer part of cell membrane (16). In another report, a consistent decrease in ZnO NPs solubility at 37°C compared with 20°C was observed (8). However, our results showed that the antimicrobial activity of ZnO NPs intensified at higher temperatures. Therefore, it is plausible to say that the other mechanism of antibacterial activity except that dissolution exists as temperature increases. In semiconductor NPs, the electrons excited by temperature are captured on active sites and then, they interact with oxygen (O2) to form superoxide radicals. The radicals produce hydrogen peroxide and hence enhance the toxicity of ZnO NPs (17). Therefore, as the temperature increases, more highly reactive oxygen species (ROS) are generated during the process, which are responsible for antibacterial activity (18, 19).
ZnO NPs may cause toxicity by different modes of action (ie, particle dissolution, oxidative stress, etc.) and these mechanisms are dependent on exposure conditions such as pH of the exposure medium and growth parameters like temperature. To verify this possibility, our data confirmed that various reaction conditions could be responsible for the disparity of ZnO NPs toxicological impact. Therefore, systematic characterization of these exposure conditions is essential for proper interpretation of toxicity data to differentiate between various modes of action and to predict the relative importance of particle-induced toxicity or dissolved Zn+2 effects in a given cultural setting.