1. Background
Senescence occurs after the development and maturation of cells (1), which means matured cells stop dividing, while they are metabolically active, mainly due to lack of DNA replication (1, 2) and unusual changes in the gene expression (3). There are evidence indicating the key role of intracellular production of reactive oxygen species (ROS) in the induction of cellular senescence (4, 5). Acrylamide is an industrial chemical compound utilized in various fields such as soil conditioning, textile, paper, and cosmetic industries (6). Recently, different levels of acrylamide are reported in high carbohydrate foods, which has increased attention to investigating its toxicity and pathogenic pathways. On the other hand, several studies showed the ability of acrylamide to induce neurotoxicity, reproductive toxicity, genotoxicity, immunotoxicity, and carcinogenicity in either cell lines or animal models. In addition, several experiments mentioned oxidative as one of the most important mechanisms in acrylamide toxicity (7, 8).
An imbalance between oxidant and antioxidant agents resulting from the release of free radicals and ROS can lead to oxidative stress and its related damages (9). Antioxidants can decrease oxidative damage and prevent mutagenesis, carcinogenesis, and senescence, mainly due to their radical‐scavenging activity (10). Antioxidants are compounds that can prevent or postpone autoxidation by blocking the pathways of free radical formation or by preventing free radicals damages through several mechanisms (11). Natural antioxidant compounds, such as flavonoids, phenolic acids, carotenoids, polysaccharides, and tocopherols, play the role of reductants by scavenging free radicals (12). Therefore, the physiological role of these compounds is to keep cellular components safe from the damage of chemical reactions resulted from free radicals (11). Carvacrol is a liquid phenolic monoterpenoid found in essential oils of various plants (13). The lipophilic properties of carvacrol are well proved. Moreover, carvacrol has a wide spectrum of biological properties, including anticarcinogenic, antioxidant, antibacterial, antifungal, and antiviral activities (14).
Astonishingly, antioxidant activities of carvacrol are higher than many of its counterparts (15). In addition, the antioxidant and anticancer properties of carvacrol on different human cancer cell lines have also been reported (16). Studies conducted using animal models also reported protective effects of carvacrol on acute pancreatitis (17) or brain, liver, and kidney damages caused by oxidative stress through its antioxidative mechanisms (18). Based on what was mentioned before, the current study aimed to, firstly, investigate the possible ability of acrylamide in inducing oxidative stress and premature cellular senescence, and, secondly, to examine the anti-senescence and antioxidant properties of carvacrol in attenuating cellular senescence in the embryonic fibroblast cell line, NIH 3T3.
2. Methods
2.1. Chemicals
Carvacrol (sigma, product number: 282197, purity ≥ 98%) and acrylamide (sigma, product number: A8887, purity ≥ 98%) were purchased from Sigma Chemical Co. (St. Louis, MO, USA). All the reagents used were analytical grade, HPLC grade, or pharmaceutical grade.
2.2. Cell Culturing and Treatment
Embryonic fibroblast cells can be found in all tissues of the mice, which plays a role in organ development. Hence, these cells are important models for in vitro experiments, especially aging and cell cycle models. The NIH 3T3 cells are mice embryonic fibroblast cell lines established from NIH Swiss mouse embryo cultures. NIH 3T3 was obtained from the Pasture Institute of Tehran, Iran. The cells were incubated at 37°C under a humidified atmosphere containing 5% CO2. After the third passage, the cells were seeded in 96-well culture plates (for MTT test) and 6-well culture plates (for β-galactosidase and oxidative stress assays) at a density of 2 × 104 and 2 × 105 cells per well, respectively. The experimental samples were sorted as groups described below:
- Control group: NIH 3T3 cells cultured in DMEM-HG medium and compound vehicle.
- Positive control group: NIH 3T3 cells exposed to H2O2 (400 μM).
- Acrylamide group: NIH 3T3 cells treated with acrylamide 5 mM (19).
- Carvacrol + acrylamide group: Various concentrations of carvacrol (50, 100, and 150 µM, carvacrol concentrations were chosen based on pre-tests and similar studies) (10, 20, 21) in combination with Acrylamide. Different concentrations of carvacrol were used in the MTT assay to determine the most effective concentration of carvacrol. Afterward, only the most effective concentration of carvacrol was used for the rest of the assays.
- Positive control + carvacrol group: The effective concentration of carvacrol in combination with H2O2 (note: carvacrol was dissolved in PBS with 2% DMSO).
2.3. Cell Viability Assay (MTT)
MTT assay was used to evaluate the viability of the treated cells. As mentioned earlier, the cells were pretreated with various concentrations of carvacrol (50, 100, and 150 µM) for 24 hours. Cell treatment with acrylamide and H2O2 was performed for 2 hours. After the incubation, the cells were rinsed twice using phosphate buffer (PBS), and then MTT solution (final concentration of 0.2 - 0.5 mg/mL) was added, followed by re-incubation at 37°C under for 4 hours. In the next step, 150 µL of DMSO solution was added, and eventually, an ELISA reader was used to record the absorbance at 570 nm. The viability of the acrylamide-treated group and the carvacrol-treated group are reported in comparison with the control group, which has been considered 100% (22).
2.4. β-galactosidase Assays
2.4.1. Senescence-Associated β-galactosidase (SA-β-gal) Staining Assay
As indicated by several studies, the β-galactosidase enzyme is only functional at a pH of 6 in senescent cells. In this study, the cells were seeded in 6-well plates and were cultured and sorted as groups mentioned before. Afterward, they were rinsed using PBS twice, followed by the addition of the fixative solution (20% formaldehyde and 2% glutaraldehyde) and incubation for 5 minutes at room temperature. In the next step, the fixative solution was removed, and the cells were rinsed with PBS twice. Then, they were stained with β-galactosidase stain and incubated at 37°C overnight (12 to 16 hours). The cells were examined under an optical microscope at 200x magnification. The observation of bluish-green color verified the presence of senescent cells (23).
2.4.2. Quantitative Identification of SA-β-gal
A mouse-specific β-galactosidase ELISA kit was used to assess β-galactosidase activity in NIH 3T3 cells (MyBioSource, cat number: MBS703814). 100 μL of standards and samples were added to each well, and then they were incubated for 2 hours at 37°C. After removing the liquid from each well, 100 μL of biotin-antibody was added, followed by incubation for 1 hour at 37°C. The wells were aspirated and rinsed, and then 100 μL of HRP-avidin (1X) was added to each well, followed by another incubation for 1 hour at 37°C. Next, aspiration and rinsing processes were performed again, and 90 μL of TMB substrate was added to each well, and they were incubated at 37°C for 30 minutes in a dark place. Eventually, 50 μL of the stop solution was added to each well, and the optical density was read at 450 nm.
2.5. Measurement of Oxidative Stress Parameters
2.5.1. Lipid Peroxidation Assay
To measure the oxidative stress damages to lipid membranes, the concentration of lipid peroxidation ending product, malondialdehyde (MDA), was evaluated using thiobarbituric acid (TBA). In this method, the cells were homogenized and mixed with 800 μL trichloroacetic acid, followed by centrifugation at 1500 xg for 30 minutes. Afterward, 150 μL TBA (1% w/v) was added to the supernatant, and the mixture was kept in a boiling water bath for 15 minutes. Subsequently, 400 μL n-buthanol was added to the mixture, and eventually, the absorbance was read at a wavelength of 532 nm using an ELISA reader (24).
2.5.2. Glutathione Content Measurement
Glutathione is a natural cellular antioxidant. Any decline in the level of the reduced form of this antioxidant (GSH) in cells can be considered a potential indicator of oxidative stress damage. In this study, to measure cellular glutathione concentration, the cells were homogenized using a glass mechanical homogenizer and then were mixed with Tris-EDTA buffer, followed by the addition of DTNB reagent and subsequent incubation for 15 minutes in a dark place at room temperature. Afterward, centrifugation at 3000 xg was performed, and, eventually, the absorbance of the supernatant was read at a wavelength of 412 nm using a spectrophotometer (25).
2.6. Statistical Analysis
The experimental data are described as mean ± standard deviation (SD). Data analysis was administered by SPSS version 13 using the one-way analysis of variance test (ANOVA) and the post hoc Tukey’s test. In addition, all graphs are drawn using Prism version 6. All assays were performed in triplet, and their mean has been used for statistical analysis. Statistical significance was considered when P-value < 0.05.
3. Results
3.1. Results of Cell Viability Assay
As demonstrated in Figure 1, acrylamide could significantly decline cell viability compared to the control group (P < 0.001). Furthermore, carvacrol increased the viability of the acrylamide-treated cells at the concentrations of 50 and 100 µM (P < 0.001). As the data indicate, H2O2 reduced the viability of the cells as much as acrylamide. Also, carvacrol (50 and 100 µM) could increase the cell viability of the H2O2-treated cells somehow, but it was not significant. Moreover, 100 µM was selected as the most effective concentration of carvacrol.
3.2. Results of β-galactosidase Assays
3.2.1. Results of SA-β-gal Staining Assay
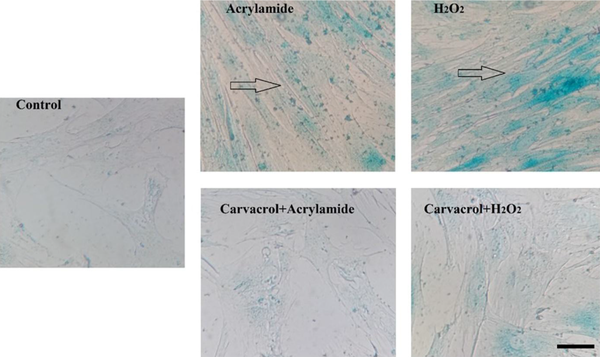
The effect of carvacrol and acrylamide on β-galactosidase activity is presented in Figure 2. The cells treated with acrylamide showed a significant rise in the level of β-galactosidase activity (verified by the appearance of the bluish-green color) as compared to the control group. On the other hand, the cells treated with carvacrol and acrylamide showed a significantly lower level of β-galactosidase activity.
3.2.2. Results of Quantitative Identification of SA-β-gal
The quantitative identification of SA-β-gal activity in different experimental groups is shown in Figure 3. As compared to the control group, the acrylamide and H2O2 groups both exhibited a significant increase in the level of β-galactosidase activity (P < 0.001). Furthermore, the carvacrol + acrylamide group, in comparison with the acrylamide group and the carvacrol + H2O2 group in comparison with H2O2 group, both demonstrated a significant decrease in the activity of SA-β-gal (P < 0.001 and P < 0.05, respectively).
The effect of carvacrol on the β-galactosidase activity of the NIH 3T3 cells exposed to acrylamide (ACR) and H2O2. Cells were treated with Carvacrol with 100 µM. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001); $ significantly different from H2O2 group (P < 0.05)].
3.3. Results of Measuring the Oxidative Stress Parameters
3.3.1. Results of Lipid Peroxidation Assay
Following the acrylamide and H2O2 treatment, an incremental pattern in the concentration of MDA was observed in studied cells. Also, carvacrol could protect the cells against damaging the effects of acrylamide and H2O2. As shown in Figure 4, exposure with acrylamide and H2O2, separately, resulted in escalated levels of lipid peroxidation (MDA concentration) in comparison to the control group (P < 0.001). Further on, the carvacrol + acrylamide group, in comparison to the acrylamide group and the carvacrol + H2O2 group compared to H2O2 group, both showed a significant decline in the level of lipid peroxidation (P < 0.001 and P < 0.01, respectively).
The effect of carvacrol on lipid peroxidation in the NIH 3T3 cells after acrylamide (ACR) and H2O2 exposure. Cells were treated with Carvacrol with 100 µM. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001); $$ significantly different from H2O2 group (P < 0.01)].
3.3.2. Results of Glutathione Content Measurement
As displayed in Figure 5, the amount of the reduced form of cellular glutathione in the acrylamide and H2O2 groups was significantly declined in comparison to the control group (P < 0.05 and P < 0.001, respectively). Instead, the carvacrol + acrylamide group compared to the acrylamide group and the carvacrol + H2O2 group in comparison with the H2O2 group showed a higher level of cellular glutathione (P < 0.05 and P < 0.01, respectively) (Figure 5).
The effect of carvacrol on glutathione concentration in the NIH 3T3 cells after acrylamide (ACR) and H2O2 exposure. Cells were treated with carvacrol with 100 µM. Data represent mean ± SD [* significantly different from the control group (P < 0.05); *** significantly different from control group (P < 0.001); # significantly different from ACR group (P < 0.01); $$ significantly different from H2O2 group (P < 0.01)].
4. Discussion
Oxidative stress-induced cellular senescence is an everlasting state of cell cycle arrest, which happens in proliferating cells as a response to stressful conditions. It has been proven that besides the permanent cell division arrest, senescent cells also exhibit noticeable changes such as resistance to apoptosis and a modified gene expression pattern (1). This study demonstrated that acrylamide behaves as an exogenous inducer of cellular senescence, as confirmed by several senescence biomarkers such as SA-β-gal concentration and different oxidative stress parameters (e.g., lipid peroxidation and cellular glutathione). In addition, it was showed that carvacrol has protective effects on this type of senescence in the mouse embryonic fibroblast cell line, NIH 3T3.
Many studies have investigated cellular senescence in several cell lines. Furthermore, beta-galactosidase activity is undetectable in immortal cells such as cancer cells, except for when these cells are manipulated for senescence evaluation by genetic or chemical procedures. Nevertheless, it can be argued that the administration of normal fibroblast cells is appropriate, especially when evaluating senescence-associated beta-galactosidase activity (26). Embryonic fibroblasts are appropriate for the evaluation of cell senescence, because not only they are available in all animal organs but also are involved in the development of various organs (23). Aging is a progressive process that accompanies by a gradual accumulation of various damages, reduced functional efficiency, and homeostasis of cells and tissues over time (27, 28). The key role of cellular senescence in aging and age-related disorders is well-established (29).
The SA-β-gal assay can also be used to investigate the onset of cellular senescence after exposure to different types of stress (30). In both in vitro and in vivo studies, the SA-β-gal assay has been widely used as a trustworthy biomarker in the detection of senescent cells (31). In the present study, β-galactosidase activity was measured using quantitative and qualitative methods. Similar to the recent studies (23), our finding demonstrated that H2O2 leads to increased β-galactosidase activity. Furthermore, a significant increase was observed in β-galactosidase activity in cells treated with acrylamide. These results are similar to those of previous studies, which showed that acrylamide also increased the b-galactosidase activity and induced senescence and aging in human umbilical vein endothelial cells (HUVECs) (32). Whereas the cell groups treated with carvacrol in combination with acrylamide or H2O2 appeared to have a lessened β-galactosidase activity compared to cells only treated with acrylamide or H2O2.
In cells exposed to oxidant agents, glutathione concentration was considerably reduced along with an increase in lipid peroxidation (33). It has also been suggested that oxygen-free radicals play a key role in brain senescence due to the elevated levels of MDA found in the brain of aged mice (34). In this study, acrylamide could increase the level of lipid peroxidation, which is attributed to the oxidative stress-inducing properties of this compound (35, 36). Furthermore, carvacrol treatment effectively reduced the level of lipid peroxidation in the acrylamide- and H2O2-treated cell groups. Earlier studies point that carvacrol inhibits lipid peroxidation by direct scavenging and inhibition of ROS production (37-39).
It has been known that glutathione concentration is decreased in the aged cells (40). Senescent organisms such as mosquitoes, adult houseflies, fruit flies, mice, rats, and humans are only a few examples of organisms in which an age-related decrement in the GSH level has been detected (41).
A negative association has been found between the concentration of GSH in the cerebral spinal fluid (CSF) and age (42), which indicates the key role of GSH levels in the process of senescence. In this study, acrylamide- and H2O2-treated cell groups both showed a declined level of GSH, which is in agreement with other similar studies. For instance, a study on the immune function of mice reported that acrylamide could induce immunotoxicity via decreasing the GSH concentration (and increasing lipid peroxidation) in the spleen of mice (43). Furthermore, the role of oxidative stress in the acrylamide induced toxicity is proved in other studies (35-37). However, carvacrol could increase the level of GSH in cells treated with acrylamide or H2O2, which is in compliance with other experimental studies (37-39).
Consequently, in this study, reduced lipid peroxidation and increased glutathione concentration, induced by carvacrol, may have an important role in the protection against senescent in fibroblast cells. Because carvacrol is a natural product with a proven record of safe human administration, it can be regarded as a promising alternative for clinical therapy.
4.1. Conclusion
In summary, this study demonstrated that acrylamide could induce cell senescence in fibroblast cells. Oxidative stress played an important role in initiating cell senescence. Furthermore, the results indicated that carvacrol could effectively reduce cell senescence in the NIH 3T3 cell line via attenuation of oxidative stress. Because carvacrol is a natural compound with a confirmed safety in humans, the beneficial effects of carvacrol may be useful in reducing adverse effects of food contaminations, such as acrylamide toxicity, in humans.
![The effect of carvacrol (CaR) on cell viability after acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub> exposure. Cytotoxicity was assessed using an MTT assay. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001)]. The effect of carvacrol (CaR) on cell viability after acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub> exposure. Cytotoxicity was assessed using an MTT assay. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001)].](https://services.brieflands.com/cdn/serve/3157f/36d599555e84afc841fc345a1800b8324020fdd9/jjnpp-16-4-109399-i001-preview.png)
![The effect of carvacrol on the β-galactosidase activity of the NIH 3T3 cells exposed to acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub>. Cells were treated with Carvacrol with 100 µM. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001); $ significantly different from H<sub>2</sub>O<sub>2</sub> group (P < 0.05)]. The effect of carvacrol on the β-galactosidase activity of the NIH 3T3 cells exposed to acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub>. Cells were treated with Carvacrol with 100 µM. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001); $ significantly different from H<sub>2</sub>O<sub>2</sub> group (P < 0.05)].](https://services.brieflands.com/cdn/serve/3157f/c2873ccc396284eddf1905e2864da34878b407e8/jjnpp-16-4-109399-i002-preview.png)
![The effect of carvacrol on lipid peroxidation in the NIH 3T3 cells after acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub> exposure. Cells were treated with Carvacrol with 100 µM. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001); $$ significantly different from H<sub>2</sub>O<sub>2</sub> group (P < 0.01)]. The effect of carvacrol on lipid peroxidation in the NIH 3T3 cells after acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub> exposure. Cells were treated with Carvacrol with 100 µM. Data represent mean ± SD [*** significantly different from the control group (P < 0.001); ### significantly different from ACR group (P < 0.001); $$ significantly different from H<sub>2</sub>O<sub>2</sub> group (P < 0.01)].](https://services.brieflands.com/cdn/serve/3157f/1a45c1c74eafe13cb53c34ce1b6254dfd1e445e0/jjnpp-16-4-109399-i003-preview.png)
![The effect of carvacrol on glutathione concentration in the NIH 3T3 cells after acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub> exposure. Cells were treated with carvacrol with 100 µM. Data represent mean ± SD [* significantly different from the control group (P < 0.05); *** significantly different from control group (P < 0.001); # significantly different from ACR group (P < 0.01); $$ significantly different from H2O2 group (P < 0.01)]. The effect of carvacrol on glutathione concentration in the NIH 3T3 cells after acrylamide (ACR) and H<sub>2</sub>O<sub>2</sub> exposure. Cells were treated with carvacrol with 100 µM. Data represent mean ± SD [* significantly different from the control group (P < 0.05); *** significantly different from control group (P < 0.001); # significantly different from ACR group (P < 0.01); $$ significantly different from H2O2 group (P < 0.01)].](https://services.brieflands.com/cdn/serve/3157f/c23d430030a2fc460fbb00fc46023300550655d6/jjnpp-16-4-109399-i004-preview.png)