1. Background
In recent years, there has been a dangerous spike in new antimicrobial resistance mechanisms worldwide, greatly threatening the ability to treat diseases of infectious origin. According to the World Health Organization (WHO), there are few replacement products in the research and development phase, and the world is moving towards a post-antibiotic era in which common infections could be fatal again (1). Thus, an important challenge for the scientific community is finding new antimicrobial compounds that can be used in cases of resistance (2).
Essential oils (EOs) are natural substances produced by aromatic plants and consist of mixtures of volatile compounds (3). The potent antibacterial activity of EOs of various origins has been demonstrated for several years. Furthermore, it has been suggested that bacteria sensitive to oils are inactivated by EOs without developing resistance (4, 5). Essential oils have been shown to have low toxicity in mammals and are rapidly degraded in the environment (6, 7).
Laurelia sempervirens, popularly known as Chilean laurel, belongs to the Atherospermataceae family and is endemic to Chile. This plant grows in the southern part of the country between 35° and 42° south latitude. In Chile, this plant is used in traditional medicine as an anti-inflammatory agent to as an expectorant and treatment for venereal diseases (8), and leaves of L. sempervirens are used by Amerindians for treating headache and as a diuretic (9). Although the chemical composition of Chilean laurel EO has been studied, only a few studies have examined its antifungal activity mainly against filamentous fungi, such as Penicillium spp. and Fusarium oxysporum (10, 11). To our knowledge, there is a scarcity of information on the effect of Chilean laurel EO on the generation and/or degradation of bacterial biofilms and the activity of this oil against yeast species that are pathogenic for humans.
2. Objectives
To determine antibiofilm and antimicrobial effects of Chilean laurel essential oil on human pathogenic strains.
3. Methods
3.1. Plant Material and Extraction of Chilean laurel EO
Fresh plant of L. sempervirens was collected in Ñuble Region (Chile) (36°51'S, 71°40'W). The plant was analysed by Dra. Olga Contreras Perez of Biology Institute of Talca University. Then, the plant was washed with distilled water and submitted to distillation (3 h; using Clevenger-type apparatus). The EO was obtained (0.73% ± 0.2), dried over anhydrous sodium sulphate, filtered and stored at 4 °C.
3.2. Chemical Composition of Chilean laurel EO
Chemical analysis of L. sempervirens EO was performed by gas chromatography coupled to mass spectrometry detector (GC-MS), using a GC Trace 1300 Thermo Fisher Scientific S.p.A equipment made in Italy. Separation was achieved using a column Rtx-5MS w/integra-guard (Cross bond 5% diphenyl - 95% dimethyl polysiloxane), 30 m × 0.25 mm i.d. × 0.25 µm d.f. The injection conditions were as follows: temperature: 250 °C, septum purge: 3 mL min-1 and split valve: 33.3. The column temperature gradient was 40 °C - 280 °C with the ramp rate of 10 °C min-1. The conditions of MS were: temperature of the transfer line: 280 °C, electronic impact ionization energy: 70 eV, mass range: 40 - 500 atomic mass units, scan rate: 20 scan s-1 and ion source temperature: 260 °C. The identification of compounds was achieved by comparing mass spectra with the National Institute of Standards and Technology Library (NIST NBS54K). And alkane standard solution C8-C20 was used as an analytical standard for measurement of retention indices (RIs) those that were calculated according to the RI Van den Dool and Kratz equation (12).
3.3. Antifungal and Antibacterial Activity of Chilean laurel EO and Determination of Minimum Inhibitory Concentration (MIC)
3.3.1. Yeast and Bacterial Strains
Candida albicans ATCC 14053, C. parapsilosis ATCC 22019, C. tropicalis ATCC 750, C. utilis ATCC 9950, C. krusei ATCC 6258, C. guillermondii ATCC 7350, C. lusitaneae ATCC 34449, Geotrichum sp. ATCC 96884, Bacillus cereus ATCC 14579, Staphylococcus saprophyticus ATCC 15305, Staphylococcus epidermidis ATCC 14990 and, Staphylococcus aureus ATCC 25923), Acinetobacter baumannii ATCC 19606, Proteus vulgaris ATCC 8427, Enterobacter aerogenes ATCC 13048, Salmonella typhi ATCC 35664, Klebsiella oxytoca ATCC 700324, Shigella sonnei ATCC 25931, Klebsiella pneumoniae ATCC 700603, Escherichia coli ATCC 25922, Enterobacter cloacae ATCC 13047, Providencia alcalifaciens ATCC 51902, Proteus mirabilis ATCC 35659, Enterococcus faecalis ATCC 29212, Citrobacter freundii ATCC 8090, Shigella flexneri ATCC 12022 and Pseudomonas aeruginosa ATCC 27853.
3.3.2. Antimicrobial Assay by the Agar Diffusion Method
Evaluation of antimicrobial activity of L. sempervirens EO was carried by the agar diffusion method. Bacterial suspension (≈ 106 colony forming unit [CFU] per mL) was streaked over Müller-Hinton (MH) agar (Merck KGaA, Darmstadt, Germany) plates, and then discs (Whatman® Antibiotic Assay Discs, 6 mm) impregnated with 5 µL of EO were placed on the inoculated plates and subsequently incubated at 37 °C for 24 h. Similarly, yeast suspension (≈ 106 CFU mL-1) was streaked over potato dextrose agar (PDA) (Merck KGaA, Darmstadt, Germany), and discs impregnated with EO were placed on the inoculated plates and were incubated for 48 h at 27 °C. For antibacterial assay, amoxicillin/sulbactam 10/10 µg (Valtek, code: 400 - 117) and discs with sterile distilled water were used as positive and negative controls respectively, and for antifungal assay, fluconazole (30 µg) (Neo-Sensitabs, Rosco diagnostica, Denmark) was used as a positive control, whereas discs without sample were used as a negative control. All the analyses were carried out in triplicate, and after the corresponding incubation time, each plate was analysed for the presence or absence of inhibition zones.
3.3.3. Minimum Inhibitory Concentration
The MIC of Chilean laurel EO was determined using the standard microdilution method (CLSI M100-S25) (13) and only against microorganisms that were susceptible in screening antimicrobial assay. Minimum inhibitory concentration was determined in Mueller Hinton (MH) broth using EO concentrations ranging from 4 µg mL-1 to 512 µg mL-1. The cell concentration was standardized to ≈ 1 × 108 CFU mL-1 of bacteria and yeasts using the McFarland standard (OD of 0.1 at 625 nm). Mueller Hinton broth medium with microorganism was tested as a positive control and MH broth without EO and without microorganism was tested as a negative control. Finally, the plates were incubated for 24 h at 37 °C for bacteria and for 24 h at 27 °C for yeasts. The MIC was determined based on the visual turbidity of the tubes and was examined in triplicate for each bacterium.
3.4. Determination of Antibiofilm Activity of Chilean laurel EO
To determine the inhibitory effect of Chilean laurel EO on the formation of bacterial biofilms, two methods already reported in the literature were used with a few modifications (3, 14, 15). Only two susceptible biofilm-forming bacteria were analysed.
3.4.1. Inhibition of Biofilm Formation
S. aureus and E. coli were grown in MH broth at 37 °C overnight. The cultures were centrifuged and rinsed with phosphate-buffered saline (PBS, pH = 7.4), and the bacteria were resuspended in MH broth (≈ 1 × 106 CFU mL-1, determined by optical density, OD). Then, 140 µL of the bacterial suspension was placed in wells of polystyrene plates, and EO was added to obtain a final concentration equivalent to 1 MIC and 2 MIC. Sterile water was used as the negative control; sterile broth was used to confirm the sterility of the medium. After incubation for 24 h at 37 °C, the broths were removed, the wells were washed with saline solution (0.89% NaCl) and the biofilm was quantified by the crystal violet staining method (3). Briefly, methanol (200 µL) was added to the wells, and after 15 min it was removed and the wells dried at room temperature. Then, an equal amount of crystal violet dye (0.5%) was added for 15 min, and after this time the stain was removed and the wells were washed and dried. Finally, 200 µL of 95% ethanol was added and absorbance (λ = 450 nm) was read using an AnthosBiochrom 2010 microplate reader.
The percentage of inhibition was obtained using the following equation:
% inhibition = 100 - [(OD450 nm experimental sample with EO/OD450 nm control sample without EO) × 100]
3.4.2. Effect of EO on Disruption of Biofilms
For this assay, 100 µL of bacterial suspension (≈ 106 CFU mL-1) was contacted with 100 µL MH broth enriched with 1% glucose, and then the plate was incubated at 37 °C for 48 h. Afterwards, the wells were washed with sterile saline solution to remove non-adherent cells, and L. sempervirens EO at MIC value was added and incubated at room temperature for 3 h. The wells were washed with saline solution, and the biofilm was quantified by the crystal violet staining method, as described previously (see point 3.4.1).
3.5. Statistical Analysis
Analysis of variance test was used to determine significant differences in the assays. A P-value of less than 0.05 was considered statistically significant.
4. Results
4.1. Chemical Composition
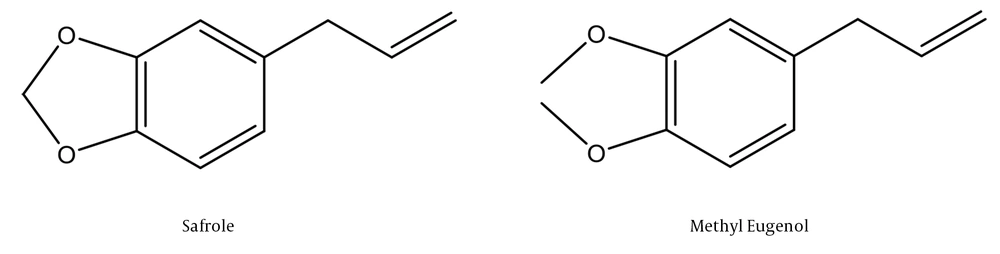
The chemical composition of Chilean laurel EO (see Appendix 1 in Supplementary File) was analysed by GC-MS, and 20 different compounds were identified that represented 98.71% of the total mixture. Safrole and Methyl-eugenol were the majority compounds with the relative abundance of 47.09% and 44.58%, respectively (Figure 1).
4.2. Antimicrobial Activity
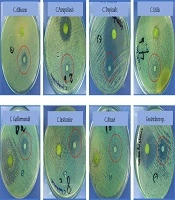
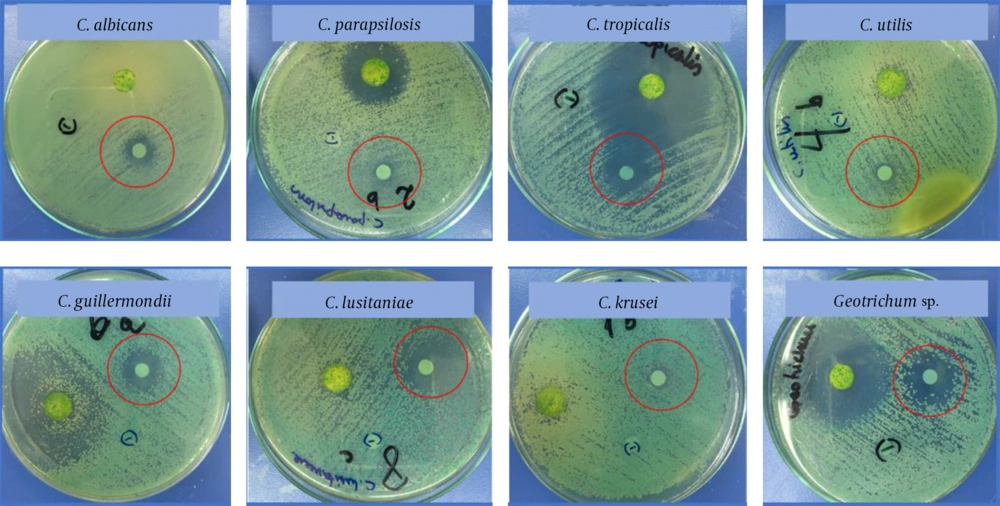
Antagonistic effect of Chilean laurel EO demonstrated extensive activity against the human pathogens studied, and antifungal activity is showed in Table 1 and Figure 2. Antibacterial activity is presented in support information (Appendix 2 in Supplementary File).
| Yeast | Inhibition Zone, mm ± RSDa |
|---|---|
| Candida albicans ATCC 14053 | 10.4 ± 0.7 |
| Candida parapsilosis ATCC 22019 | 9.8 ± 0.3 |
| Candida tropicalis ATCC 750 | 12.1 ± 0.6 |
| Candida utilis ATCC 9950 | 7.9 ± 0.7 |
| Candida guillermondii ATCC 7350 | 10.0 ± 0.5 |
| Candida lusitaniae ATCC 34449 | 11.2 ± 0.4 |
| Candida krusei ATCC 6258 | 9.3 ± 0.8 |
| Geotrichum sp. ATCC 96884 | 12.8 ± 0.3 |
Abbreviation: NA, not active.
aValues are expressed as means ± SD (n = 3) and include disc diameter (6 mm).
4.3. Minimum Inhibitory Concentration (MIC)
Minimum inhibitory concentration was determined for the microorganisms that were found most susceptible in the screening test, and the results are shown in Table 2.
| Microorganisms | MIC, µgmL-1 |
|---|---|
| Acinetobacter baumannii ATCC 19606 | 128 |
| Escherichia coli ATCC 25922 | 128 |
| Shigella flexneri ATCC 12022 | 64 |
| Bacillus cereus ATCC 14579 | 32 |
| Staphylococcus aureus ATCC 25923 | 64 |
| Staphylococcus epidermidis ATCC 14990 | 64 |
| Staphylococcus saprophyticus ATCC 15305 | 64 |
| Candida albicans ATCC 14053a | 64 |
aMIC fluconazole to C. albicans ≥ 64 µg mL-1.
4.4. Antibiofilm Activity
The antibiofilm activity was tested against S. aureus and E. coli, and the results are presented in Table 3.
| Bacteria | Inhibition of Biofilm Formation, % ± RSD | Effect on Disruption of Biofilms, % ± RSD | ||
|---|---|---|---|---|
| 1 MIC | 2 MIC | 1 MIC | 2 MIC | |
| S. aureus | 56 ± 5 | 67 ± 6 | 29 ± 2 | 32 ± 4 |
| E. coli | 35 ± 6 | 45 ± 3 | 22 ± 2 | 25 ± 2 |
5. Discussion
It has been known that essential oils can contain between 20 to 60 components in different concentrations and two or three that are found in high concentrations (20% - 70%) (7). In this study, the most abundant compounds of Chilean laurel EO were the aromatic compounds safrole and methyl eugenol. Tricyclic sesquiterpenoid, spathulenol and other components identified were present in amounts less than 1.1%. These findings are consistent with other reports published on the composition of L. sempervirens EO (8, 16).
Antibacterial activity of Chilean laurel EO against S. aureus, Bacillus subtillis, A. baumanni, P. aeruginosa, Salmonella spp, S. marcescens and E. coli has been demonstrated (9, 10). However, this study revealed its activity against important strains of human pathogens such as, P. vulgaris, E. aerogenes, S. typhi, K. oxytoca, S. sonnei, E. cloacae, P. alcalifaciens, S. flexneri, P. mirabilis, B. cereus, S. epidermidis, S. saprophyticus, C. albicans, C. parapsilosis, C. tropicalis, C. utilis, C. guillermondii and C. krusei. The antimicrobial activity of Chilean laurel EO could be largely due to the presence of safrole. It has been documented that this molecule inhibits the production of intracellular enzymes, such as amylases and proteases, causing wall deterioration and a high degree of cell lysis (11). Also, the presence of hydrophobic compounds, such as eugenol, α-terpineol and γ-terpinene, has been shown to cause bacterial cell membrane disruption (17). Some authors have proposed that EO components with the lowest proportion play a critical role in antimicrobial activity, possibly due to a synergistic effect between them (10). Due to the variability of compounds present, some possible mechanisms have been proposed that are associated with alteration of the outer membrane of Gram-negative bacteria with the release of lipopolysaccharides, interaction with membrane proteins (ATPases and others), destabilization of the proton motive force with ion leakage, coagulation of cell content and inhibition of enzyme synthesis (18).
Chilean laurel EO also had activity against Candida albicans, which is a human pathogen responsible for 90% of vulvovaginal infections caused by fungi. This information is relevant, since it has been reported that the management of fungal infections is currently complicated mainly due to the limited number of antifungal drugs, toxicity, high resistance to antifungal drugs and high costs (19). The mechanisms of antifungal activity of essential oils that have been described so far are quite similar to those described for their antibacterial activity, and they have to do with irreversible damage to the cell membrane (18).
Finally, Chilean laurel EO showed antagonistic activity against the formation of biofilms. Bacterial biofilms are communities of bacteria that adhere to surfaces through the extracellular production of polymeric substances, mainly polysaccharides and proteins (20). In this study, the EO activity decreased when the biofilms were already formed. It must be considered that biofilm formation implies two phases, an initial reversible binding phase followed by an irreversible binding phase (21). Therefore, the decrease in the activity of Chilean laurel EO for the disruption of biofilms may be due to the fact that the biofilms were already in an irreversible binding phase, and an even higher concentration should be considered to exert a considerable effect.
5.1. Conclusions
In sum, although moderate activity has been seen in terms of S. aureus biofilm disruption and low E. coli antibiofilm activity, we must consider, as mentioned above, that the test carried out in this work (crystal violet staining method) serves only as an indicator of bound biomass in a biofilm and does not reveal the metabolic status of cells. Therefore, we must continue with a metabolic analysis through which we can identify if the cells remain viable or not at the concentration the EO was used.
Taken together, it can be stated that Chilean laurel EO is a promising alternative candidate to the conventional antibacterial and antifungal agents, and it can be used as a possible antibiofilm agent. Thus, it can have various applications in the food and cosmetics industries among others. In view of these promising properties, in vivo studies and clinical trials are required to justify the use of this EO in humans.