1. Background
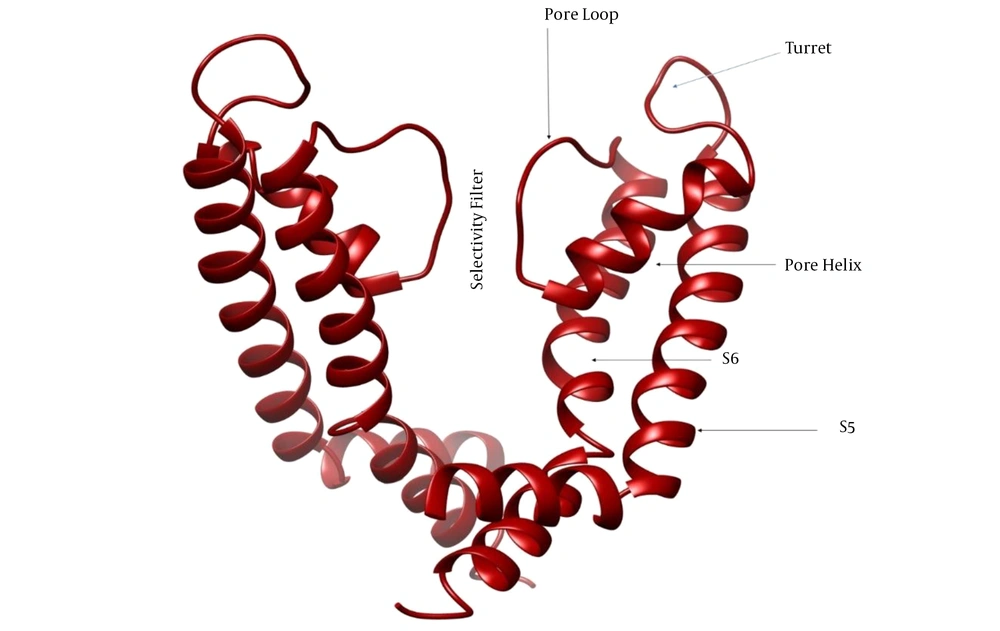
Voltage-gated potassium channels (Kv) are a significant and very varied group of potassium channels, characterized by 12 families (Kv1 to Kv12) (1). The Kv channels have an essential effect on generating and propagating the action potential by detecting fluctuations in transmembrane voltage and linking this detection to channel opening and closing that controls the efflux and influx of K+ ions into the cells (2). The Kv channels are tetrameric transmembrane proteins in which every subunit comprises a membrane-spanning and three extracellular parts. The membrane-spanning part consists of six helical segments (S1 to S6), and the extracellular parts involve the S1-S2 linker, the S3-S4 linker, and the pore region. The pore region includes the pore helix, pore loop, and turret (Figure 1). The S1-S4 linkers form the voltage-sensing domain, and S5-S6 form the pore-forming domain. Voltage-sensing domains sense diverse stimuli, and then K+ ions are transported through the pore (3).
Kv1.3 is preferentially expressed in T cells and is crucial for the activation of these immune cells, suggesting that Kv1.3 plays an essential role in autoimmune diseases, Multiple Sclerosis (MS), rheumatoid arthritis (4), type 1 diabetes (5), asthma (6), psoriasis (7), and neuroinflammatory human disorders such as stroke, epilepsy, Alzheimer’s disease, and Parkinson’s disease (8). Some studies have demonstrated that the blockers of Kv1.3 can selectively inhibit TEM (effector memory T) lymphocyte activation and improve the immunologic damage (4-6). Accordingly, Kv1.3 has been considered a target for treating T cell-mediated autoimmune diseases, and Kv1.3 blockers have great promise for use as immunosuppressant drugs in autoimmune diseases (8).
Scorpion toxins affecting potassium channels (KTxs) represent the potential to apply as valuable tools for developing novel medicines (9). Some peptides derived from scorpion venom are found to block Kv1.3. Up to now, different scorpion Kv1.3 blockers have been characterized, such as OSK1, a toxin from Orthochirus scrobiculosus scorpion venom (10), BmP02 (11), BmKTX, and BmKDfsin4 (12) from Mesobuthus martensii, Kaliotoxin from Androctonus mauritanicus (13), Charybdotoxin from Leiurus quinquestriatus (14), maurotoxin from Scorpio maurus (15), noxiustoxin from Centruroides noxius (16), pi1 toxin from Pandinus imperator (17), and Vm24 from Vaejovis smithi (18). Among them, ShK derivatives, ShK-186 and ShK-192, are mainly used to treat multiple sclerosis by targeting Kv1.3 channels (8).
Three interaction modes, including gating modifying, pore blocking, and turret blocking, have thoughtfully been described between KTxs and K channels (19). Gating modifier toxins bind to the voltage-sensing domain of K channels and block the channel by changing the closed state stability. Pore-blocking toxins bind at the K channel pore and occlude the ion conduction pathway. Some pore-blocker toxins like agitoxin-2 (20) directly occlude the channel pore with a residue (commonly Lys) occupying the pore of the channel, whereas the others bind to the region far from the central channel pore, i.e., the turret region (21). The exact mechanism by which these toxins block potassium channels remains unclear, while turret block, a general concept whereby the toxin functions as a lid on top of the pore entry, is suggested (19, 21).
Although many KTxs have been characterized from scorpion venom (22), there are still many unknown K+ channel blocker peptides whose identification can be helpful in the world of pharmacology and toxicology research. Moreover, most potassium channels are still in peptide blockers’ shortage, and the detection of new KTxs targeting these channels waits for an enormous challenge (23).
2. Objectives
Our study integrated homology modeling, docking analysis, and molecular dynamics simulation to understand the behavior of meuK2-2, a toxin derived from the scorpion Mesobuthus eupeus venom gland when exposed to the Kv1.3 channel. Docking analysis and molecular dynamics simulations determined the binding regions of meuk2-2 to Kv1.3 and identified residues involved in the interaction. The findings could suggest a new mechanism for meuK2-2 as a blocker of Kv1.3.
3. Methods
3.1. cDNA Library Construction and Screening
The cDNA library was constructed from the Iranian M. eupeus venom gland as described previously (24). Clones carrying a DNA insert of more than 700 base pairs, which theoretically can encode venom peptide precursors, were selected for cDNA sequencing. We deposited the nucleotide sequence applied in the current study in the GenBank database (http://www.ncbi.nlm.nih.gov) under accession number KU253401 (referred to as meuK2-2). By searching the GenBank database, BLASTP (https://blast.ncbi.nlm.nih.gov/Blast.cgi?PAGE = Proteins) uncovered that the homologs of meuK2-2 are mostly potent blockers of Kv1.3. The molecular weight and Isoelectric Point (IP) of meuK2-2 were calculated using the proteomics tool from the INNOVAGEN server (http://www.innovagen.com/proteomics-tools).
3.2. Generation and Refinement of Three-Dimensional Structures
The three-dimensional structures of the newly obtained KTx, meuK2-2, and KV1.3 were provided by program MODELLER (25) using homology-based protein structure models. A search against the Protein Data Bank Proteins (PDB) database was done to find the most similar structures to meuK2-2 and Kv1.3. The most similar peptide/protein with more than 75% identity was selected for homology modeling. Besides, BmP03 (PDB ID: 1WM8) and Kv1.2 (PDB ID: 5WIE) were selected as templates for modeling meuK2-2 and KV1.3, respectively. Following the modeling process with MODELLER, we selected the best model based on parameters, such as the smallest DOPE assessment score and the highest GA341 assessment score.
3.3. Molecular Docking of meuK2-2 with Kv1.3 Channel
Software HADDOCK (26) was used to investigate how the meuK2-2 interacts with the Kv1.3 channel. This program performs protein-protein docking using the molecular dynamics simulation technique entirely flexibly. Next, the residues playing a direct role in the toxin and potassium channel interaction were determined. These residues are defined as Ambiguous Interaction Restraints (AIR). Ambiguous interaction restraints are employed for molecular docking and defined through a list of residues that fall under two categories: active and passive. In general, active residues are of central importance for interaction. These active residues throughout the simulation must be part of the interface; otherwise, they will be penalized for scoring. Passive residues are those contributing to the interaction but are deemed of less importance.
After molecular docking, intermolecular interaction energy is used to rank the docking results. This energy is the sum of electrostatic and van der Waals energies. In HADDOCK software, amino acids with a direct effect on the interaction are called active amino acids, and the amino acids around the active amino acids are called passive amino acids. Amino acids with a water exposure level greater than 50% are considered active amino acids (26). Here, all ligand residues and amino acids in the pore of the Kv1.3 channel were defined as active amino acids. In the first cluster of docking results, the most regular conformation was selected to evaluate the complex stability by molecular dynamics simulation.
3.4. Molecular Dynamics Simulation
To evaluate the binding interaction between meuK2-2 and Kv1.3, we simulated the meuK2-2/Kv1.3 complex using a protein-water system in the NVT ensemble by Gromacs 2019.6 (27). Before the simulation, the CHARMM-GUI server (28) placed the meuK2-2/Kv1.3 complex inside the phospholipid membrane and prepared input files for simulation. Phosphatidylcholine (POPC) was used to make the phospholipid membrane. A total of 262 POPCs were applied for two layers of the phospholipid membrane. To balance the net charge of the system, we added Na+ and Cl− ions to the simulation box to 150 mM. The steepest descent algorithm was used for energy minimization. While running the simulation, the temperature and pressure were made constant using the Berendsen algorithm at 310 K and 1 atm, respectively. Finally, the ultimate complex simulation was run with the help of Gromacs 2019.6 with a time step of 2 fs for 100 ns (50 million steps). The PME algorithm was applied to calculate the electrostatic interactions. The SHAKE algorithm was developed to keep the length of the covalent bonds constant.
The software VMD was applied to visualize the trajectories and perform analyses. To evaluate the stability of the peptide in the binding region on the Kv1.3 channel, we measured the root-mean-square deviation (RMSD) (29). The RMSD changes related to alpha carbon atoms of ligand were calculated during the running time of simulation relative to the original structure. Finally, RMSD was used to evaluate the stability of meuK2-2 at the binding site on the Kv1.3 channel. The structure of the meuK2-2/Kv1.3 complex before and after simulation was visualized and compared with a molecular visualization program, UCSF ChimeraX (30).
4. Results
4.1. Characterization and Modeling of meuK2-2
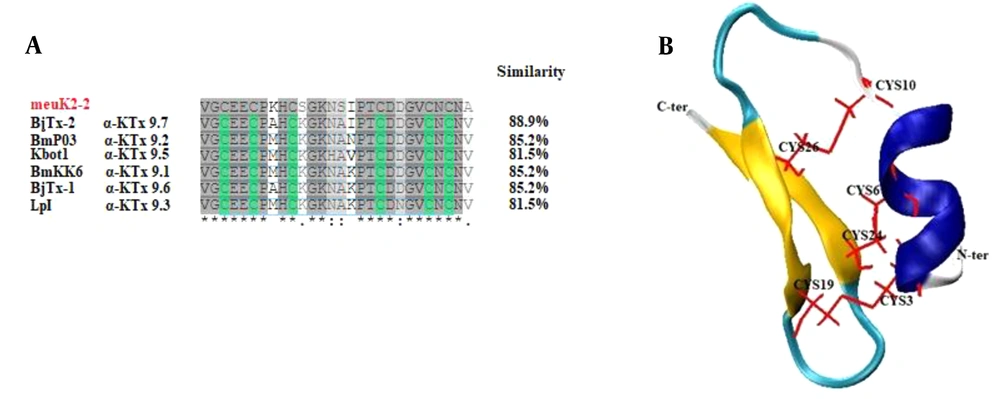
MeuK2-2 is a 28-amino acid peptide with molecular weight and IP of 2884 Dalton and 4.42, respectively. Based on IP, meuK2-2 is an acidic peptide. Sequence alignment demonstrated that meuK2-2 had extensive homology with α-KTx9 subfamily members (Figure 2A). The meuK2-2 final 3-D structure is shown in Figure 2B. As shown, meuK2-2 contains six Cys residues forming three disulfide bridges, and presents two structural domains: one α-helix and a double-stranded beta-sheet, indicating that they adopt a CSαβ (cysteine-stabilized α-helical and β-sheet) folding. The structure of CSαβ is created from a single α-helix connecting to a double or triple stranded β-sheet employing three or four disulfide bonds (31). Two random coils were also detected in the structure of meuK2-2.
A, multiple sequence alignment of meuK2-2 with other α-KTx9 members; B, 3-D view of the modeled meuK2-2. Disulfide bonds (red lines) connect the β–sheet (yellow) to the alpha-helix (dark blue), facilitating the folding of the peptide into a CSαβ structure. Random coils are shown in light blue.
4.2. meuK2-2/Kv1.3 Interaction
The modeled meuK2-2 was docked with Kv1.3 to generate their binding mode. The results of the docking are shown in Table 1. The binding energy between Kv1.3 and meuK2-2 of the first four clusters accompanied by the member number of each cluster is shown in this table. Cluster 1 has 16 members and the most significant number of the structures, but it shows the weakest interaction among the other three clusters in terms of energy. Due to the slight difference in the number of members related to clusters 1 and 2, cluster 2 was used to study the molecular dynamics and evaluate the stability of meuK2-2 in the binding region.
| Cluster | HADDOCK Score | Cluster Size |
|---|---|---|
| 1 | -20.193 | 16 |
| 2 | -65.92 | 12 |
| 3 | -43.130 | 10 |
| 4 | -63.505 | 10 |
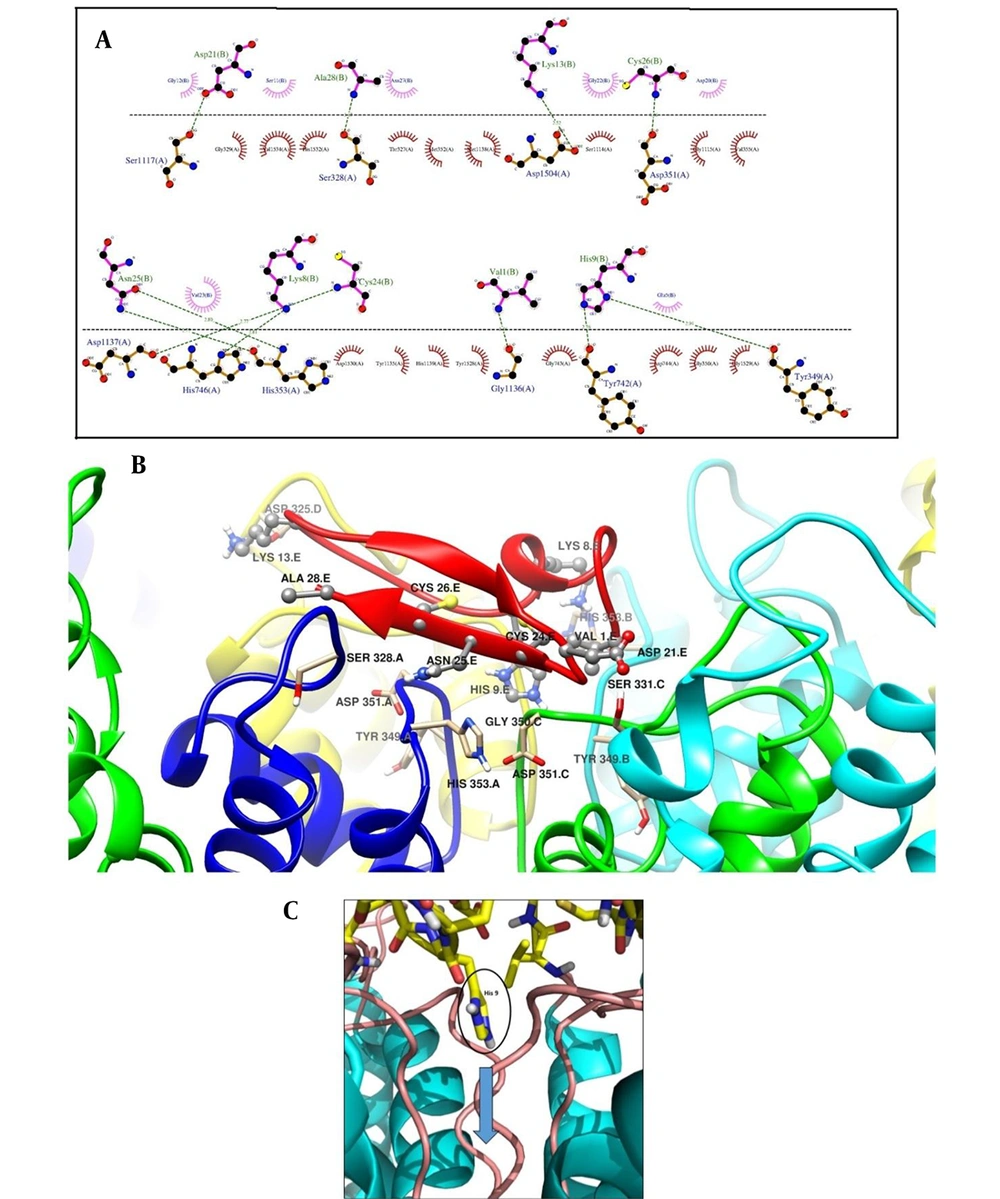
The interacting amino acids in the docking of meuK2-2 towards Kv1.3 are shown in Figure 3A. Nine residues of meuK2-2 (Val1, Lys8, His9, Lys13, Asp21, Cys24, Asn25, Cys26, and Ala28) were found to involve in interactions with 10 residues (Ser328, Tyr349, Asp351, His353, Tyr742, His749, Ser117, Gly1136, Asp1137, and Asp1504) on the surface of the vestibule of Kv1.3 channel through H-bonds.
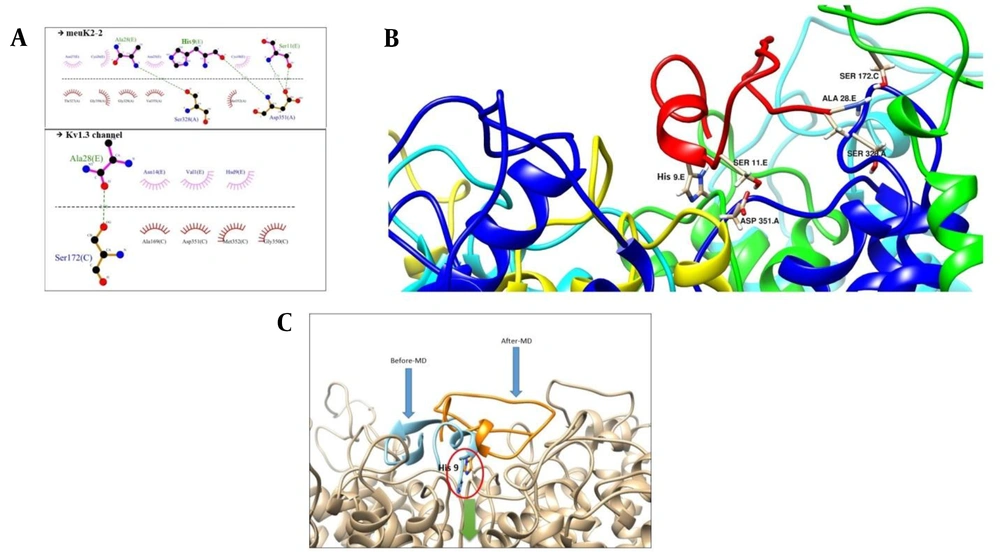
A, the interaction of meuK2-2 with the Kv1.3 channel with possible H-bonds resulting from docking. The dashed lines represent H-bond interactions. Critical residues of meuK2-2 are shown in pink color and crucial residues of Kv1.3 in orange color; B, 3-D presentation of meuK2-2 (red color) and Kv1.3 interactions. Residues of meuK2-2 and Kv1.3 contribute to the interaction; C, Kv1.3 channel blockade with the entry of residue His9. The pore path is indicated by a blue arrow.
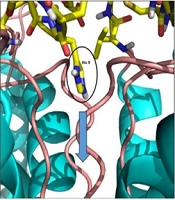
The 3-D view of meuk2-2, when docked with Kv1.3, is illustrated in Figure 3B. As shown in Figure 4C, after the interaction of the meuk2-2 with the amino acids on the surface of Kv1.3, His9 entered directly into the channel's pore. So, it could block the channel.
4.3. Structural Stability Analysis Upon meuK2-2 Binding Using MD Simulation
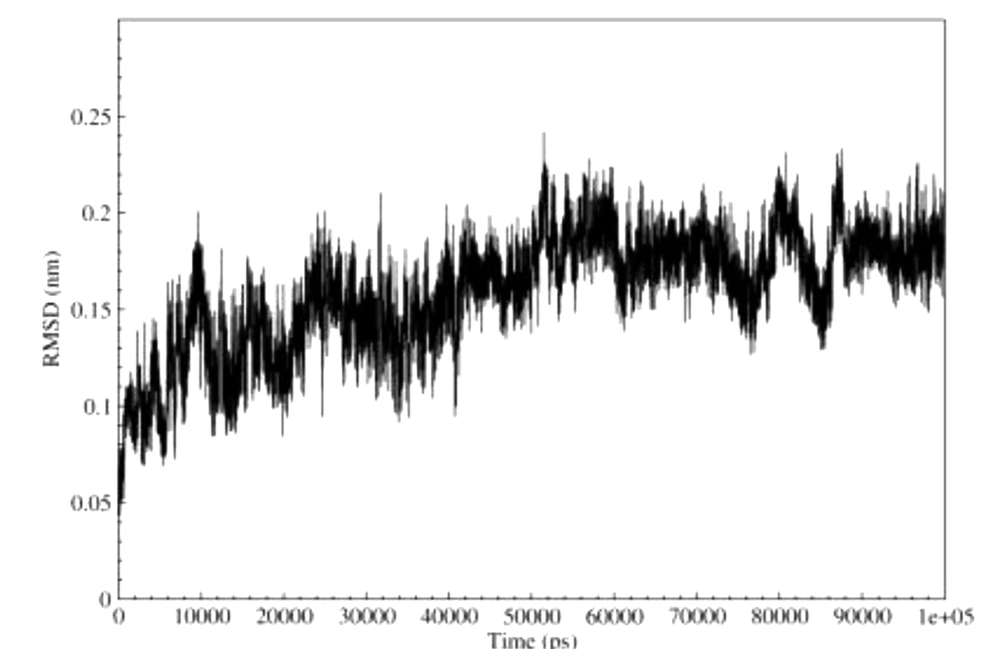
We calculated the RMSD changes of meuK2-2 to analyze meuK2-2 behavior during simulation. As a general rule, RMSD values reflect the local flexibility and mobility of the protein during MD simulation. So, a greater residue RMSD value indicates greater mobility and vice versa. We calculated the RMSD values for meuk2-2 using MD simulation. The results are shown in Figure 4. At the beginning of the simulation, the RMSD chart shows a slight upward trend, and this increase lasts up to 50,000 ps so that at this time, the RMSD value is equal to about 0.18 nm. From this time to the end of the simulation, the RMSD value remains constant at 0.18 nm, indicating the peptide structure's stability in the binding region after the time of 50,000 ps.
Figure 5 shows the conformational changes of meuK2-2 after simulation. According to this figure, the peptide binding conformation is shifted after simulation, and some interactions between the peptide and the channel are lost. The peptide, however, still retains its binding to the channel via three residues (His9, Ser11, and Ala28). After simulation, Ala28, in addition to Ser328, reacts with Ser172 of the channel, and His9 reacts with Asp351 instead of Tyr349. Moreover, Ser11, which had not participated in the reaction before simulation, enters the reaction with Asp351 after simulation. The interesting point is the location of His9, which penetrated the channel's pore in both cases before and after simulation (Figure 5C).
A, crucial residues in the interaction of meuK2-2 to Kv1.3 after simulation; B, 3-D view of participating residues in the meuK2-2/Kv1.3 interactions. meuK2-2 is in red; C, conformational changes of meuK2-2 in the binding mode on the vestibule of Kv1.3, before (blue) and after MD-simulation (orange). The green arrow indicates the channel pore. Note the position of His9, before and after MD simulation.
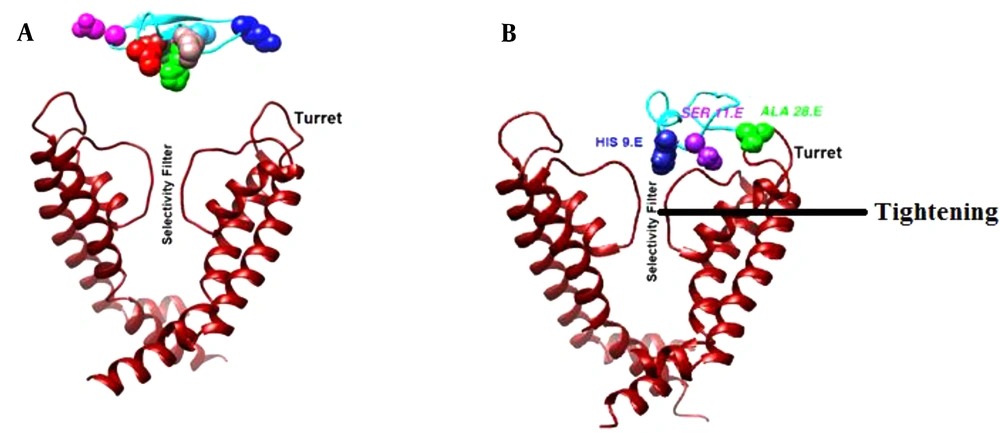
A better illustration of the hypothesized model of meuK2-2/Kv1.3 interaction and conformational changes of Kv1.3 induced by meuK2-2 is shown in Figure 6. As shown, Ala28 was bound to the turret region and Ser11 to pore the loop. The binding of Ala28 to the turret bent it inward close to the pore entry. His9 occluded the pore, and other parts of meuK2-2 acted as a lid above the pore entry. Moreover, the selectivity filter of Kv1.3 was tightened due to binding meuK2-2 to the channel.
Conformation changes of Kv1.3, induced by meuK2-2. A, before binding of meuK2-2; B, after binding of meuK2-2. The turret has bent inward close to the pore entry, the selectivity filter has become tighter, His9 has penetrated the pore, and other parts of meuK2-2 act as a lid above the pore entry.
5. Discussion
Some research has proved the effects of scorpion KTxs on some potassium channels consisting of SKCa, Kv1.x, BKCa, Kv11.1, and IKCa (24, 25). However, new KTx inhibitors are required for channel structure-function studies as potential drugs for the treatment of potassium channel-related disorders for which an inhibitor does not exist (25). Basic scorpion KTxs have been extensively employed in studies of toxin/potassium channel interaction assays. However, research on acidic KTxs has been progressed slowly (32). The findings of the current study show how an acidic peptide, meuK2-2, derived from the scorpion M. eupeus venom, can be an effector of the potassium channel Kv1.3. To produce a trustworthy 3-D model of the meuK2-2/ Kv1.3 complex, a computational protocol employing homology modeling, followed by molecular docking experiments, pose clustering, and molecular dynamic simulations (100 ns), was developed. The results showed that meuK2-2 has a modified CSαβ structure, and when it encounters the Kv1.3 channel, it interacts with the residues of the turret and pore loop of Kv1.3; eventually, His9 penetrates the pore of the channel and blocks the ion permeation pathway. Moreover, the position of meuK2-2 as a lid above the Kv1.3 helps block the channel.
The vast majority of scorpion venom peptides are basic. However, a few anionic peptides with no toxicity properties have previously been characterized from the venom gland of Mesobuthus martensii [BmKa1, BmKa2 (33), and HAP-1 (34)], Tityus costatus (four peptides) (35), Scorpiops jendeki (SJE098.1 and SJE098.2) (35), Buthus occitanus Israelis (BoiTx776 from) (36), Hottentotta judaicus (HjVP) (37), and Androctonus bicolor (38). The function of most of these peptides has not yet been characterized. It was previously thought that these peptides only play a part in balancing the pH of scorpion venom liquid (33). However, Shi et al. recently showed that HAP-1 has an inhibitory effect on the antimicrobial peptide activity of M. martensii Karsch venom (34). Now, acidic peptides are known as a new class of scorpion peptides characterized by (1) IP < 5.0; (2) being highly hydrophilic; and (3) having random coil regions and some α-helical domains (34). Due to IP (4.42), the presence of random coil, and α-helical domains in the 3-D structure, meuK2-2 is an acidic toxin that can make it unique as a toxic KTx. This peptide appears to be a bifunctional peptide with two roles in the venom gland of M. eupeus, blocking the K channel and balancing the pH of the venom liquid.
The homology modeling of meuK2-2 revealed a CSαβ folding. Peptides with the CSαβ structure are the most predominant components of scorpion venom-affecting potassium channels (39). A study using the molecular evolutionary analysis showed that most CSα/β toxin scaffolds underwent the periodic effect of positive selection, compared to non-CSα/β linear toxins (40). KTxs are structurally classified into seven different groups, i.e., α, β, γ, κ, δ, λ, and ε-KTx. Seven different scaffold motifs have been identified within the structure of scorpion toxins: CS-α/β, CS-α/α, ICK (inhibitor cystine knot), DDH (disulfide-directed β-hairpin), Kunitz-type structural fold with a double-stranded antiparallel β-sheet flanked by an α-helix, and a knotting type fold, stabilized by four disulfide bridges (41). The CSα/β motif occurs in α-, β-, and γ-KTxs subfamilies (39). MeuK2-2 was also classified as an α-KTx, confirmed by the presence of the CSα/β motif in its structure.
In one classification, scorpion toxins were classified into six main groups and 16 subgroups, based on both functional and structural characteristics (40). The CSα/β toxins were classified as the leading group with six subgroups, including CSαβ‘alpha, CSαβ’beta, CSαβ’chlorotoxin, CSαβ’lipo, CSαβ’scorpine, CSαβ’long-chain, and CSαβ'short-chain. Only the last three subgroups affect the potassium channels (40). Excluding CSαβ’scorpine’, which contains β-KTxs, meuK2-2 is classified in the CSαβ’short-chain’ group because of its short length (28 amino acids).
Mutagenesis and computational experiments have suggested various interactions between potassium channels and scorpion toxins (24). Our docking experiments determined that meuK2-2, like charybdotoxin (42), is a pore blocker toxin that binds to the pore loop through a residue (Ser11), and like ErgTx1 (43) and Cs1 (19), two turret blocker toxins, binds to the turret through a hydrophobic residue (Ala28). Unlike Charybdotoxin that blocks the channel by entering the side chain of Lys residue in the pore, meuK2-2 occludes the pore with the His residue. Both Lys and His are classified in amino acids with positively charged side chains.
The importance of hydrophobic side chains in the stable binding of potassium channel blockers has previously been identified. Leneaus et al. demonstrated that hydrophobic side chains stabilize the binding of quaternary ammonium (QA) compounds (potassium channel blockers) to the surface of the potassium channel cavity (44). Therefore, the hydrophobic interaction between meuK2-2 and Kv1.3 through Ala28 and Ser172 could play an essential role in stabilizing the meuK2-2/ Kv1.3 complex.
Some scorpion toxins like charybdotoxin did not induce conformational changes in binding to potassium channels (42), while noticeable conformational changes were induced by some other scorpion toxins like kaliotoxin (45) and ADWX-1 (46). Conformational changes predominantly occur in potassium channels' channel turret or filter region upon different scorpion toxin bindings (47). Computational simulations revealed that during the binding of ADWX-1 toxin to the Kv1.3 channel, the channel turret from one Kv1.3 chain makes close contact with ADWX-1 toxin, while the rest of the chains bend outward and move away from the ADWX-1 toxin (46). These findings are in line with what was found here for meuK2-2. When meuK2-2 associates with the Kv1.3 channel, it binds to the turret and bends it inward close to the pore entry (Figure 6).
In contrast to ADWX-1, the selectivity filter plays a vital role in binding the Kv1.3 channel to the scorpion toxin, maurotoxin. During the binding of maurotoxin to the Kv1.3 channel, conformational changes were induced in the channel filter region rather than the channel turret (48). MeuK2-2, in addition to the turret channel, binds to the pore loop simultaneously. The binding of meuK2-2 to the pore loop induces conformational changes, leading to the selectivity filter getting tighter (Figure 6).
In an overall comparison, meuK2-2 behaves somewhat like pore blocker toxins and somewhat like turret blocker toxins. Pore blocker toxins plug the K+ ion conduction pathway by entering a residue in the channel pore, whereas turret blockers locate as a lid at the pore entry and prevent K+ ions currents (21). MeuK2-2 has both features in the interaction with Kv1.3. Moreover, a tighter selectivity filter results from the meuK2-2/ Kv1.3 interactions. We propose it as a new mechanism of scorpion toxins (or possibly basic scorpion toxins) and potassium channels interactions. Further research can shed new light on meuK2-2/Kv1.3 interactions.
The Kv channel blockers are known to be new tools to inhibit the activation of T-cells, resulting in specific immunosuppression. Besides, Kv1.3 is the primary channel on human effector memory T lymphocytes; consequently, it composes an appealing pharmacological target for autoimmune diseases mediated by T cell immunomodulation (49). Functional characterization of meuK2-2 as a potent Kv1.3-targeted peptide will create new therapeutic agents for human autoimmune diseases.