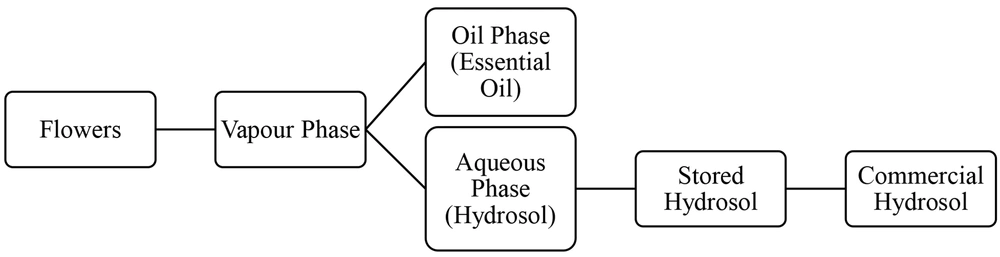
Antioxidants are substances that can prevent or reduce the formation of free radicals (
16). The accumulation of reactive oxygen species (ROS) has been identified as the cause of skin aging, resulting in dryness, loss of subcutaneous tissue, and wrinkle formation. Natural antioxidants are essential for skincare products and are widely used in the cosmetics industry (
30). Several assay types can be used to evaluate the antioxidant activity of plant hydrosol. Based on Degirmenci and Erkurt (
15), antioxidant activity is expressed as a percentage of scavenging (%) and 50% scavenging concentrations (IC
50 g/mL).
Table 5 shows the antioxidant potential and biochemical composition of plant hydrosol.
According to Francezon and Stevanovic (
14), antioxidant activity was assessed using two separate chemical pathways [i.e., 2,2-diphenyl-1-picrylhydrazyl hydrate (DPPH) and oxygen radical absorbance capacity (ORAC) assay] to gain a better understanding of the antioxidant potential based on hydrogen atom transfer. Not just that, natural phenolic compounds, such as phenolic acids and flavonoids, have been claimed to contribute to the biological activity of plant materials and play an essential role as antioxidant properties (
15), where they can be used to assess hydrosol's antioxidant potential (
31). Due to the capacity of hydrogen donation atoms to free radicals, phenolic and flavonoid molecules are significant antioxidant components that deactivate free radicals. They also have the perfect structural properties for scavenging free radicals (
32).
The chemical composition of hydrosol, in particular, a high level of phenolic and flavonoids, was found to be associated with antioxidant activity measured using the DPPH method (
22). The difference in the result obtained could be related to the distinct mechanisms involved or the presence of encapsulated bioactive compounds (
3). According to Wollinger et al. (
16), the DPPH assays were always chosen since they are a quick and simple way to look at total antioxidant scavenging activity. In addition, the experimental approach may be readily adapted to the analytic experiments. The analysis of antioxidant activity may need further research and require more than one method in order to determine the suitable method for producing the best result to avoid misunderstanding in the result of the study. According to Johari and Khong (
33), determining the phenolic content in samples is known as total phenolic content (TPC) activity. The redox characteristics of phenolic chemicals found in plants allow them to act as antioxidants. The Folin-Ciocalteu colorimetric test was used to obtain the TPC values (
15). Phenolic compounds have direct antioxidant properties due to the presence of hydroxy groups acting as hydrogen donors; thus, they might contribute to antioxidant activity. Shen et al. (
3) reported that total phenolic levels of hydrosols from several sections of arecanut and coconut ranged from 0.17 to 3.88 μg/mL of gallic acid equivalent. Other than that, flavonoids are secondary antioxidant metabolites where the strength is determined by the amount and position of free hydroxy (OH) groups (
32). The total flavonoid content (TFC) values were calculated using the aluminum chloride colorimetric method and represented in milligrams of quercetin equivalents per gram of extract (mg QE/g). Research by Degirmenci and Erkurt (
15) reported that the hydrosol of
Citrus aurantium flowers, the TFC, was found to be lower than essential oil.