1. Background
2. Objectives
3. Methods
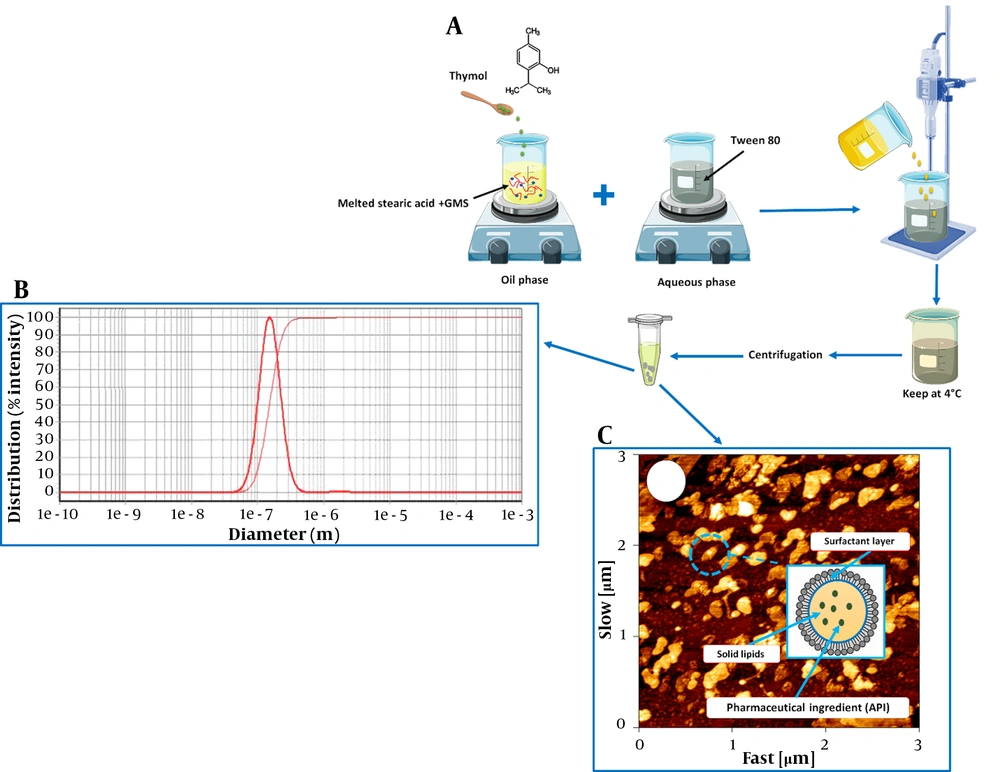
3.1. Preparation of Solid Lipid Nanoparticles
3.2. Determination of Encapsulation Efficacy
3.3. Particle Size and Morphology Study
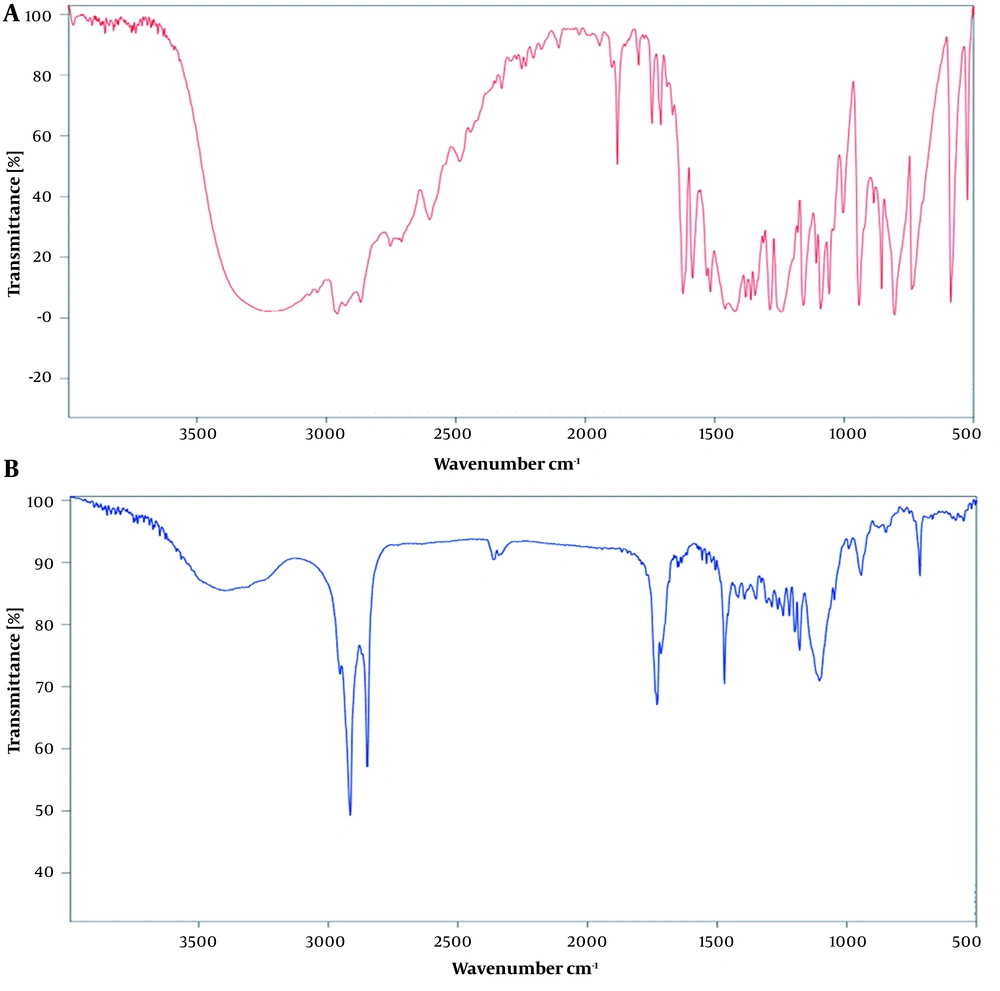
3.4. Fourier Transform Infrared Analyses
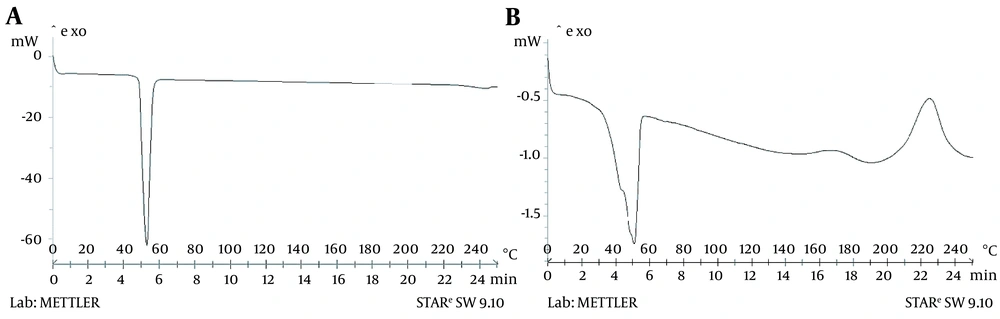
3.5. Differential Scanning Calorimetry
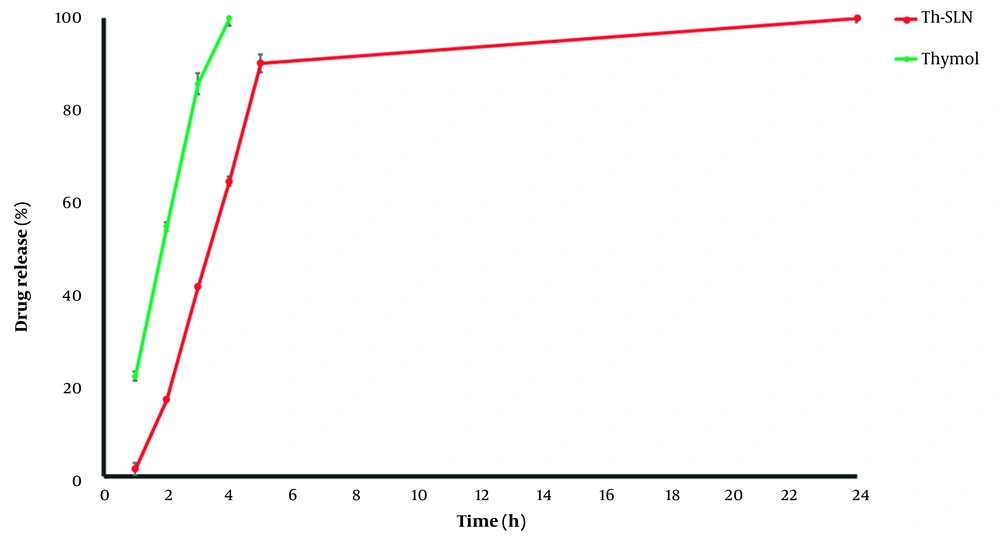
3.6. In vitro Drug Release
3.7. In-vitro Cytotoxicity Assays
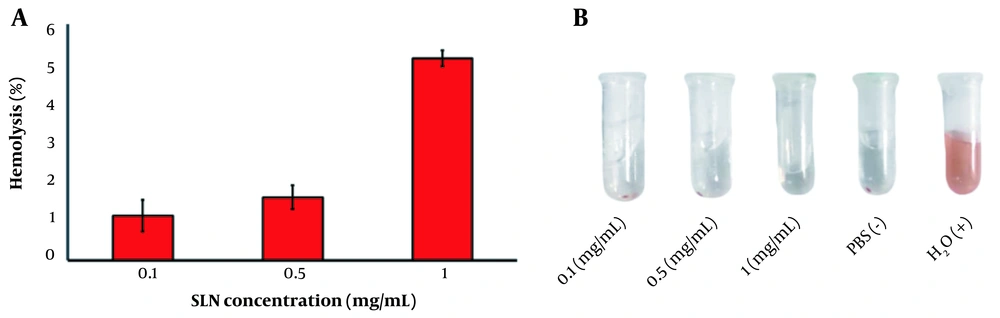
3.8. Hemolysis Assay
3.9. Statistical Analysis
4. Results and Discussion
| Formulation | EE% | Particle Size (nm) |
|---|---|---|
| Th-loaded SLNs | 63 | 145 |
Abbreviation: SLNs, solid lipid nanoparticles.
4.1. Fourier Transform Infrared Analyses
4.2. Differential Scanning Calorimetry Analysis
4.3. In vitro Release Study
| Parameters | Models | |||||||
|---|---|---|---|---|---|---|---|---|
| Zero-order | First Order | Higuchi | Korsmeyer–Peppas | Hixcon | Weibull | Linear Wagner | Logarithmic Wagner | |
| R2 | 0.9930 | 0.8682 | 0.9625 | 0.9716 | 0.9293 | 0.9948 | 0.9874 | 0.9729 |
| MPE% | 48.82 | 405.29 | 99.12 | 25.42 | 209.69 | 7.99 | 17.74 | 15.67 |
Abbreviations: R2, regression coefficient; MPE%, mean percentage error.
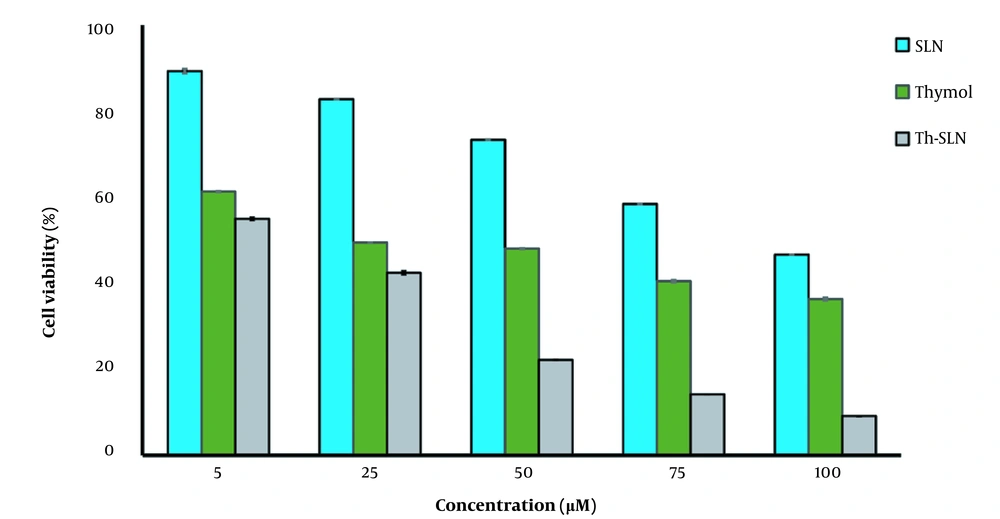
4.4. Cytotoxicity Evaluation
| Formulation | IC50 (μM) |
|---|---|
| Thymol | 39.22 ± 0.9 |
| SLNs | 94.87 ± 1.1 |
| Th-SLNs | 7.88 ± 0.7 |
Abbreviations: SLNs, solid lipid nanoparticles; Th-SLNs, thymol-loaded SLNs.