1. Background
The use of medicinal plants has significantly increased in recent years, and they are now widely regarded by researchers and the pharmaceutical industry as promising therapeutic options (1). Haloxylon ammodendron is a xerophytic perennial shrub with a high tolerance to drought. In many Asian desert regions, this plant predominates as the flora (2). Antibiotic resistance among bacteria such as Klebsiella pneumoniae, Escherichia coli, Salmonella typhimurium, Proteus vulgaris, Corynebacterium xerosis, and Candida albicans is one of the factors that make the treatment of infections challenging (3). It appears essential to discover and use effective natural compounds with lower costs and fewer complications against microorganisms, considering the increasing prevalence of infectious diseases and occurrences of antibiotic resistance. Plants’ secondary metabolites, known as polyphenols, have potent antioxidant properties. The molecular structure of flavonoids and other polyphenols makes them excellent for inhibiting free radicals (4).
There is no study on the antibacterial and antifungal effects of H. ammodendron, but studies have been conducted on other species in this family. Previous studies on several species of Haloxylon have led to the identification and isolation of alkaloid compounds. Other chemical compounds in this plant include tannins, saponins, and glycosides (5). Chemical evaluations showed that two alkaloids (haloxylin A and B) from Haloxylon salicornicum have antifungal and cholinesterase enzyme inhibitory effects (6). The alkaloids isolated from H. salicornicum were effective in treating gastric ulcer symptoms caused by indomethacin in animal models (7). Synephrine and N-methyltyramine, compounds isolated from the aqueous extract of H. salicornicum, revealed inhibitory effects on spontaneous uterine contractions in mice (8). Crude extracts of several species of Haloxylon were evaluated. The ethyl acetate extract of Haloxylon scoparium has an inhibitory effect on Staphylococcus aureus (9). Ullah et al. reported that the aqueous extract of H. salicornicum has anti-inflammatory, antipyretic, analgesic, and antioxidant activities without significant toxicity (10).
This study is the first to assess the antibacterial and antifungal properties of H. ammodendron ethanolic extract and provides an analysis of the plant’s antioxidant capacity. Additionally, the research offers insights into the phenolic content of the plant’s ethanolic extract, which is directly linked to its bioactivity. This study positions H. ammodendron as a promising candidate for natural antimicrobial and antioxidant agents, with implications for developing new strategies in combating microbial resistance and oxidative stress-related conditions.
2. Objectives
This study evaluated the antioxidant, antibacterial, and antifungal activities of H. ammodendron ethanolic extract, along with its phenolic content. The method offers significant advantages: Efficient extraction of bioactive compounds using ethanol, precise phenolic quantification, and reliable antimicrobial assessment through standardized minimum inhibitory concentration (MIC) determination. These strengths provide a comprehensive analysis of the plant’s therapeutic potential.
3. Methods
3.1. Preparation of Plant Extract
Haloxylon ammodendron was identified by an expert at the Medicinal Plants Research Center of Ahvaz Jundishapur University of Medical Sciences, using botanical tests based on the herbal pharmacopeia of Iran. A voucher specimen (A230070200ArP) was deposited in the School of Pharmacy, Ahvaz Jundishapur University of Medical Sciences. The plant material was dried at room temperature to reduce moisture content, ensuring that heat-sensitive compounds remained intact. The dried material was then ground into a fine powder to increase the surface area, facilitating better contact with the solvent during extraction and improving compound recovery efficiency. The powdered plant material was macerated in 70% ethanol, a solvent commonly used because it balances the extraction of polar (water-soluble) and non-polar (ethanol-soluble) compounds. The mixture was macerated for 24 hours, with occasional stirring. During the cold maceration process, filtration was performed every 24 hours to separate the liquid extract from the plant residue using filter paper. After each filtration, fresh 70% ethanol was added to the remaining plant material to continue the extraction process, and this step was repeated three times. The filtered liquid extract was then concentrated using a rotary evaporator to remove excess solvent, followed by drying in a freeze-dryer. This drying step, conducted at low pressure and temperature, minimized the degradation of heat-sensitive compounds while efficiently concentrating the extract. The dried plant extract was stored in a suitable container for further applications (11).
3.2. Total Phenolic Content
The Folin–Ciocalteu technique was used to calculate the total phenolic content. One gram of tannic acid (as standard) was dissolved in 100 milliliters of ethanol to create this standard. From this solution, dilutions of 75, 100, 150, and 200 μg/mL were created. One milliliter of each dilution and 5 mL of Folin-Ciocalteu reagent was mixed. Ten minutes later, 4 mL of a 75 mg/mL sodium bicarbonate solution was added, and all combinations were allowed to incubate at room temperature for two hours in the dark. The absorbance of each solution was determined at 765 nm. The absorbance data was used to draw a calibration curve. One milliliter of the plant ethanolic extract (1 mg/mL) was mixed with the same reagents to measure the absorbance of the mixture to determine the phenolic content of the extract. Each measurement was performed in triplicate. By utilizing the calibration curve, the total phenolic content of the extract was determined as mg of tannic acid equivalents per g of extract (12).
3.3. Antioxidant Activity
The antioxidant activity was evaluated using 1,1-diphenyl-2-picrylhydrazyl (DPPH). Several concentrations (25, 50, 100, 200, 500, and 1000 ppm) of the extract were mixed with DPPH, and after 30 minutes of storage in darkness at room temperature, the absorbance of the solutions was measured at 517 nm (13). A methanolic solution of butylated hydroxytoluene was used as the positive control. The percentage of antioxidant activity was calculated using the formula: % Antioxidant activity = [(Ac - As)/Ac] × 100, where Ac is the control reaction absorbance and As is the testing specimen absorbance.
3.4. Bacterial Strains
Standard strains were used to evaluate the antimicrobial effects of the extracts. The standard strains of bacteria included S. aureus (ATCC 29213), Streptococcus mutans (ATCC 35668), K. pneumoniae (ATCC 700721), E. coli (ATCC 25922), Acinetobacter baumannii (ATCC 19606), Pseudomonas aeruginosa (ATCC 27853), and C. albicans (ATCC 10231). These standard strains were collected from the laboratory of the Microbiology Department of Jundishapur University of Medical Sciences and maintained there. The bacteria were cultured twice in BHI medium (Merck, Germany) at 37°C for 18 hours (14). This step was essential to confirm the purity and consistency of the bacterial cultures, as any contamination or loss of viability could affect the reliability of the results.
3.5. Preparation of Diluted Extracts
To achieve a 50 mg/mL concentration of each extract, 500 mg of dried plant extract was diluted in 10 mL of 5% dimethyl sulfoxide (DMSO) and sterilized by filtering through a 0.45 µm membrane filter. The extract was prepared in various concentrations (50, 25, 12.5, 6.25, and 3.12 mg/mL) using the serial dilution method (15).
3.6. Agar Well Diffusion Method
The antimicrobial activity of H. ammodendron extract on gram-positive and gram-negative bacteria was assessed using the well diffusion method (16). Dilutions of 50, 25, 12.5, 6.25, and 3.12 mg/mL of the extract were added to each 6-mm-deep well punched into Müller Hinton agar plates, which had been seeded with 0.5 McFarland of the test bacterium. The total volume was 30 µL. The apparent inhibitory zone diameter that developed around each well after 24 hours of incubation at 37°C was measured in millimeters. Dimethyl sulfoxide was used as the negative control (with no inhibitory zone) (15).
3.7. Minimum Inhibitory Concentration Procedure
The MIC is defined as the lowest concentration of a substance that prevents observable growth of an organism following overnight incubation (17). Serial dilutions, including 1000, 500, 250, 125, and 62.5 µg/mL of the extracts, were prepared in trypticase broth medium and added to each 10-mL tube of microbial suspension, equivalent to 0.5 McFarland. The tubes were incubated for 18 hours at 37°C, and the first tube that showed no turbidity was considered the MIC. This process was repeated at least three times for each dilution, and the results were recorded as the MIC of the extract.
4. Results
4.1. Total Phenolic Content
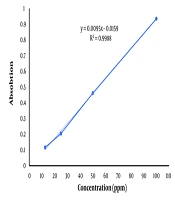
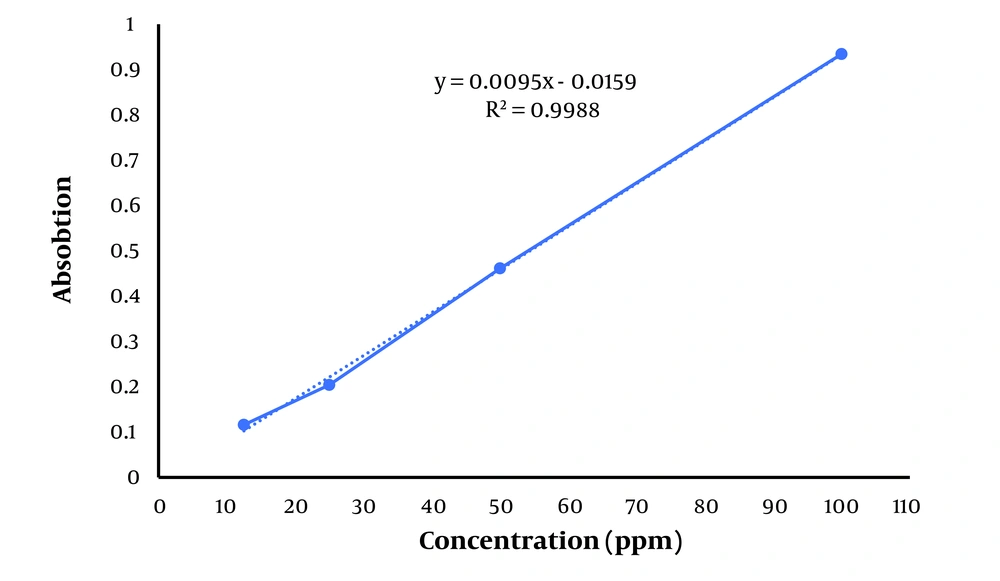
The calibration curve presented in Figure 1 shows the relationship between absorbance and concentration of the standard (tannic acid). The linear equation is (y = 0.0095x - 0.0159), with an (R2 = 0.9988), indicating excellent linearity across the concentration range tested (10 - 100 ppm). The high R2 value (close to 1) suggests a strong correlation between the concentration of the phenolic compounds and the absorbance, ensuring the reliability of the curve for quantification. The total phenolic content was 58.27 ± 0.80 mg per gram of the H. ammodendron ethanolic extract. The standard curve of tannic acid is presented in Figure 1.
4.2. Antioxidant Activity
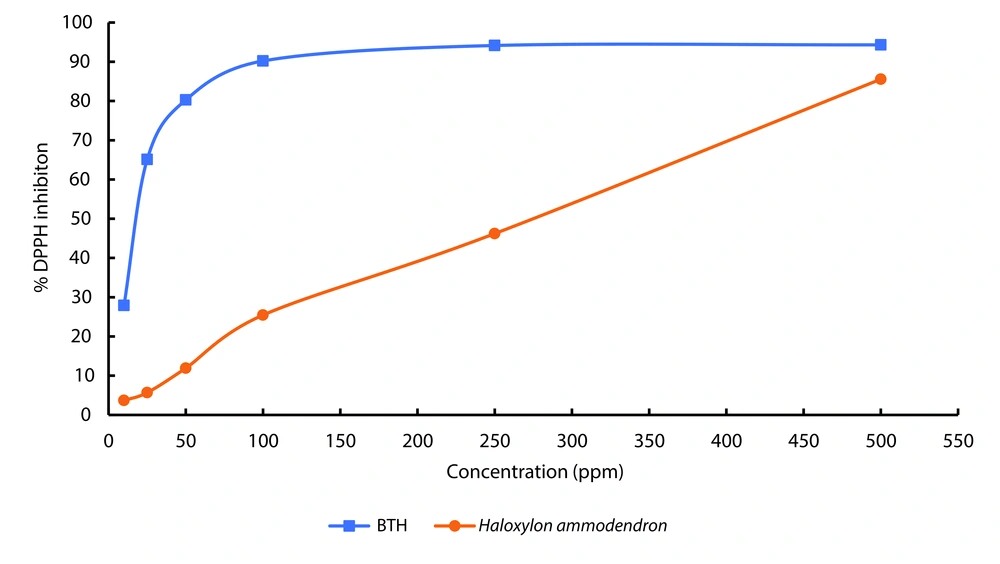
The DPPH test was used to measure the antioxidant properties of the ethanolic extract. The results of the antioxidant activity test of the H. ammodendron extract, repeated three times, demonstrate that the ethanolic extract of this plant possesses antioxidant properties. The percentage of DPPH radical inhibition at different concentrations of H. ammodendron ethanol extract compared to BHT is presented in Figure 2. The graph displaying antioxidant activity data does not include error bars due to the exceptionally small standard deviations observed. These minimal variations indicate a high level of consistency in the data, making the error bars too small to be visible on the graph. This suggests that the antioxidant activity measurements were highly reproducible and precise across the replicates. The antioxidant activity of the extract is directly related to its concentration, indicating that as the concentration of the extract increases, its antioxidant activity also increases.
The IC50 value measured for BHT as a standard was 19.16 ppm, while this value was 265.9 ppm for the ethanolic extract of H. ammodendron. This result indicates that the ethanolic extract of H. ammodendron exhibits lower antioxidant activity compared to BHT, which is expected.
4.3. Antimicrobial Activity
The results of the antimicrobial effect of H. ammodendron extract, determined by the well diffusion method, are presented in Table 1. The maximum inhibitory zone diameters at a concentration of 50 mg/mL were 14 mm for S. aureus and S. mutans, followed by C. albicans and P. aeruginosa with a non-growth halo of 12 mm. Additionally, the extract exhibited the lowest inhibition, resulting in the least effective impact on Acinetobacter, K. pneumoniae, and E. coli bacteria.
| Organisms | The Concentration of the Haloxylon ammodendron Extract (mg/mL) | Control Negative (DMSO) | ||||
|---|---|---|---|---|---|---|
| 50 | 25 | 12.5 | 6.25 | 3.12 | ||
| Streptococcus mutans (ATCC 35668) | 14 | 10 | 8 | 6 | - a | - |
| Staphylococcus aureus (ATCC 29213) | 14 | 10 | 8 | 6 | - | - |
| Klebsiella pneumoniae (ATCC 700721) | 10 | 7 | - | - | - | - |
| Acinetobacter baumannii (ATCC 19606) | 8 | 6 | - | - | - | - |
| Pseudomonas aeruginosa (ATCC 27853) | 12 | 10 | - | - | - | - |
| Escherichia coli (ATCC 25922) | 10 | - | - | - | - | - |
| Candida albicans(ATCC 10231) | 12 | 10 | 8 | 6 | - | - |
Abbreviation: DMSO, dimethyl sulfoxide.
a The inhibitory zone was not seen.
4.4. Minimum Inhibitory Concentration
In this study, the MIC of H. ammodendron extract against S. aureus, S. mutans, K. pneumoniae, E. coli, A. baumannii, P. aeruginosa, and C. albicans was investigated. The results of the MIC of H. ammodendron extract are presented in Table 2. The results showed that the MIC of this extract was 250 µg/mL for S. aureus, S. mutans, and C. albicans; 500 µg/mL for P. aeruginosa, A. baumannii, and K. pneumoniae; and 1000 µg/mL for E. coli.
| Organisms | Growth in the Concentration of the Haloxylon ammodendron Extract (µg/mL) | Control Negative (DMSO) | ||||
|---|---|---|---|---|---|---|
| 1000 | 500 | 250 | 125 | 62.5 | ||
| Streptococcus mutans (ATCC 35668) | - | - | - | + | + | - |
| Staphylococcus aureus (ATCC 29213) | - | - | - | + | + | - |
| Klebsiella pneumoniae (ATCC 700721) | - | - | + | + | + | - |
| Acinetobacter baumannii (ATCC 19606) | - | - | + | + | + | - |
| Pseudomonas aeruginosa (ATCC 27853) | - | - | + | + | + | - |
| Escherichia coli (ATCC 25922) | - | + | + | + | + | - |
| Candida albicans (ATCC 10231) | - | - | - | + | + | - |
Abbreviation: DMSO, dimethyl sulfoxide.
a -: Bacterial growth.
b +: No-growth of bacteria.
5. Discussion
This study aimed to investigate the antioxidant and antimicrobial activity of the ethanolic extract of H. ammodendron. Natural antioxidants can scavenge free radicals before they initiate oxidative chain reactions in the cell membrane or lipid-containing organelles of the cell. The collection of reactive radical species has an indicative effect on the stability of vulnerable cellular compounds and ensures the health of body cells and tissues. Today, medicinal plants are considered one of the important sources of antioxidant compounds and have a privileged position in clinical research (18).
Research indicates that phenolic compounds in plants are responsible for their antioxidant properties. Practically all plant parts contain phenolic compounds that are essential to several physiological functions, such as fruit ripening, seed germination, and cell growth (19, 20). Plants are essential raw materials in the medical and food sectors, providing critical nutrients while being extensively employed in the treatment of numerous diseases. The high concentration of phenolic compounds in certain plants has garnered considerable scientific attention due to their bioactive properties. The potential application of these phenolic compounds, particularly their potent antioxidant capacity, is of significant interest for therapeutic and nutritional advancements (21).
Phenolic compounds are a large group of naturally occurring plant metabolites characterized by one or more hydroxyl groups attached to an aromatic ring. They are known for their antioxidant properties, which help protect plants and humans from oxidative stress by neutralizing free radicals. Phenolic compounds include flavonoids, phenolic acids, tannins, and lignans, commonly found in fruits, vegetables, tea, coffee, and wine. Phenolic compounds are associated with health benefits, such as reducing the risk of chronic diseases like cardiovascular diseases, cancer, and neurodegenerative conditions (22).
Phenolic compounds are powerful antioxidants due to their unique chemical structure, which allows them to interact with reactive oxygen species (ROS) and other free radicals in multiple ways. First, the hydroxyl groups attached to their aromatic rings can donate hydrogen atoms or electrons to neutralize free radicals, forming a more stable phenoxyl radical that prevents further propagation of oxidative chain reactions. This free radical scavenging property is essential in reducing damage to lipids, proteins, and DNA, which are often targets of oxidative stress.
In addition to neutralizing free radicals, phenolic compounds are effective metal ion chelators. They bind transition metals like iron (Fe2+) and copper (Cu2+), which catalyze the production of ROS through Fenton reactions. By chelating these metals, phenolic compounds reduce the formation of hydroxyl radicals, one of the most damaging ROS, and protect cells from metal-induced oxidative damage.
Another important antioxidant mechanism of phenolic compounds is their ability to modulate antioxidant enzyme systems in the body. Polyphenols like resveratrol, quercetin, and curcumin have been shown to upregulate the activity of key antioxidant enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx). These enzymes play crucial roles in detoxifying harmful ROS and maintaining redox balance within cells, providing an additional layer of protection against oxidative stress.
Finally, phenolic compounds can inhibit lipid peroxidation, a destructive process in which free radicals attack polyunsaturated fatty acids in cell membranes. Lipid peroxidation can lead to cell membrane damage, loss of cellular integrity, and ultimately cell death. Phenolics like epigallocatechin gallate (EGCG) in green tea and flavonoids in various fruits and vegetables effectively inhibit lipid peroxidation, contributing to the preservation of cellular health. These diverse antioxidant mechanisms of phenolic compounds make them effective in protecting against a range of oxidative stress-related diseases, including cardiovascular disease, neurodegenerative disorders, diabetes, and certain cancers (21-23).
The stable DPPH radical scavenging model is widely used to evaluate the ability of various compounds (such as plant extracts) to scavenge free radicals. This method is based on the decolorization of the DPPH solution, which is performed by the antioxidants in the extracts through the inhibition of free radicals (18). In the present study, the DPPH technique was used to evaluate the antioxidant properties of H. ammodendron extract. The findings of this study demonstrate a clear correlation between the concentration of H. ammodendron ethanolic extract and its antioxidant activity, with higher concentrations yielding increased activity. This dose-dependent behavior is characteristic of plant-derived phenolic compounds, which typically exert their antioxidant effects by scavenging free radicals and donating hydrogen atoms or electrons.
The measured IC50 value for the H. ammodendron extract is significantly higher than that of the synthetic antioxidant BHT, indicating a lower overall antioxidant efficacy. Nevertheless, the observed antioxidant activity of the H. ammodendron extract remains noteworthy, particularly considering the increasing demand for natural antioxidants due to safety concerns associated with synthetic compounds. Although less potent than BHT, the extract may offer a safer alternative in food preservation or therapeutic applications when used at higher concentrations.
Studies have shown that plants with high levels of phenol and flavonoid compounds have relatively high antioxidant properties (24, 25). In the study by Amzad Hossain et al., the antioxidant activity of the essential oil and extract of the native plant Merremia borneensis was evaluated. The researchers evaluated the total phenolic content (with gallic acid as standard) and flavonoids (with quercetin as standard) of different extracts of M. borneensis, including those obtained with different solvents such as ethanol, methanol, hexane, chloroform, ethyl acetate, and butanol. The results showed that essential oils and extracts had significant antioxidant activity, primarily attributed to their phenolic compounds. Among them, flavonoids and phenolic acids were found to be the main factors. These compounds demonstrated strong free radical scavenging abilities and were effective in reducing oxidative stress. The antioxidant properties of the extracts were assessed using standard assays, including DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging and Ferric Reducing Antioxidant Power (FRAP) assays (24).
In another study, the antioxidant capacity and total phenolic content of different medicinal plants in Iran (Descurainia sophia, Plantago major, Trachyspermum copticum, Coriandrum sativum, Trigonella foenum-graecum) were investigated. The researchers measured the total phenolic content with gallic acid as standard and evaluated the antioxidant activity using different assays, such as DPPH radical measurement and total antioxidant capacity assay. In this study, P. major and T. foenum-graecum had the highest and lowest amounts of total and antioxidant properties, respectively (25).
In the present study, investigating the antimicrobial properties of H. ammodendron extract showed that S. aureus and S. mutans bacteria exhibit less resistance with increasing extract concentration, resulting in a larger diameter of the non-growth zone. However, in E. coli bacteria, the zone of non-growth was observed only at the highest concentration (50 mg/mL). Polyphenols exhibit broad-spectrum antimicrobial activity through multiple mechanisms. These compounds inhibit bacterial virulence factors, such as enzymes and toxins, by interfering with quorum-sensing pathways, thereby reducing bacterial pathogenicity. They interact directly with the bacterial cytoplasmic membrane, disrupting lipid bilayers, increasing permeability, and causing leakage of cellular contents, as observed with quercetin and catechins. Furthermore, polyphenols suppress biofilm formation, a key bacterial defense mechanism, by disrupting extracellular polymeric substances and interfering with adhesion molecules, effectively reducing biofilm development in pathogens like S. aureus and P. aeruginosa. Additionally, these polyphenols exhibit synergistic effects with antibiotics, restoring antibiotic efficacy by inhibiting efflux pumps and enhancing bacterial susceptibility to drugs such as ciprofloxacin and ampicillin. These properties position polyphenols as promising agents for combating multidrug-resistant bacteria and enhancing the efficacy of existing antimicrobial treatments (26-29).
The resistance of gram-negative bacteria to plant-based antimicrobials is primarily due to their unique cell wall structure. Unlike gram-positive bacteria, which possess a thick peptidoglycan layer, gram-negative bacteria have a more complex and effective permeability barrier. This barrier consists of an outer membrane containing lipopolysaccharides (LPS), which forms a protective layer that limits the penetration of antimicrobial compounds, including plant extracts. The outer membrane also features porins, proteins that regulate the passage of molecules, further restricting access to harmful substances. Gram-negative bacteria have a periplasmic space, which is not present in gram-positive bacteria. The periplasmic space also contains enzymes that can break down foreign molecules that enter (26). Due to this robust defense, gram-negative bacteria tend to be more resistant to plant-derived antimicrobials and often exhibit minimal or no inhibitory response. In contrast, gram-positive bacteria lack this outer membrane and have a much thicker peptidoglycan layer, which is easier for plant extracts to penetrate. The more open and accessible nature of the gram-positive cell wall allows these antimicrobials to disrupt essential cellular functions, leading to a higher susceptibility (26, 30).
Numerous studies have shown that gram-negative bacteria, such as E. coli and Salmonella, are more resilient to plant-based antimicrobial agents compared to gram-positive species like S. aureus and Bacillus cereus. This difference in susceptibility is a well-documented factor in the development of antimicrobial treatments, making it crucial to tailor strategies based on the specific bacterial structure (31, 32). In a study, Biswas et al. demonstrated the antimicrobial properties of guava leaf extract (Psidium guajava) in four different solvents (hexane, methanol, ethanol, and water) against two gram-negative bacteria (E. coli and Salmonella enteritidis) and two gram-positive bacteria (S. aureus and B. cereus). Their results indicated that the methanolic and ethanolic extracts effectively inhibited gram-positive bacteria, while gram-negative bacteria resisted all tested extracts (33). Mahfuzul Hoque et al. reported no antibacterial activity of the ethanolic extract of guava against E. coli and S. enteritidis (34). Similarly, Nascimento et al. found that guava extract could inhibit Staphylococcus and Bacillus species but had no impact on Escherichia and Salmonella (35).
In a study, the aqueous, ethanol, and chloroform leaf extract of six medicinal plants, Ocimum sanctum (Tulsi), Citrus limon (Lemon), Nerium oleander (Nerium), Azadirachta indica (Neem), Hibiscus rosasinensis (Hibiscus), Eucalyptus globulus (Eucalyptus), was evaluated against E. coli, Pseudomonas spp., and Klebsiella spp. The lemon aqueous extracts showed a good inhibitory effect against E. coli. The best antibacterial activity against Klebsiella was observed with Eucalyptus ethanolic extracts, and Pseudomonas was observed with Tulsi ethanolic extracts. Pseudomonas was resistant to all other plant extracts (36).
Liu et al. conducted a comparative analysis of the antibacterial properties of four types of tea, including green, oolong, black, and Fuzhuan tea against gram-positive bacteria (Enterococcus faecalis and S. aureus) and gram-negative bacteria (E. coli and S. typhimurium). The study demonstrated that all tea extracts exhibited antibacterial activity, with gram-positive bacteria showing greater susceptibility to the tea extracts than gram-negative bacteria. Subsequent investigations revealed that catechins, polyphenolic compounds present in tea, disrupted bacterial cell membranes by increasing membrane permeability, leading to alterations in relative electrical conductivity and the leakage of intracellular components. The study found that catechins more significantly compromised the membrane integrity of S. aureus compared to E. coli, which may explain the stronger antibacterial effect observed against gram-positive bacteria (30). The reduced efficacy of catechins against gram-negative bacteria is likely due to their inability to penetrate the outer lipopolysaccharide layer, whereas they can directly interact with the exposed peptidoglycan layer in gram-positive bacteria. These structural differences between gram-positive and gram-negative bacteria are considered the primary reason for the differential antibacterial activity of catechins (37).
The antimicrobial properties of H. ammodendron have been evaluated in different contexts, yielding complementary insights. For instance, the Saudi H. ammodendron demonstrated strong antimicrobial activity through silver nanoparticle biosynthesis, which effectively inhibited both gram-positive and gram-negative bacteria, as well as exhibiting anticancer properties. In contrast, our study found that the ethanolic extract of H. ammodendron exhibited selective antimicrobial activity, with significant inhibition of gram-positive bacteria (S. aureus and S. mutans) but limited effects on gram-negative strains (38).
Additionally, the metabolomic analysis of H. ammodendron and Heracleum persicum under drought stress highlighted the increased production of secondary metabolites, such as phenolics, which are known to contribute to antioxidant and antimicrobial activities. Our results align with this observation, as the ethanolic extract showed significant antioxidant activity (IC50: 265.9 ppm) and a notable phenolic content (58.27 ± 0.80 mg/g extract) (39).
The comprehensive review of the Haloxylon genus emphasized its pharmacological properties, including anti-inflammatory, antioxidant, and antimicrobial effects, attributed to compounds such as alkaloids, tannins, and saponins. While our study corroborates these findings, particularly regarding antioxidant and antibacterial activities, the broader pharmacological potential of H. ammodendron requires further exploration, particularly for antifungal and therapeutic applications (40).
This comparison underscores the unique and overlapping bioactivities of H. ammodendron, positioning our findings within the broader context of existing research on the genus.
In a study, Wahab et al. evaluated the pharmacological and toxicological effects of Haloxylon recurvum methanolic extract and butanol fraction. These extracts showed significant activity against E. coli, Shigella flexneri, and S. aureus, but had weak to moderate antifungal activity against Aspergillus flavus, Trichophyton longifusus, and C. albicans. Methanolic extract and other fractions did not show significant activity against Microsporum canis, Candida glabrata, and Fusarium solani (41). Lamchouri et al. investigated the antibacterial effect of aqueous, methanolic, ethyl acetate, petroleum ether, and chloroform extract of H. scoparium against S. aureus, E. coli, S. xylanases, P. aeruginosa, K. pneumonia, and Branhamella catarrhalis by disk diffusion method. Results showed that only the ethyl acetate extract has an inhibitory effect on S. aureus (9).
The results of this in vitro study indicate that the ethanolic extract of H. ammodendron exhibits antimicrobial activity against S. aureus and S. mutans and inhibits the growth of C. albicans. With further investigations in animal studies, this extract could potentially be used as a suitable plant for developing new herbal medicines. Based on the findings, H. ammodendron extract holds the potential for development into natural antioxidants or antimicrobial agents, possibly for use in treating infections caused by gram-positive bacteria or as an adjunct in managing oxidative stress-related disorders. Future clinical studies and formulation development are needed to evaluate its therapeutic potential.
5.1. Conclusions
Plants exhibit a broad spectrum of antimicrobial and antioxidant activity. The results of this study indicate that the ethanolic extract of H. ammodendron possesses antimicrobial and free radical inhibitory properties. Additionally, a significant amount of phenolic compounds was observed in the extract of H. ammodendron. Considering the limited number of studies performed on H. ammodendron, the results of this investigation can be regarded as a preliminary report on this plant’s beneficial role in terms of antimicrobial and antioxidant activity. The findings of this research can serve as a basis for further studies aimed at isolating the active ingredients of this plant. Additional investigations can be conducted to develop appropriate pharmaceutical formulations and explore its effects in clinical trials.