1. Background
Anabolic androgenic steroids (AAS) are synthetic derivatives of testosterone, designed to promote muscle growth (anabolic effects) and enhance male physical characteristics (androgenic effects) (1). Their use has increased significantly, particularly among athletes and bodybuilders seeking to enhance performance and improve physique (2). One of the most well-known anabolic steroids is dianabol, also referred to as methandrostenolone or methandienone. Introduced in the late 1950s, dianabol was initially developed for medical purposes (3). However, it quickly gained popularity among athletes and bodybuilders due to its powerful anabolic properties (4). Dianabol functions by binding to androgen receptors in muscle cells, triggering protein synthesis and enhancing nitrogen retention, both essential for muscle growth (5). Users often experience significant increases in muscle mass and strength in a relatively short period.
While dianabol may temporarily delay muscle cell death, it can cause oxidative damage and toxicity in other organs, such as the kidneys (6). Additionally, dianabol is linked to various adverse effects, including cardiovascular issues, hormonal disruptions, and psychological impacts such as increased aggression and mood swings (7). Moreover, apoptosis, a programmed cell death mechanism, can be exacerbated by oxidative stress induced by dianabol, leading to severe liver damage.
Selenium and vitamin E are well-documented antioxidants that play crucial roles in mitigating oxidative stress and enhancing cellular defense mechanisms (8). Previous research has demonstrated that dietary supplementation with these antioxidants can significantly reduce lipid peroxidation and improve enzymatic activity in the liver, thereby offering a protective effect against steroid-induced damage (9, 10).
Clove (Syzygium aromaticum) is a spice obtained from the dried flower buds of an evergreen tree native to Indonesia. Bioactive components of clove extracts include compounds such as eugenol, eugenyl acetate, β-caryophyllene, gallic acid, and flavonoids. Clove, rich in these bioactive compounds, has been shown to positively impact liver function by reducing elevated serum levels of liver enzymes like aspartate aminotransferase (AST) and alanine aminotransferase (ALT), which are indicative of liver damage. These compounds also contribute to clove’s potent antioxidant capacity, as evidenced by its ability to scavenge various reactive oxygen species (ROS) and enhance antioxidant enzyme activity, thereby protecting against oxidative stress and lipid peroxidation (11, 12). Renowned for its distinct flavor and aroma, clove is widely used in cooking, particularly in curries, baked goods, and beverages. Beyond its culinary uses, clove has a long history of use in traditional medicine, primarily due to its high antioxidant content (13). Research suggests that clove may offer various health benefits, including improved blood sugar regulation and digestive support (14).
Moreover, AAS, such as dianabol, can decrease levels of superoxide dismutase (SOD), the body’s primary enzymatic defense against free radicals (15). Consequently, dianabol misuse may increase the release of apoptotic factors, leading to decreased cell survival and increased cell death (16). It may function as an oxidant, resulting in oxidative damage to hepatocytes by elevating free radical production during cellular oxidation processes in liver tissue. The increasing prevalence of AAS such as dianabol, particularly among athletes and bodybuilders, raises significant concerns regarding their impact on health and performance (17). Dianabol has gained notoriety for its potent anabolic effects, leading to widespread misuse in pursuit of enhanced athletic performance (18). This misuse is not without consequences. While users may experience rapid increases in muscle mass and strength, the associated risks include severe liver damage, cardiovascular issues, and hormonal imbalances (19). Furthermore, the oxidative stress induced by AAS can exacerbate apoptosis in liver cells, leading to further complications. Dianabol-induced liver damage can lead to the activation of Kupffer cells, which then release inflammatory mediators and ROS, further exacerbating liver damage (20).
Given the adverse effects of AAS such as dianabol on liver function, it is crucial to explore potential protective strategies, such as the supplementation of antioxidants like selenium and vitamin E, as well as natural compounds like clove extract, which may mitigate the hepatotoxic effects of steroids and support liver health.
2. Objectives
The present study aims to investigate the protective effects of selenium, vitamin E, and the methanolic extract of clove on the expression of apoptotic genes in the liver of rats subjected to dianabol administration. By elucidating the interplay between these antioxidants and apoptotic pathways, this study intends to contribute valuable insights into potential therapeutic strategies for mitigating dianabol-induced hepatotoxicity.
3. Methods
3.1. Chemicals
The materials used in the experiments included the AST and ALT kits, both sourced from Iran and produced by Pars Azmoon Company. Additionally, the total antioxidant capacity (TAC) kit was obtained from Zell Bio Company in Iran. Dianabol and selenium were supplied by Power Pnorma and Hi Health, respectively, also based in Iran. Vitamin E was provided by Karen Pharmaceutical Company, and clove was sourced locally in Iran. The clove was authenticated by a specialist to confirm its identity and quality before use. Double-distilled water was obtained from Sky Pharmaceutical, a manufacturer in Iran. These materials were essential for conducting the experiments and ensuring accurate results.
3.2. Animal Experimental Design
In this research, thirty-three adult male Wistar rats, each with an average weight of 250 g, were obtained from the Royan Research Institute, Iran. The experimental protocol was approved by the Ethics Committee of Islamic Azad University, Falavarjan Branch, following ethical standards (IR.IAU.FALA.RCE.1402.014). The rats were maintained under a 12-hour dark-light cycle and had free access to food and water. They were divided into six groups, with bedding (sawdust) changed every other day, and the daily intake of water and food was monitored. The cages were cleaned and disinfected every ten days using a 10% saline solution and 70% ethanol.
Prior to the test procedures, the animals were carefully weighed and grouped. For one week, the rats were treated with amoxicillin at a dosage of 1 mg/L, followed by a one-week rest period before the practical phase of the study commenced. Amoxicillin was used to prevent bacterial infections and maintain a healthy microbial environment, which helps reduce confounding variables and ensures that observed effects are more reliably attributed to the experimental treatments.
The nutrient levels in the rat food per 100 grams included crude protein at 11.5 - 23.5 g, crude fat at 4.5 - 9.5 g, and crude fiber at 4 - 4.5 g. The ash content was a maximum of 21 g, while calcium was present at 1.35 - 2 g and phosphorus at 1.65 - 1.7 g. Salt content was included at a level of 1.51 - 1.55 g, with a maximum moisture content of 21 g. Additionally, the food contained methionine at 1.99 g, lysine at 2.25 g, methionine plus cysteine at 1.79 g, and tryptophan at 1.15 g per 100 grams of food.
The rats were randomly divided into six groups, each containing six members. The control group (group 1) received 0.3 mL of physiological serum daily via oral gavage, while group 2 was treated with dianabol at 15 mg/kg of body weight. Group 3 received dianabol at 15 mg/kg along with vitamin E at 100 IU/kg, and group 4 received dianabol at 15 mg/kg combined with selenium at 0.5 mg/kg. Group 5 was treated with dianabol at 15 mg/kg along with clove extract at 4 mg/kg, while group 6 received a combination of dianabol at 15 mg/kg, vitamin E at 100 IU/kg, clove extract at 4 mg/kg, and selenium at 0.5 mg/kg. All treatments were administered daily via oral gavage at 8:00 AM for a duration of 42 days following a one-week rest period after the administration of amoxicillin.
3.3. Supplementation
Tablets containing 10 mg of dianabol were dissolved in physiological serum, and 1 mL of this solution was administered via gavage to each rat daily. Selenium was prepared weekly for the rats using 200 µg tablets, with a daily oral gavage dose of 1.5 mg. The methanolic clove extract was prepared using the maceration method. Dried cloves were first ground into a fine powder. Ten grams of the powdered cloves were soaked in methanol at room temperature in a sealed container. The mixture was left to macerate for 24 - 72 hours, during which it was occasionally stirred to enhance the extraction of bioactive compounds. After the maceration period, the mixture was filtered. The resulting methanolic extract was then concentrated by evaporating the solvent under reduced pressure using a rotary evaporator, yielding a concentrated clove extract. This extract was dissolved in normal saline and administered at a dosage of 4 mg/kg in the study, with fresh clove extract prepared weekly to ensure optimal potency and efficacy (21, 22). For vitamin E supplementation, vitamin E was mixed with olive oil, which was then stored in the refrigerator. One mL of this mixture was administered to the rats via gavage daily. Based on previous research demonstrating the protective effects of selenium and vitamin E against steroid-induced damage, as well as the antioxidant properties of clove, the extract dose was selected to investigate their combined impact on apoptotic gene expression in the liver of rats treated with dianabol (9-11). A pilot study was also conducted to inform the selection of these doses. Selenium and vitamin E were selected in this study due to their well-documented antioxidant properties and their established roles in mitigating oxidative stress and enhancing cellular defense mechanisms (8).
3.4. Blood Sampling and Separation of Serum
Blood sampling from rats was performed 24 hours after the last injection, utilizing a syringe to collect blood directly from the heart through the cardiac puncture method while the rats were anesthetized with a ketamine and xylazine mixture to minimize interference with liver enzymes. After collection, the blood samples were allowed to clot at 37°C for 10 minutes, then centrifuged at 3,000 rpm for 5 minutes to separate the serum, which was stored at -20°C until further testing. Following blood sampling, the rats were euthanized humanely using an overdose of the ketamine and xylazine mixture to ensure they were unconscious and insensible to pain. Once euthanasia was confirmed, the abdominal cavity was opened using sterile surgical instruments to access the liver. The liver was carefully excised, rinsed with sterile saline to remove blood and debris, and handled aseptically to prevent contamination. A portion of the liver was immediately placed in cryogenic tubes and stored at -70°C for RNA extraction and molecular analyses, while additional samples could be fixed in formalin for histopathological studies if needed.
3.5. Biochemical Estimations
After blood collection, the serum samples were transported to the laboratory for analysis, where liver function tests, specifically ALT and AST, were measured using the Pars Azmoun autoanalyzer and corresponding kits.
3.6. RNA Extraction and cDNA Synthesis
RNA extraction was performed using the SambioTM column extraction kit, starting with 10 mg of rat liver tissue, which was homogenized and treated with a lysing solution containing phenol and chloroform. After centrifugation, the supernatant was mixed with cold isopropanol to precipitate RNA, which was then washed with ethanol and eluted for storage at -70°C. cDNA synthesis was conducted using the SambioTM kit, involving a mixture that underwent incubation for reverse transcription followed by heating to terminate the reaction, with the synthesized cDNA stored at -20°C for future analyses.
3.7. Quantitative Reverse Transcription Polymerase Chain Reaction
Specific primers were designed to assess the expression levels of the following genes: BAX (forward: 5'-TTG CTA CAG GGT TTC ATC CA-3', reverse: 5'-GAG TAC CTG AAC CGG CAT CT-3'), BCL2 (forward: 5'-GAG TAC CTG AAC CGG CAT CT-3', reverse: 5'-GAA ATC AAA CAG AGG TCG CA-5'), and GAPDH (forward: 5'-AGATTTGGACCTGCGAGCG-3', reverse: 5'-GAGCGGCTGTCTCCACAAGT-3'). GAPDH was used as an internal control. The relative expression level of the target gene was calculated using the 2-ΔΔCt approach, which is based on the difference between the threshold cycle (Ct) of the target gene and that of the internal control gene.
3.8. Statistical Analysis
We employed one-way ANOVA and the Kruskal-Wallis test to compare means across six groups, including a control group, based on data distribution. If the data followed a normal distribution, one-way ANOVA was used; otherwise, the Kruskal-Wallis test was applied. A significance level of 0.05 was set for all analyses, which were conducted using SPSS version 20 software.
4. Results
4.1. Assessment of Aspartate Aminotransferase and Alanine Aminotransferase Activities
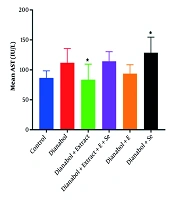
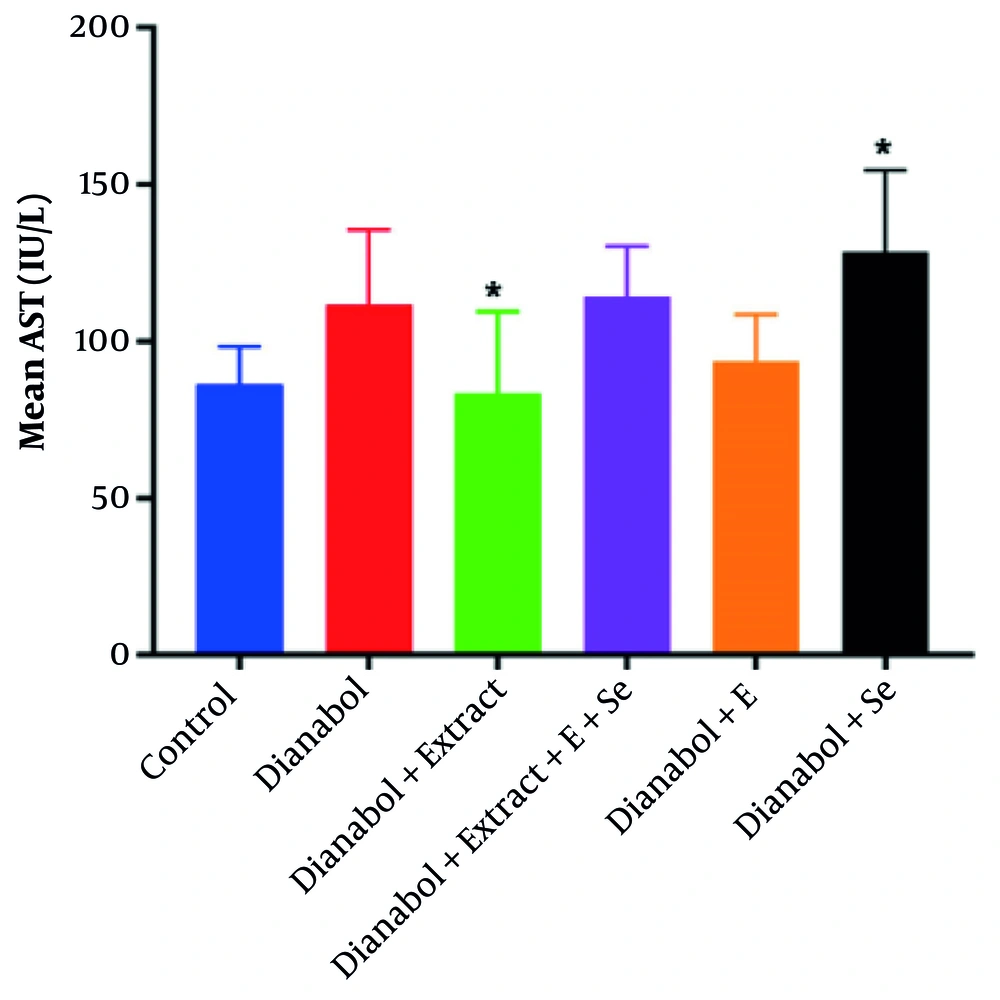
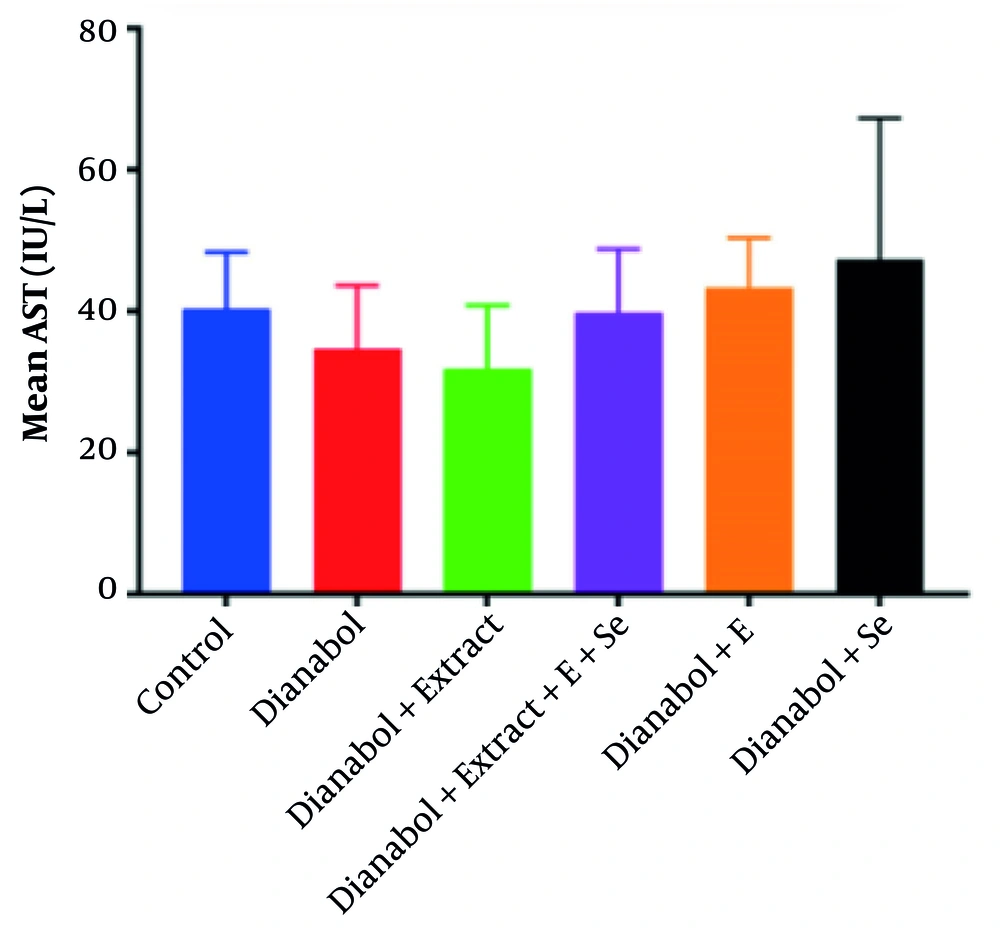
A significant increase in AST activity was observed in the group that received a combination of dianabol and selenium compared to the control group (P = 0.027) (Figure 1). Conversely, a significant decrease in AST activity was noted in the group treated with a combination of dianabol and clove extract compared to the control group (P = 0.022). No significant differences in ALT activity were found between any of the treated and untreated groups (Figure 2).
Aspartate aminotransferase (AST) activity levels in the treated groups. Data are presented comparing the control group with groups receiving “dianabol”, “dianabol + extract”, “dianabol + extract + E (Vitamin E) + Se (selenium)”, “dianabol + E” and “dianabol + Se”. A significant increase in AST activity was observed in the dianabol + Se group compared to the control group (P = 0.027), and a significant decrease was observed in the dianabol + extract group compared to the control group (P = 0.022). * Indicates significant difference compared with control group at P < 0.05.
Alanine aminotransferase (ALT) activity levels in the treated groups. Data are presented comparing the control group with groups receiving “dianabol”, “dianabol + extract”, “dianabol + Extract + E (vitamin E) + Se (selenium)”, “dianabol + E” and “dianabol + Se”. No significant differences were found between ALT values among the treated and untreated group.
4.2. Investigating Total Antioxidant Capacity Activity
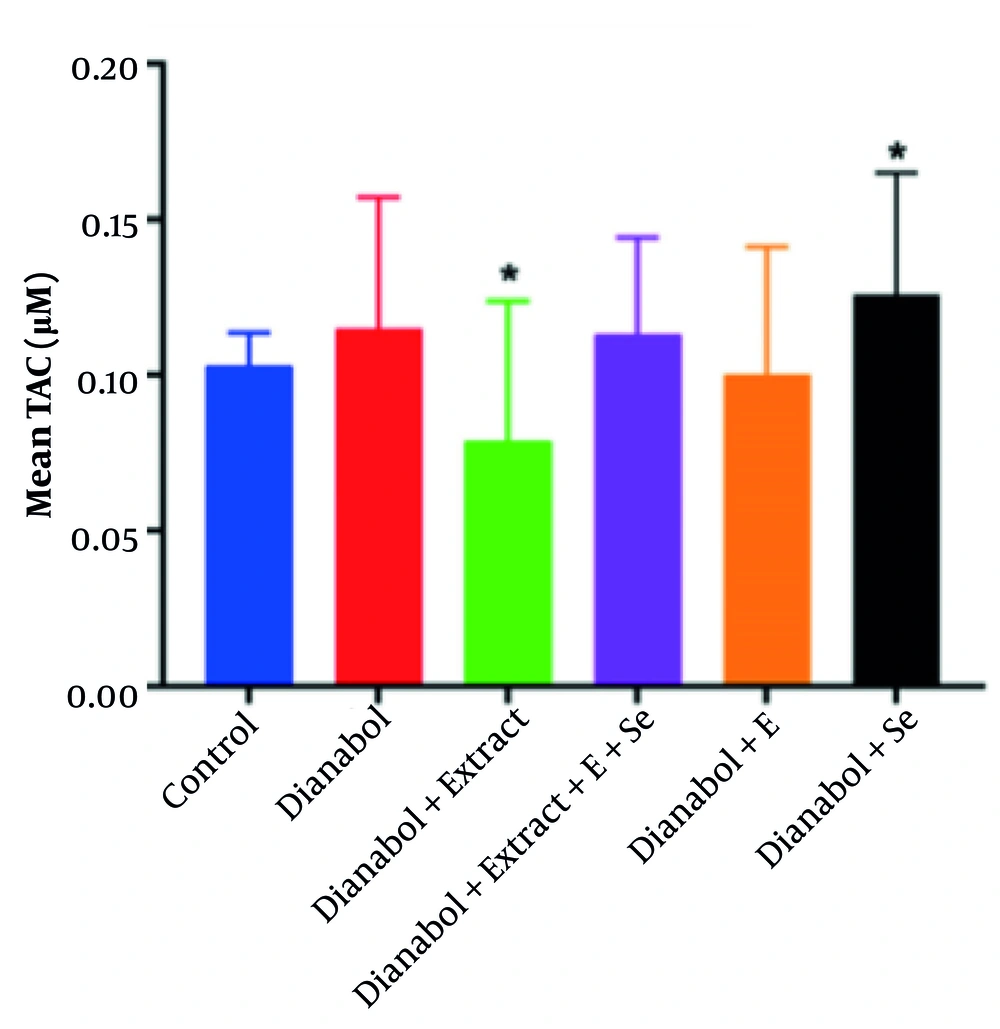
Total antioxidant capacity is a measure of the cumulative antioxidant capacity of all the antioxidant compounds present in a given sample. Assessing TAC allows evaluation of the compounds’ overall ability to counteract oxidative stress damage. This metric provides valuable insights into the total antioxidant levels in the body and serves as an indicator of the body’s defense mechanisms against the harmful effects of free radicals and ROS. A significant increase in TAC was observed in the group that received a combination of dianabol and selenium compared to the control group (P < 0.05) (Figure 3). Conversely, the group that received a combination of dianabol and clove exhibited a significant decrease in TAC activity compared to the control group (P < 0.05). However, the one-way ANOVA parametric test did not reveal any significant differences among all six groups (P = 0.417).
Total antioxidant capacity (TAC) in the treated groups. Data are presented comparing the control group, dianabol group, dianabol + extract group, dianabol + extract + E (vitamin E) + selenium group, dianabol + vitamin E group and dianabol + selenium group. * Indicates significant difference compared with control group at P < 0.05.
4.3. Evaluation of the Expression of Apoptosis-Related Genes
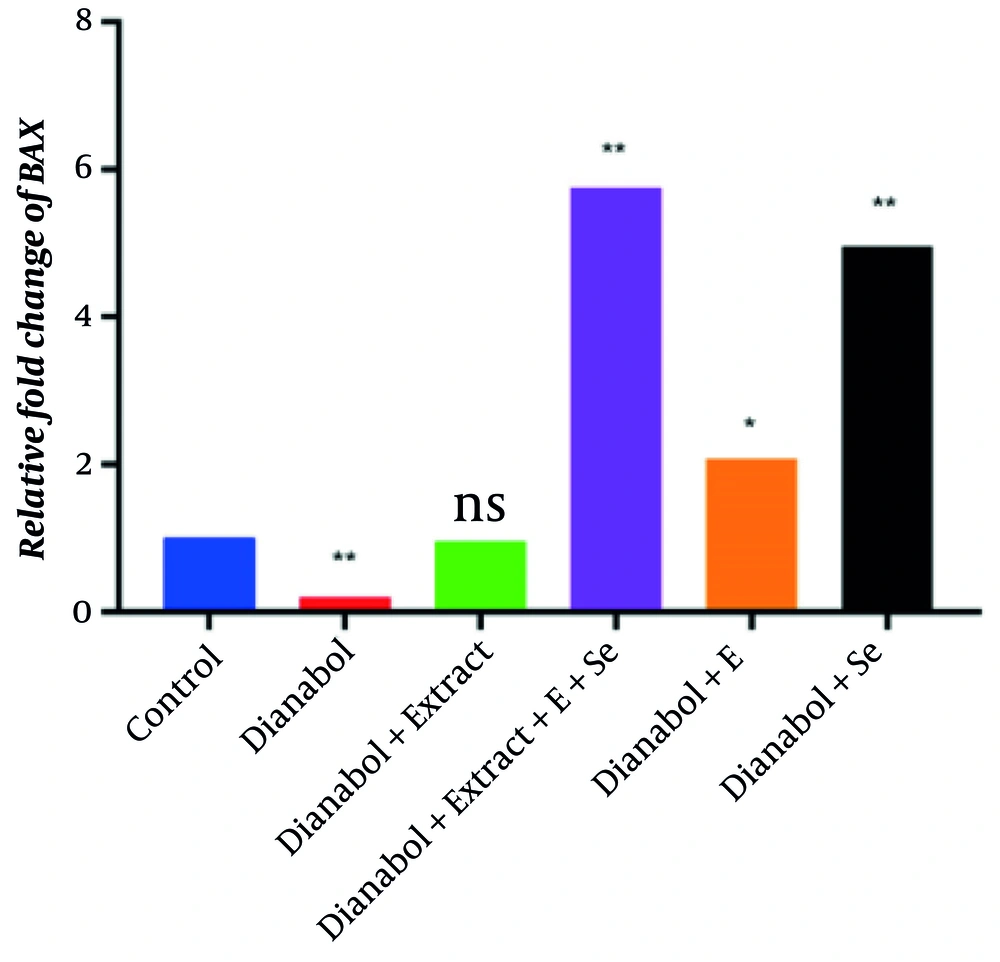
The qRT-PCR analysis assessed BAX and BCL2 gene expression to evaluate the apoptotic effects of various compounds. The steroid treatment group exhibited the lowest BAX expression and the highest BCL2 expression, indicating that steroids significantly suppress pro-apoptotic gene expression (P < 0.01). In contrast, the dianabol and selenium treatment group showed the highest BAX expression, suggesting that selenium counteracts the suppressive effects of dianabol on apoptosis. Clove extract administration increased BAX expression to levels comparable to the control group, with no significant difference. However, selenium and combined treatments with clove, selenium, and vitamin E significantly increased BAX expression beyond control levels (P < 0.01) (Figure 4). These findings suggest that while dianabol treatment reduces BAX expression, the addition of clove, selenium, or vitamin E can counteract this effect and enhance pro-apoptotic signaling.
Relative fold change of BAX gene in control group and groups treated with dianabol, dianabol + extract, dianabol + Se (selenium), dianabol + vitamin E (E), and dianabol + extract + Se + E. * and ** indicate significant difference compared with control group at P < 0.05 and P < 0.01, respectively.
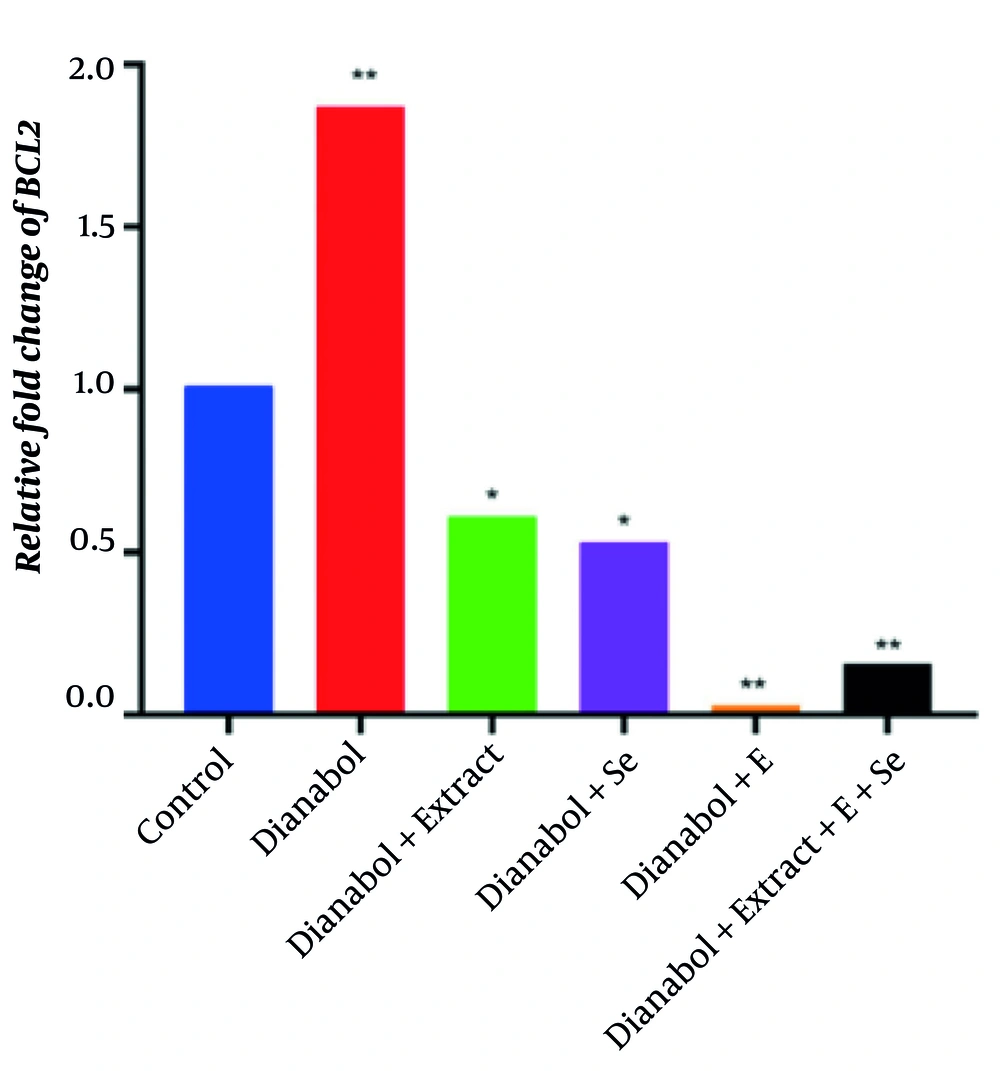
The comparison of BCL2 gene expression results between the experimental groups and the control group is presented in Figure 5. Pairwise comparisons of the mean results using the t-test showed that the dianabol and vitamin E treatment group had the lowest BCL2 gene expression, while the dianabol treatment group exhibited the highest expression level, indicating that dianabol treatment significantly increased BCL2 gene expression compared to the control group (P < 0.01). However, the administration of vitamin E and the combined treatment with clove, selenium, and vitamin E mitigated the dianabol effect and reduced BCL2 gene expression levels (P < 0.01). Furthermore, the groups treated with clove and those treated with selenium also exhibited a significant reduction in BCL2 gene expression level compared to the control group (P < 0.05). These findings suggest that while dianabol treatment significantly increased BCL2 gene expression, the addition of vitamin E, either alone or in combination with clove and selenium, mitigated this effect and reduced BCL2 gene expression to levels lower than those observed in the control group. The clove and selenium treatments also demonstrated a moderating effect on BCL2 gene expression compared to the control group.
Relative fold change of BCL2 gene in control group and groups treated with control group and groups treated with dianabol, dianabol + extract, dianabol + Se (selenium), dianabol + vitamin E (E), and dianabol + extract + Se + E. * and ** indicate significant difference compared with control group at P < 0.05 and P < 0.01, respectively.
5. Discussion
This study investigated the impact of selenium, vitamin E, and clove extract, both alone and in combination, on AST and ALT activity, TAC, and the expression of apoptosis-related genes in the liver of rats treated with dianabol. Our results indicated a significant increase in AST activity due to the combined effects of dianabol and selenium. This finding aligns with previous studies demonstrating that dianabol contributes to elevated liver enzyme levels, particularly AST, which serves as a crucial marker of hepatocellular injury. Elevated AST levels are often associated with liver stress and damage, reinforcing concerns about the hepatotoxic effects of anabolic steroid use (23). The increase in AST activity suggests that while selenium may provide some protective effects against the hepatotoxicity associated with dianabol, the combination still results in elevated liver enzyme levels, indicating ongoing liver stress and damage.
Conversely, the group that received a combination of dianabol and clove extract exhibited a significant decrease in AST activity compared to the control group. This suggests that clove extract may possess hepatoprotective properties, potentially mitigating the liver toxicity induced by dianabol. The antioxidant and anti-inflammatory compounds present in clove extract could play a crucial role in reducing liver enzyme levels and protecting hepatocytes from damage. This protective effect has been observed in previous studies, which have demonstrated that eugenol-rich fractions of clove can reverse biochemical and histopathological changes in liver cirrhosis and inhibit hepatic cell proliferation (24). Additionally, clove bud polyphenols have been shown to modulate alcohol-induced oxidative stress and inflammation, further supporting their hepatoprotective potential (25).
Clove contains a diverse array of bioactive compounds, including phenolic compounds like eugenol, eugenol acetate, gallic acid, and hydroxybenzoic acids. Additionally, it contains terpenoids such as β-caryophyllene, α-humulene, monoterpenes, and sesquiterpenes. Other components include flavonoids (e.g., luteolin, quercetin), tannins, triterpenoids, chromones, and coumarins. These diverse bioactive compounds play a significant role in exerting protective effects against oxidative stress by mitigating oxidant reactions (11, 12). Phenolic compounds such as eugenol, eugenol acetate, and thymol have been reported to contribute to clove’s strong antioxidant capabilities through multiple mechanisms. These bioactive compounds not only neutralize free radicals but also exhibit antimicrobial effects, making clove a valuable natural preservative in food systems (26). Additionally, clove essential oil has demonstrated therapeutic potential by modulating pro-inflammatory mediators, oxidative stress, and metabolic enzyme activities, further enhancing its health benefits (27).
The TAC is a critical biomarker used to assess the cumulative antioxidant potential within biological samples, providing a measure of the organism’s capacity to mitigate oxidative stress. In this study, a significant increase in TAC was observed in the experimental group receiving the combination of dianabol and selenium, compared to the control group. This suggests that the combination may enhance the organism’s overall antioxidant defense mechanisms, potentially offering a protective effect against the oxidative damage induced by dianabol. The increase in TAC, particularly in the presence of selenium, implies a more efficient neutralization of free radicals and ROS, thus contributing to the maintenance of liver health and reducing oxidative stress.
The group receiving a combination of dianabol and clove showed a significant decrease in TAC compared to the control group, raising questions about the interaction between clove and dianabol. This is a somewhat unexpected finding. It suggests that clove extract, despite its known antioxidant properties (13), may have interfered with the TAC assay or had a complex interaction with dianabol affecting overall antioxidant capacity. The observed decrease in TAC in the dianabol + clove group compared to controls presents a complex scenario. This seemingly contradictory result may stem from several possibilities: The methanolic clove extract may be interfering with the TAC assay itself, leading to inaccurate readings, or clove components interacting with dianabol and altering their redox properties to exhibit a pro-oxidant effect under these specific conditions. The fact that the addition of selenium shows an increase in TAC while the addition of clove decreases highlights the complex interaction between dianabol and the extract and the need for further research to understand how dietary antioxidants and anabolic steroids affect oxidative stress and liver function.
In this study, we observed significant alterations in the gene expressions of BAX and BCL2 among the experimental groups. The group treated with dianabol showed a marked reduction in BAX expression and an increase in BCL2 expression compared to the control group. These findings suggest that anabolic steroids may modulate the balance between pro-apoptotic and anti-apoptotic proteins, potentially suppressing pro-apoptotic gene expression while enhancing cell survival. The lower BAX expression and higher BCL2 expression in the steroid-treated group indicate a potential shift toward a more anti-apoptotic environment, which may favor muscle cell survival and protect against oxidative or stress-induced damage (28, 29). These results highlight the significant impact of anabolic steroids on apoptosis regulation, which could have important implications for tissue survival in muscle and other affected tissues.
In contrast, the group treated with dianabol and selenium exhibited increased BAX expression, suggesting that selenium may counteract the suppressive effects of dianabol on apoptosis. This could be attributed to selenium’s antioxidant properties and its ability to modulate cellular signaling pathways. Interestingly, clove extract treatment resulted in normalized BAX expression levels comparable to the control group, while simultaneously decreasing BCL2 expression. This suggests that clove extract may help restore the apoptotic signaling disrupted by dianabol (30). Certain selenium compounds have been shown to activate caspases, which are essential enzymes in the apoptosis process, thereby facilitating cell death. Furthermore, combined treatments of clove, selenium, and vitamin E significantly increased BAX expression while decreasing BCL2 expression beyond the levels observed in the control group, indicating a potential synergistic effect in promoting apoptosis (31). The increased BAX expression in this combination group suggests that selenium may enhance pro-apoptotic signaling, potentially offering a protective effect against steroid-induced cellular alterations.
Co-administration of clove extract with dianabol resulted in the normalization of BAX gene expression to levels comparable to those in the control group, accompanied by a reduction in BCL2 expression. This suggests that clove extract may effectively restore apoptotic signaling pathways that are disrupted by dianabol use. Moreover, the combination of clove extract, selenium, and vitamin E led to a significant increase in BAX expression and a decrease in BCL2 expression beyond the levels observed in the control group, indicating a possible synergistic effect in promoting apoptosis. These findings strengthen the notion that antioxidants can modulate pro-apoptotic signaling in the presence of steroid-induced alterations.
This study has limitations in its scope of analysis. While the study focused on gene expression and select biochemical markers, it did not include histological examination of liver tissue, which could have provided direct evidence of cellular damage and structural changes. Furthermore, electron microscopy could have offered ultrastructural insights into organelle changes and cellular stress. In addition, the study did not quantify changes in key proteins and enzymes involved in antioxidant activity (e.g., SOD, glutathione peroxidase) or apoptosis (e.g., caspase-3 activation, cytochrome c release), which would have provided a more comprehensive understanding of the mechanisms involved. Future studies should incorporate these additional analyses to better characterize the protective or detrimental effects of selenium, vitamin E, and clove extract on dianabol-induced hepatotoxicity.
5.1. Conclusions
In conclusion, this study provides valuable insights into the effects of dianabol on liver function and apoptotic gene expression, as well as the potential protective role of antioxidants such as selenium, clove extract, and vitamin E. By demonstrating how dianabol significantly alters AST activity, TAC, and apoptosis-related gene expression, this research fills an important gap in understanding the molecular mechanisms behind steroid-induced liver damage. The study also underscores the potential of combining antioxidants to mitigate the harmful effects of anabolic steroid misuse, particularly in bodybuilding and sports. These findings have significant implications for athletes and bodybuilders who may misuse anabolic steroids to enhance performance or physique. The results suggest that supplementation with antioxidants such as selenium, clove, and vitamin E could offer a therapeutic strategy to protect against the oxidative damage and apoptotic effects associated with long-term steroid use, promoting better liver health and overall well-being.