1. Background
Breast cancer remains one of the most common malignancies among women worldwide, with an estimated 2.3 million new cases diagnosed in 2020. The MCF7 cell line, derived from a pleural effusion of a patient with metastatic breast cancer, is widely used as a model for studying estrogen receptor-positive (ER+) breast cancer. MCF7 cells exhibit characteristics typical of luminal A breast cancer, making them a valuable tool for investigating hormone-dependent pathways and therapeutic responses (1, 2). Conversely, ovarian cancer is often diagnosed at an advanced stage due to its asymptomatic nature in early stages, leading to a poor prognosis. The A2780 cell line, established from a patient with ovarian serous papillary carcinoma, serves as a representative model for studying high-grade serous ovarian cancer. These cells are known for their aggressive behavior and resistance to chemotherapy, particularly platinum-based agents like cisplatin (3).
The contrasting characteristics of MCF7 and A2780 cells provide an excellent opportunity to study the effects of steviol glycosides across different cancer types. Steviol glycosides, natural sweeteners derived from the leaves of Stevia rebaudiana, have gained increasing attention in recent years not only for their use as sugar substitutes but also for their potential therapeutic effects, particularly in cancer treatment. These compounds, including steviol glycoside and rebaudioside A, are known to possess various biological activities, including antioxidant, anti-inflammatory, and anticancer properties (4).
Given the rising incidence of breast and ovarian cancers, understanding the effects of steviol glycosides on cancer cell lines such as MCF7 and A2780 is of significant clinical relevance. Research has indicated that steviol glycosides can induce apoptosis in various cancer cell lines through several mechanisms (5). One key pathway involves the modulation of the mitochondrial apoptotic pathway. Studies have shown that treatment with steviol glycosides can increase the expression of pro-apoptotic proteins such as Bax while decreasing anti-apoptotic proteins like Bcl-2. This shift in the Bax/Bcl-2 ratio promotes mitochondrial membrane permeabilization and subsequent release of cytochrome c into the cytosol, triggering caspase activation and apoptosis (6, 7).
In addition to inducing apoptosis, steviol glycosides have been found to affect other cellular functions critical for cancer progression. For instance, they can inhibit cell proliferation by interfering with cell cycle progression. Research has demonstrated that steviol glycosides can induce G0/G1 phase arrest in various cancer cell lines, thereby limiting their ability to proliferate. This effect is particularly relevant in the context of breast and ovarian cancers, where uncontrolled cell division is a hallmark of malignancy (5, 6).
Steviol glycosides have shown promise as anticancer agents due to their multifaceted effects on tumor cell lines. They inhibit glucose metabolism in pancreatic cancer cells, reducing proliferation via the mTOR signaling pathway. Steviol induces apoptosis primarily through the mitochondrial pathway, increasing the Bax/Bcl-2 ratio and activating caspases, while also upregulating tumor suppressor proteins like p53 and p21. Additionally, steviol promotes G1 phase cell cycle arrest by modulating key regulatory proteins such as Cyclin D1. The disruption of translation initiation further enhances its anticancer effects by inhibiting protein synthesis essential for tumor growth. Collectively, these findings suggest steviol glycosides could be valuable therapeutic agents in cancer treatment, warranting further investigation into their mechanisms and clinical applications (4).
The comparative study of MCF7 and A2780 cell lines provides valuable insights into how steviol glycosides exert their effects differently based on the inherent characteristics of each cell type. For instance, MCF7 cells are ER+ and respond to estrogen stimulation; therefore, the impact of steviol glycosides on these cells may involve modulation of estrogen signaling pathways. Research has suggested that steviol glycosides can inhibit estrogen-induced proliferation in MCF7 cells by downregulating estrogen receptor expression or blocking downstream signaling pathways associated with cell proliferation (2). Conversely, A2780 cells are typically ER-negative and exhibit a different set of signaling pathways driving their growth and survival. The aggressive nature of A2780 cells often leads to resistance against conventional chemotherapeutics. Steviol glycosides offer a promising approach to cancer treatment due to their broad anticancer activity, low toxicity, selective targeting of cancer cells, and potential to enhance chemotherapy efficacy. Investigating how steviol glycosides interact with these resistant pathways could reveal novel therapeutic strategies for overcoming drug resistance in ovarian cancer (3).
The exploration of steviol glycosides' effects on MCF7 and A2780 cell lines provides valuable insights into their potential as anticancer agents in breast and ovarian cancers. Through mechanisms such as apoptosis induction, modulation of cellular functions like proliferation and migration, and potential synergy with conventional therapies, steviol glycosides present an intriguing avenue for further research. It is also worth noting that stevioside is less toxic to normal cells even at higher doses (4, 6, 7).
2. Objectives
The rationale for selecting the MCF-7 and A2780 cell lines lies in their distinct molecular profiles and relevance to studying breast and ovarian cancers, respectively. MCF-7 is an estrogen receptor-positive (ER+) breast cancer cell line, making it a widely used model for hormone-responsive cancers. It allows researchers to investigate the role of estrogen signaling in tumor proliferation and response to endocrine therapies. On the other hand, A2780 is a human ovarian cancer cell line derived from an untreated patient, frequently used to study chemoresistance mechanisms and drug action in ovarian carcinoma. These cell lines were chosen over others due to their ability to represent different molecular subtypes of cancer (e.g., ER+ versus ER-negative) and their established use in modeling hormone-driven and chemoresistant tumor behaviors. This selection facilitates comparative analyses of apoptosis and therapeutic responses across two distinct yet clinically significant cancer types.
3. Methods
3.1. Cell Culture
In this study, we aimed to investigate the effects of steviol glycosides on two cancer cell lines obtained from Institut Pasteur Iran: MCF-7, derived from human breast cancer, and A2780, originating from human ovarian cancer. Both cell lines were cultured under standard conditions in RPMI-1640 medium (Gibco), supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin solution (Gibco). Once the cells reached an appropriate density of approximately 80%, they were treated with varying concentrations of steviol glycosides (0, 10, 20, 30, 40, and 50 µM) for different time points (24, 48, and 72 hours). The concentrations used in these studies were determined based on preliminary findings from the MTT assay, which assessed cell viability after exposure to various concentrations of steviol glycoside.
3.2. Cell Viability Test
To assess the impact of steviol glycoside on cell viability, we employed the MTT assay. After treatment, MTT solution was added to each well, allowing viable cells to convert the MTT into formazan crystals. Following a 4-hour incubation period, the crystals were dissolved in dimethyl sulfoxide (DMSO), and the absorbance was measured at 570 nm using a microplate reader. This enabled the calculation of cell viability as a percentage of the untreated control group.
3.3. Apoptosis Assays
RNA extraction was performed using the ZiAzole kit from ZiAViZ Company. For the apoptosis assay, the expression levels of BAX, BAD, and BCL2 genes were evaluated, using β-actin as the reference gene. The primer sequences are listed in Table 1.
| Primer Name | Sequence of Primer | Melting Temperature°C |
|---|---|---|
| Bata-actin | F: 5'-GGAGTCCTGTGGCATCCACG-3' | 62.86 |
| R: 5'-CTAGAAGCATTTGCGGTGGA-3' | 58.26 | |
| BAX | F: 5'- GGCCCACCAGCTCTGAGCAGA-3' | 66.52 |
| R: 5'- GCCACGTGGGCGGTCCCAAAGT-3' | 69.96 | |
| BAD | F: 5'-CAGTGATCTGCTCCACATTC-3' | 56.22 |
| R: 5'-TCCAGCTAGGATGATAGGAC-3' | 55.20 | |
| Bcl-2 | F: 5'-GTGGAGGAGCTCTTCAGGGA-3' | 60.62 |
| R: 5'-AGGCACCCAGGGTGATGCAA-3' | 64.29 |
Primer Sequences Used in This Study
3.4. Statistical Analysis
Statistical analyses were performed using a t-test, with P-values calculated based on a significance threshold of 0.05. Graphs were generated using GraphPad Prism software. A P-value of less than 0.05 is indicated with (*), a P-value of less than 0.01 is indicated with (**), and a P-value of less than 0.001 is indicated with (***).
4. Results
4.1. Cell Viability
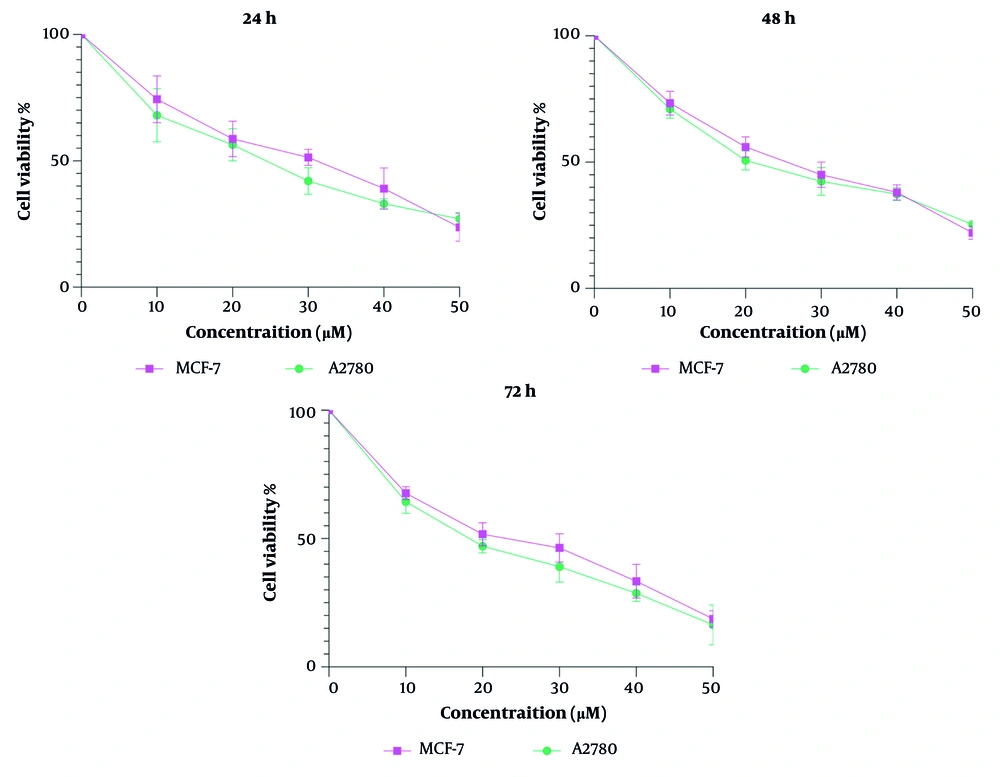
The MTT assay results (Figure 1) demonstrated that steviol glycoside exhibits significant cytotoxic effects on both MCF-7 and A2780 cell lines, which represent breast and ovarian cancer, respectively. The inhibitory concentration 50% (IC50) values for MCF-7 cells were 30, 24, and 22 µM at 24, 48, and 72 hours, respectively. For A2780 cells, the IC50 values were 24, 20, and 19 µM at the same time points. The biological significance of the reported IC50 lies in its ability to provide insights into the potency, efficacy, and safety of therapeutic agents. This measurement is invaluable across various stages of drug development and research, allowing scientists to make informed decisions and enhance our understanding of biological interactions and therapeutic strategies.
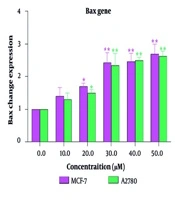
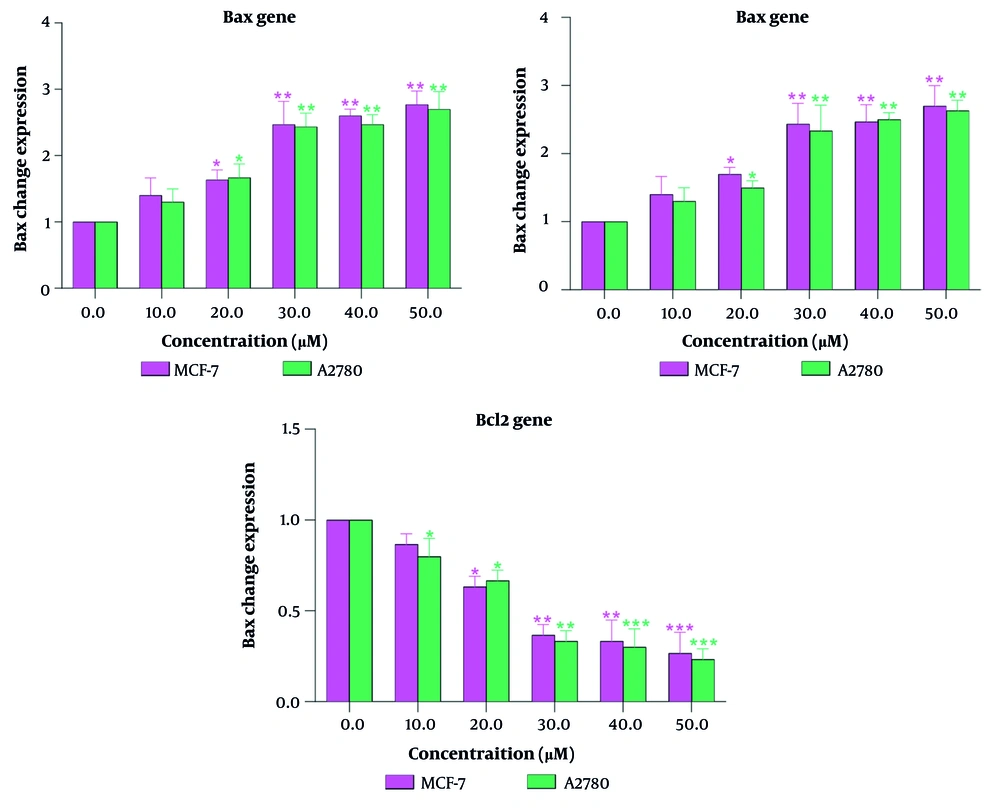
4.2. Gene Expression of Bax, Bad, and Bcl-2
Specifically, treatment with steviol glycoside led to an increase in the expression of apoptotic proteins, such as the pro-apoptotic genes Bax and Bad, and a decrease in the expression of the anti-apoptotic gene Bcl-2 in the MCF-7 cell line, indicating its potential as an effective anticancer agent. Similar findings were observed in the A2780 cell line (Figure 2), where the cytotoxicity of steviol glycoside was attributed to its ability to trigger apoptosis. The fold change of the Bax and Bad genes increased as the concentration of steviol glycoside was increased. At concentrations above 30 μM, this increase reached twice the baseline level. Conversely, the expression of the Bcl-2 gene decreased to half its control level. Overall, the data suggest that steviol glycoside not only reduces cell viability in both cancer cell lines but also promotes apoptotic pathways, highlighting its potential therapeutic applications in treating breast and ovarian cancers. Notably, the changes in apoptosis across different concentrations and time points were similar between the two cell lines.
5. Discussion
Researchers are actively engaged in developing new pharmaceutical agents, therapeutics, natural products, and innovative drug delivery systems to enhance treatment efficacy and minimize side effects (8-10). Steviol glycoside, a natural compound derived from the S. rebaudiana plant, has gained recognition for its potential as an anticancer agent. This discussion explores the mechanisms by which steviol glycoside induces apoptosis in cancer cells, particularly focusing on its effects on breast (MCF-7) and ovarian (A2780) cancer cell lines. The findings suggest that steviol glycoside operates through multiple pathways, including the generation of reactive oxygen species (ROS), modulation of apoptotic proteins, and regulation of the cell cycle (5).
Another significant aspect of steviol glycoside's anticancer effects is its ability to modulate cell cycle regulators. Specifically, it induces G1 phase arrest in various cancer cell lines by upregulating proteins such as p21 and p53 while downregulating Cyclin D1. This cell cycle arrest not only inhibits proliferation but also sensitizes cells to undergo apoptosis when exposed to steviol glycoside (11). The concentration-dependent effects observed in studies indicate that higher concentrations of steviol glycoside correlate with increased apoptosis rates. This relationship reinforces the notion that dosage plays a crucial role in its anticancer efficacy. Identifying effective concentrations for inducing apoptosis without adversely affecting normal cells could enhance therapeutic applications.
Beyond inducing apoptosis, steviol glycoside may also influence cellular migration and invasion, key processes involved in metastasis. In vitro studies have shown that these compounds can inhibit the migratory capabilities of cancer cells by downregulating matrix metalloproteinases (MMPs), enzymes involved in extracellular matrix degradation (12). By inhibiting MMP expression and activity, steviol glycosides may reduce the invasive potential of both MCF-7 and A2780 cells.
Results from MTT assays indicate that steviol glycoside exhibits significant cytotoxic effects on both MCF-7 and A2780 cell lines, highlighting its potential as an effective anticancer agent. The observed increase in pro-apoptotic transcripts such as Bax and Bad, coupled with a decrease in Bcl-2 levels in the MCF-7 cell line, underscores the compound's ability to induce apoptosis. The consistency of these findings across both MCF-7 and A2780 cell lines reinforces the idea that steviol glycoside could be a promising candidate for further development as an anticancer therapeutic.
Data collectively suggest that steviol glycoside not only reduces cell viability but also actively promotes apoptotic pathways in both cancer cell lines. Notably, changes in apoptosis across different concentrations and time points were similar between MCF-7 and A2780 cells, indicating a potentially universal mechanism of action for steviol glycoside against various cancer types. Additionally, exploring potential synergistic effects with existing chemotherapeutic agents may reveal new therapeutic strategies for treating breast and ovarian cancers. Combining steviol glycoside with other treatments could enhance overall efficacy while minimizing side effects associated with conventional chemotherapy.
The promising results from studies on steviol glycoside highlight its potential as a novel anticancer agent. Its ability to induce apoptosis through modulation of key apoptotic proteins positions it as a candidate for further research aimed at developing effective treatments for breast and ovarian cancers. The investigation into the effects of steviol glycoside on MCF-7 and A2780 cell lines reveals significant insights into its potential as an anticancer agent (13, 14). The capacity of steviol glycoside to induce apoptosis at varying concentrations and time points further suggests a dose-dependent relationship crucial for optimizing its therapeutic application. Moreover, consistent changes in apoptotic pathways across both cell lines highlight its promise as a candidate for further research and development in cancer therapy.
In conclusion, findings from this study support the hypothesis that steviol glycoside possesses significant anticancer properties through its ability to induce apoptosis in breast and ovarian cancer cell lines. Future studies on steviol glycosides can focus on understanding the precise mechanisms by which they induce apoptosis in cancer cells, particularly breast and ovarian cancers. Expanding research to include diverse cancer cell lines and conducting in vivo studies using animal models will be essential to evaluate their safety, bioavailability, and therapeutic potential in living systems. Exploring their use in combination therapies with existing chemotherapeutics and assessing their long-term safety profile in humans are also important directions.
Additionally, investigating the pharmacokinetics and bioavailability of steviol glycosides will shed light on how they are absorbed, metabolized, and excreted. Research should also aim to identify specific molecular targets and pathways affected by steviol glycosides, while exploring potential synergistic effects with other compounds. Utilizing patient-derived xenograft models can provide more accurate insights into their efficacy in human-like tumors. Ultimately, clinical trials will be necessary to validate preclinical findings and establish evidence-based guidelines for their use. Public awareness efforts will also play a role in educating healthcare providers and patients about the potential benefits and risks of steviol glycosides as anticancer agents.
While these findings highlight the therapeutic potential of steviol glycoside, critical gaps remain in translating these in vitro findings to clinical applications. Key challenges include defining dose-response relationships, assessing interactions with concurrent therapies, and validating safety and efficacy through systematic preclinical and clinical trials. Acknowledging these limitations strengthens the study’s transparency and underscores the need for rigorous follow-up research to advance toward human trials.