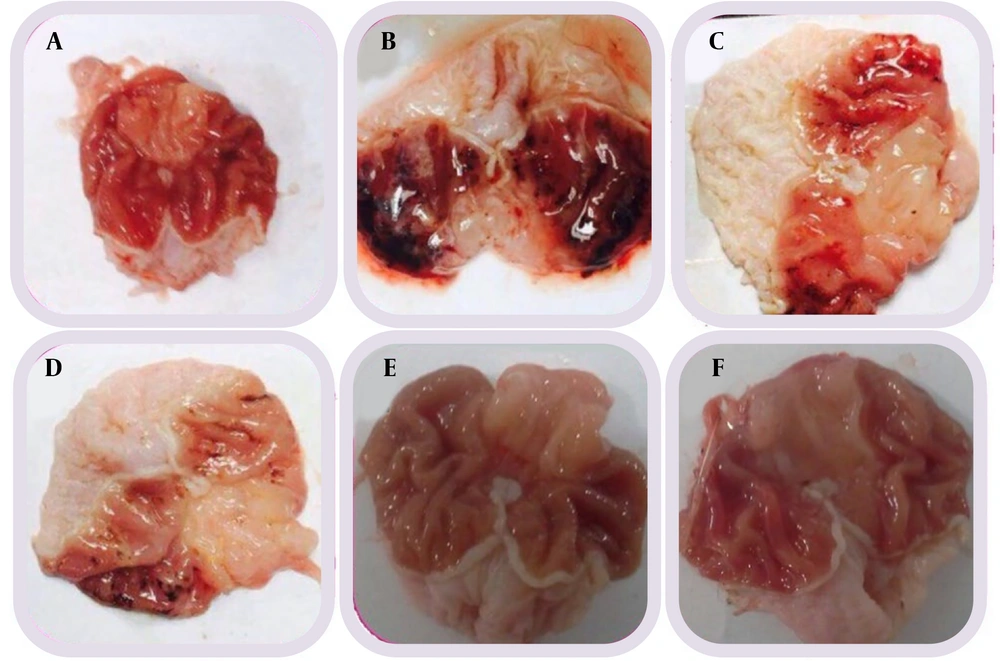
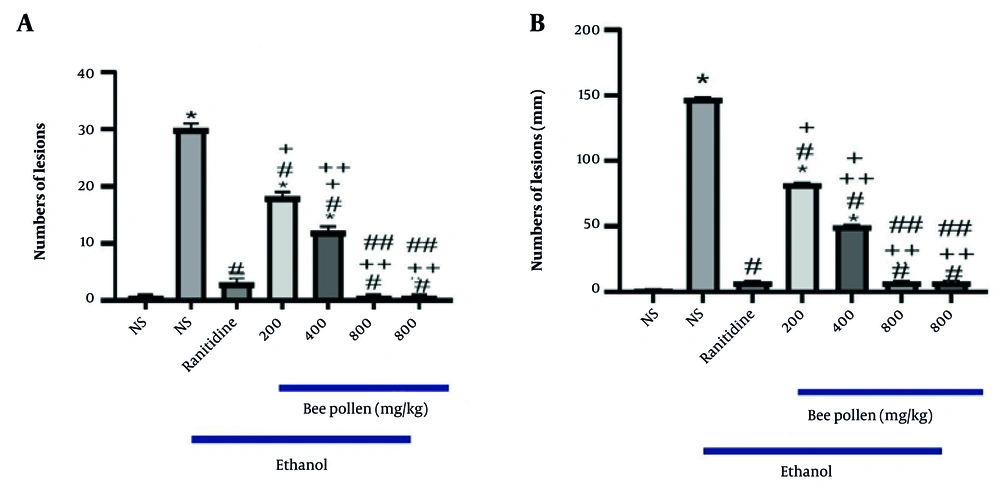
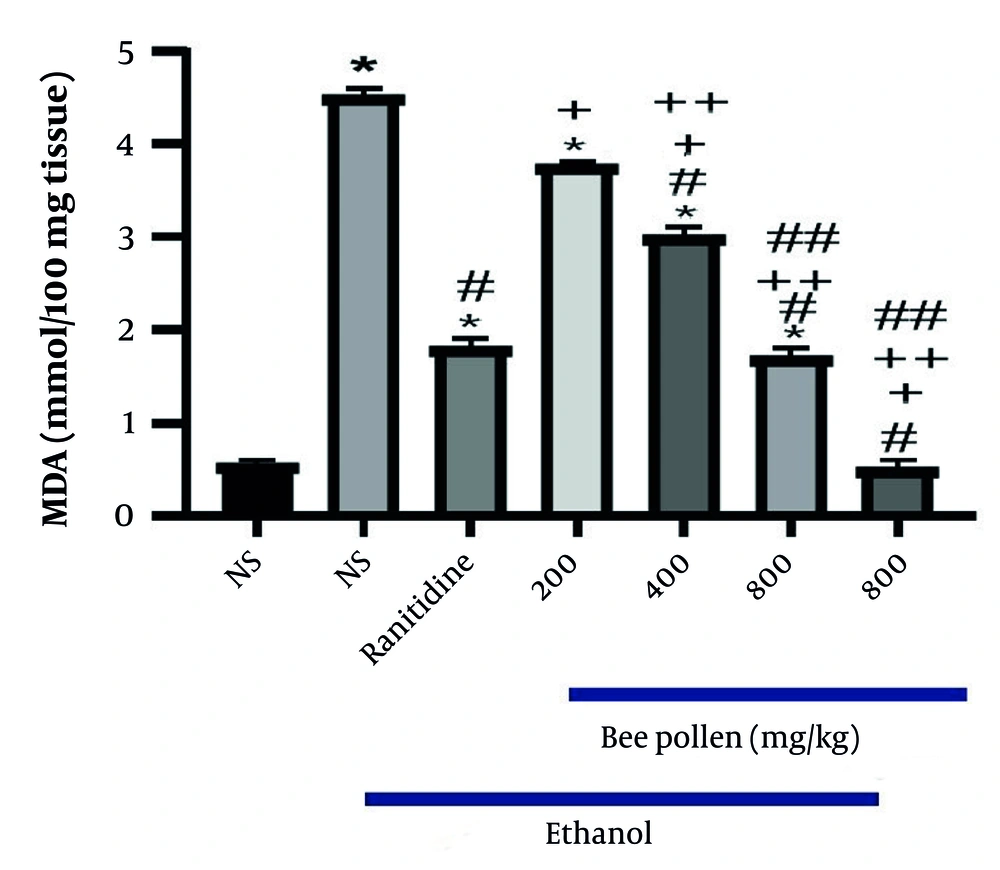
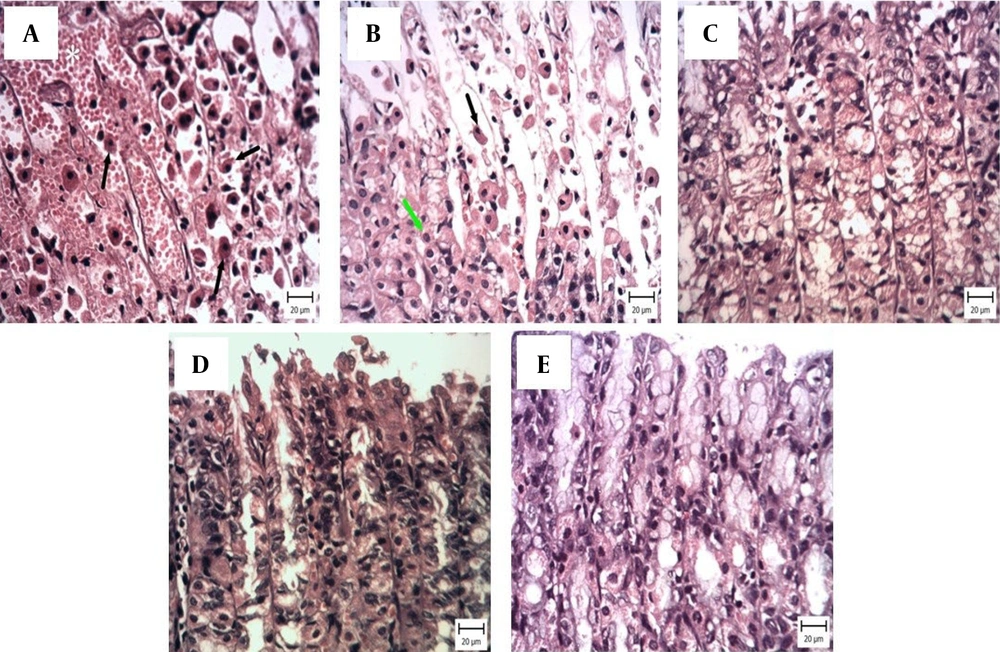
Gastrointestinal diseases, including gastric ulcers, are prevalent and have been the subject of numerous studies. Many investigations have focused on methods to prevent gastric ulcers (
17). Ethanol-induced gastric ulcers are one of the peptic ulcer models widely used to examine the gastroprotective properties of various substances in laboratory settings (
18). The pathogenesis of ethanol-induced gastric ulcers is multifactorial, involving factors such as necrosis of surface cells and the production of superoxide anions and free radicals through ethanol metabolism in the body. These mediators lead to reduced microvascular blood flow to the gastric mucosa and ultimately result in mucosal damage. Additionally, ethanol disrupts the integrity of the gastric mucosa by interfering with the production of gastric mucosal surfactant (
19). Ethanol can reduce the secretion of bicarbonate and gastric mucus. Furthermore, ethanol decreases mucosal blood flow, induces oxidative stress, increases the production of xanthine oxidase and malondialdehyde, and decreases glutathione production (
20).
Various studies have been conducted to identify new gastroprotective agents against oxidative inflammatory conditions induced by ethanol in the stomach (
15). Thus, attention to natural substances aims to achieve drugs with minimal side effects. The current study aimed to investigate the therapeutic effects of Bee Pollen on ethanol-induced gastric ulcers in rats. Bee Pollen is essentially a collection of pollen particles bound together by nectar and the saliva of honeybees (
21). Additionally, Bee Pollen is an abundant source of hydrophilic antioxidants that safeguard against oxidative stress in the cytoplasm, intracellular organelles, and extracellular fluid. Notable among these hydrophilic antioxidants are compounds like ascorbate (vitamin C) and phenolic acids, which have been isolated (
22).
Flavonoids comprise a large group of substances containing 5,000 different compounds with a wide range of biological effects. Various mechanisms for their gastric healing effects have been proposed, including increased prostaglandin levels, increased gastric mucus, reduced histamine secretion from mast cells by inhibiting the enzyme histidine decarboxylase, scavenging free radicals, and inhibiting the growth of Helicobacter pylori (
23). Vitamin C derived from Bee Pollen can reduce reactive oxygen species. It has also been shown that Bee Pollen can produce a suitable level of nitric oxide, which can relax arteriolar smooth muscles. The relaxation of arteriolar smooth muscles is one of the most important factors responsible for regulating gastric mucosal blood flow (
24). This may explain the protective effects of HBP on the histopathological characteristics of ethanol-induced gastric ulcers.
Although HBP exhibits protective tissue effects, the molecular and cellular mechanisms behind these effects remain incompletely understood. However, it is likely that Bee Pollen alleviates inflammatory and oxidative conditions and releases mediators such as nitric oxide, leading to vascular vasodilation and increased blood supply to the damaged area, thereby accelerating gastric ulcer healing (
22). The present study demonstrated that HBP can significantly reduce the number of ulcers, ulcer length, and MDA levels as a primary marker of lipid peroxidation in the stomach of rats in a dose-dependent manner.
The results of this study align with research conducted by other investigators. For example, Eraslan et al. in 2008 demonstrated that Bee Pollen acts as an anti-lipoprotein peroxidant, with the bioflavonoids present in Bee Pollen inhibiting free radicals, leading to a reduction in MDA levels and an increase in antioxidant enzyme levels (
25). Sarić et al. in 2009 examined the antioxidant and anti-estrogenic effects of flavonoids in Bee Pollen in mice. The results showed that Bee Pollen is a vast source of antioxidants and has high potential for protecting the body and maintaining the health of various organs (
26). Jue in 1989 studied the anti-aging effects of Bee Pollen compounds. In this study, for two months, 10 grams of pollen were given daily to old mice, and parameters such as liver and heart tissue health and lipid levels were examined, finding that Bee Pollen reduced tissue degradation and aging (
27). Leja et al. in 2007 investigated the antioxidant properties in 12 species of Bee Pollen and found that all species contained significant amounts of phenolic compounds with antioxidant properties, revealing that daily intake of Bee Pollen in these mice compared to the control group could have a significant impact on maintaining the health of various organs (
28).
In the present study, the effects of doses of 200 mg/kg, 400 mg/kg, and 800 mg/kg of HBP on ethanol-induced gastric ulcers in rats were examined. Factors such as the number of ulcers, ulcer length, and MDA levels were evaluated. The results and pathological and laboratory data assessments indicated that the protective effect of this extract is likely due to the elimination of free radicals and the natural protection of cells. The dose of 800 mg/kg of HBP was selected as the optimal dose for controlling various factors.
5.1. Conclusions
The present study showed that HBP has a therapeutic effect on ethanol-induced gastric ulcers. HBP can significantly reduce the elevated levels of MDA caused by ethanol. Notably, all tissue protective effects and antioxidant properties of Bee Pollen are dose-dependent. The results suggest that HBP may serve as an adjunct or primary treatment for gastric ulcer prevention, pending further studies on various animal models and adherence to existing regulations and guidelines for human trials.