1. Background
Allicin (diallyl thiosulfinate) as a major bioactive molecule of garlic consists more than 70% of organosulfur compounds found in garlic extract. Allicin has been reported to possess various biological properties. It exhibits antimicrobial, antithrombotic, anticancer, anti-platelet aggregation and antioxidant activities (1). In addition, it has been shown that allicin neutralizes microbial virulence factors (2, 3). Due to its high pharmacological values, allicin is considered to be a drug candidate for the treatment of several pathological conditions.
Commercial availability of allicin is limited because of its instability. Researchers generally provide the substance by synthesis or isolation from garlic extract in their laboratories. Several laboratories described different techniques to extract allicin. Allicin can be chemically synthesized by oxidation of the analogous polysulfides (4) or purified from garlic extract (5, 6). However applying abovementioned techniques requires the use of laborious synthesis and purification procedures.
2. Objectives
In this report, we described a simple chromatographic technique for semi-preparative purification of allicin from garlic extract.
3. Materials and Methods
3.1. Materials
Reagent grade standard allicin with a purity of 99.39% was purchased from LKT laboratories Inc. (St Paul, MN, USA) and stored at -80°C until analysis. HPLC grade methanol, ethylparaben (99% purity) and diethyl ether were obtained from Merck Company (Darmstadt, Germany).
3.2. Instrument (HPLC System)
HPLC procedures were performed on a Jasco liquid chromatography system (Jasco Co, Tokyo, Japan), equipped with a HSS1500 system control, a model PU-1580 pump, a model UV-1575 UV/VIS absorbance detector, a model 7725i Rheodyne manual injector and a model Jasco–Borwin data processing software (JMBS Developments, Grenoble, France). A 20 µL and 1 mL loops were used for analytical and semi-preparative HPLC procedures, respectively.
3.3. Semi-Preparative HPLC Purification Procedure
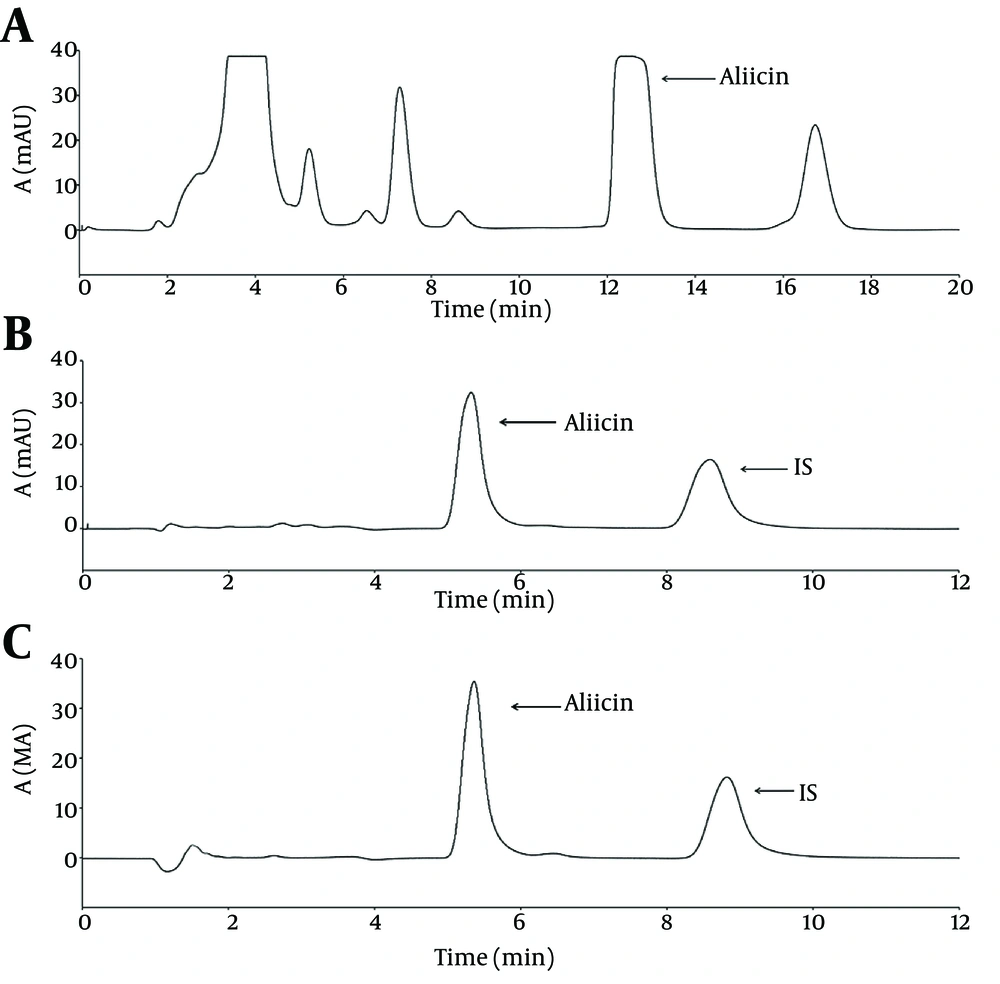
Garlic extract was prepared as previously reported (7). The proteins were separated using methanol (50:50, v/v) and supernatant was filtered through a 0.22 µm membrane. A 0.5-mL solution of garlic extract was injected onto HPLC system equipped with a C18, Nucleosil 100 ODS (5 µm) semi-preparative column with a dimensions of 150 mm × 10 mm (Teknokroma S. Coop. C. Ltda., Barcelona, Spain). The column was eluted with methanol–water (50:50, v/v). The chromatographic analyses were performed at ambient temperature at a flow rate of 2 mL/min. The allicin in the effluents was monitored at 220 nm. The fractions containing allicin was collected into 50 mL falcon tubes and stored at -80°C until use.
3.4. Concentrating of Allicin
To concentrate allicin, collected fractions were pooled. Equal volume of non-polar solvent “diethyl ether” was added and the mixture was poured into a separating funnel; the funnel was shaken vigorously and left to stand for 15 minutes, until two distinct phases appeared. The organic phase was collected. Appropriate amount of double-distilled water was added and then the organic phase was evaporated under reduced pressure using a rotary evaporator at 33°C (8). The allicin solution was stored at -80°C for further analysis. The concentration of allicin was determined by analytical HPLC. The percent yield of allicin was calculated as follows:
.
3.5. Quantitative Analysis of Allicin by Analytical HPLC
The content of purified allicin was quantitatively determined by high performance liquid chromatography (HPLC) as reported by authors (7). Briefly, 10 µL of purified allicin was dissolved in 15 µg/mL Ethylparaben solution. Then, 20 µL of prepared solution was injected onto HPLC system (Jasco, Tokyo, Japan) equipped with a C18, Nucleosil 100 ODS (5 µm) analytical column with a dimensions of 4.6 mm × 150 mm (Alltech Grom GmbH, Rottenburg, Germany). The column was eluted with methanol/water (50:50, v/v) at flow rate of 1 mL/min. Allicin was detected in the effluents at 220 nm and quantified by comparing the peak area to standard allicin.
3.6. Biological Activity Assay
Biological activity of purified allicin was assayed using its antibacterial property in comparison with the standard allicin. For this purpose, a Gram positive Staphylococcus aureus strain ATCC 25923 was used. The bacteria were streaked on nutrient agar plate and incubated for 18 hours at 37°C. Then, 3 - 5 colonies were picked up and diluted in sterile normal saline to turbidity comparable to that of a 0.5 McFarland turbidity standard. The surface of a Mueller Hinton agar plate (4 cm diameter) was cultured uniformly with a sterilized cotton swab. Filter paper disks placed on the plate and a 30 µL solution containing 45, 65 and 95 µg amounts of isolated allicin and standard allicin was put on the disks. Plates were incubated 24 hours at 37°C. The biological activity was determined as the diameter of growth inhibition halo. In parallel, the same experiment was performed using standard allicin.
4. Results
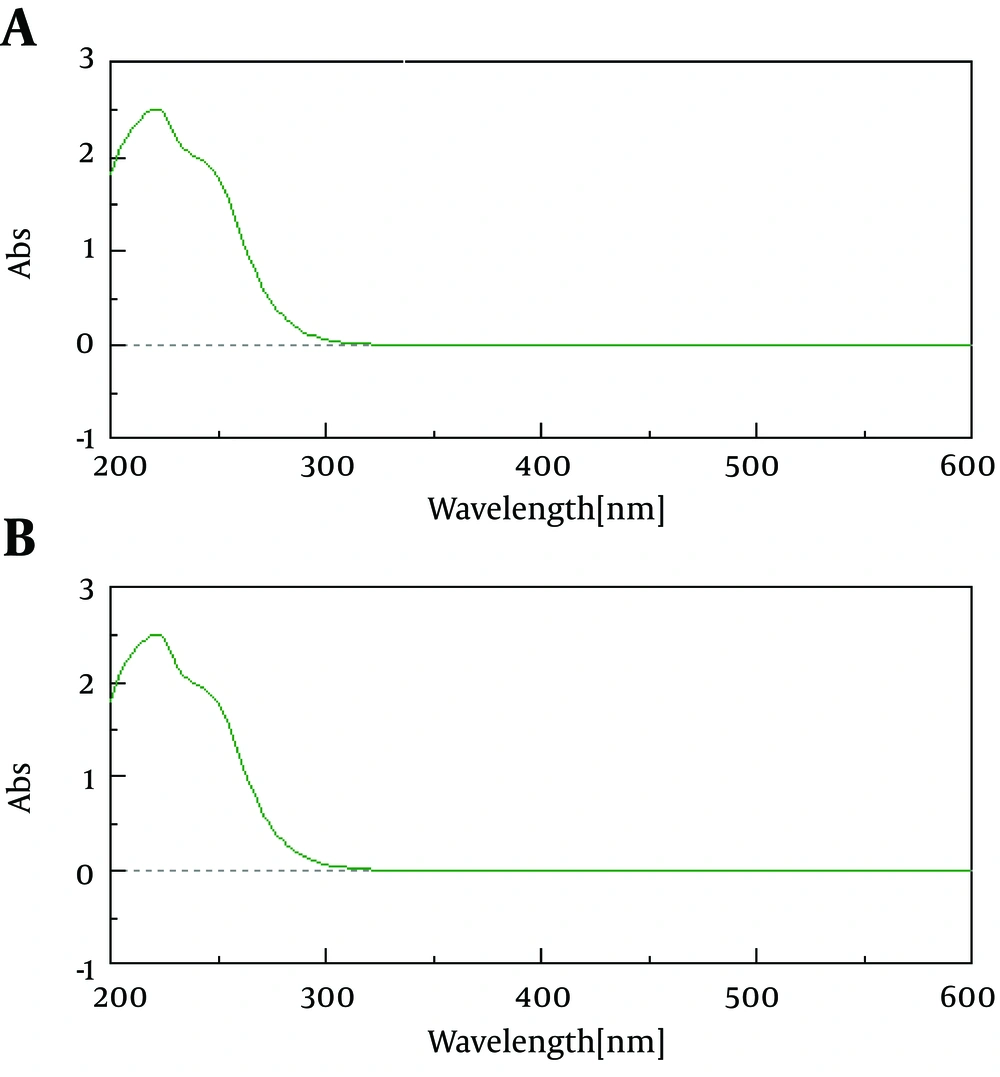
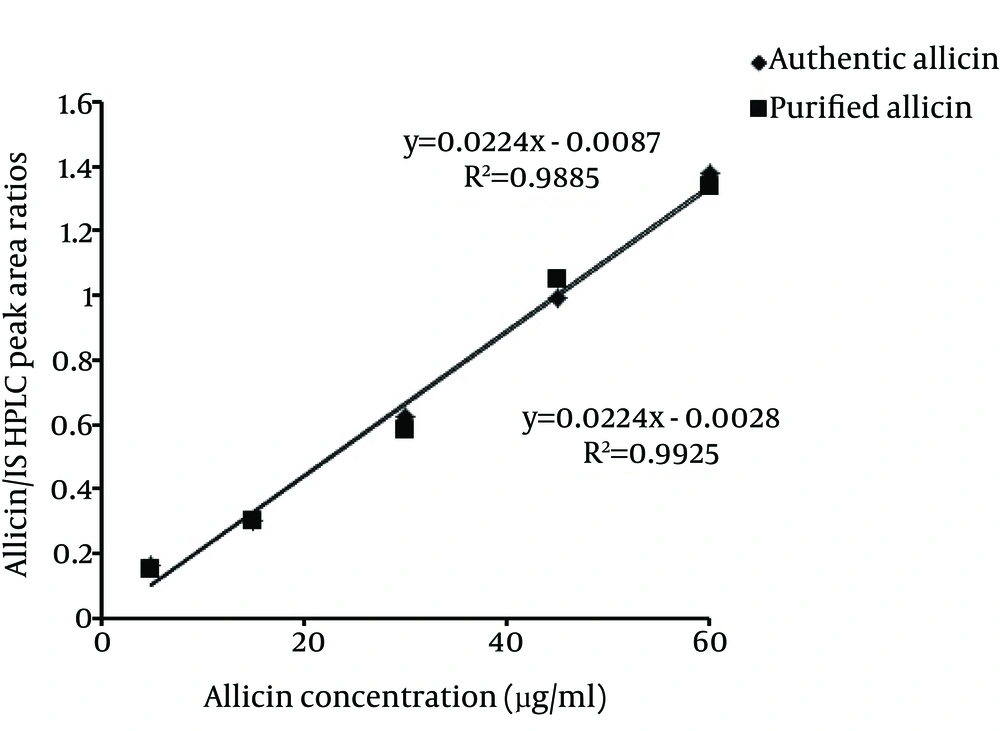
Typical representative chromatogram of garlic methanol extract is shown in Figure 1 A. As shown in the chromatogram, semi-preparative HPLC yielded a distinct peak with a retention time of 12.28 minute Results revealed peak corresponding to allicin was separated by a return to baseline and there were no interfering peaks between allicin and other components in the extract. Analytical HPLC analyses showed collected fraction yield a single peak with a retention time of 5.22 minutes, which corresponded to standard allicin and constituted ≥ 95% of the total detectable peak areas (Figure 1 B, C). Absorbance spectrum (200 - 600 nm) analyses of purified allicin peak revealed similar pattern as that for standard allicin (Figure 2). The purity of prepared allicin was compared to a commercially standard allicin. Purified allicin gave a peak area comparable to standard allicin at several concentrations (Figure 3).
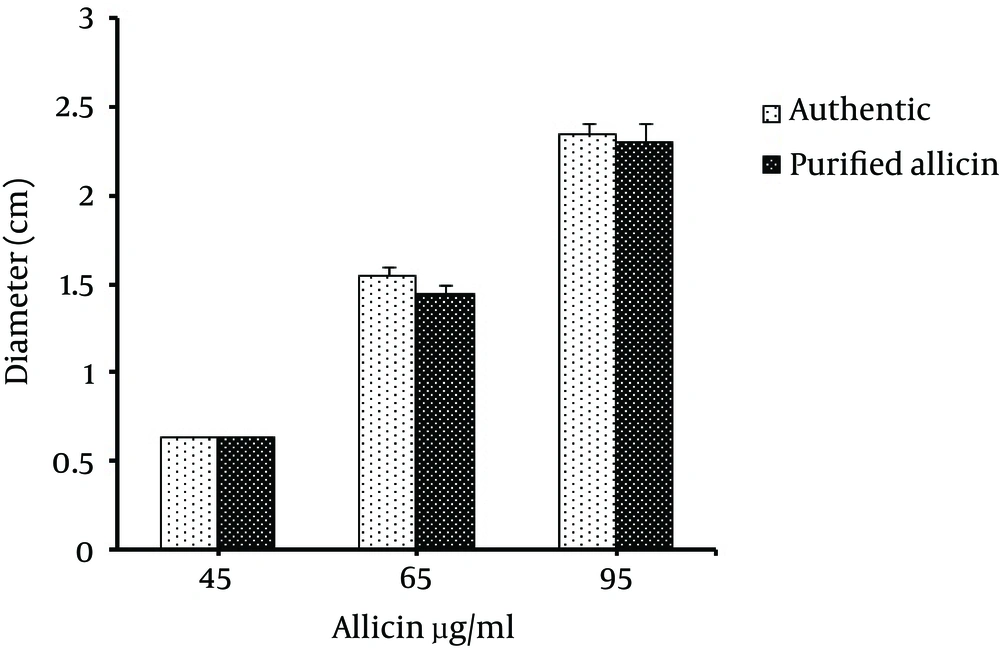
Biological activity of purified allicin was tested using its antibacterial activity against Staphylococcus aureus. Results showed that purified allicin gave an inhibition zone size comparable to standard allicin at different concentrations (P < 0.05) (Figure 4).
Purification method yielded highly purified allicin. Dried garlic bulb, garlic extract and purified solution composed 3.2% (1 fold), 12% (~8 fold) and 95% (~ 30 fold) of allicin, respectively for weight to weight ratio. The percent yield of allicin in peak collection and concentrating steps were 100% and 73.34 ± 2%, respectively.
5. Discussion
Several laboratories described different methods for purification or synthesis of allicin. Usually, the methods used for purification of allicin from garlic extract use relatively high temperature for extraction (5). It has been previously shown that allicin is not stable in high temperatures and generates numerous sulfur-containing compounds (4, 9). This lowers the yield and purity of the production. These types of methods need further purification process to produce highly purified allicin. In the synthetic methods, allicin is synthesized by oxidation of allyl sulfiede as precursor using agents such as hydrogen peroxide (10), m-chloroperbenzoic acid (11) and magnesium monoperoxy hydrate (12). These methods also need further purification process to obtain pure allicin. Bat-Chen et al. (8) developed a simple method for direct extraction of allicin from garlic extract. In another study, Lee et al. developed a recycling preparative HPLC method for separation and purification of thiosulfinates in garlic extract (13). The purification method we developed yields high purity allicin from a single HPLC fractionation. The purity and biological activity of purified allicin were the same as the commercially available standard allicin based on antibacterial activity and HPLC assays, respectively. Produced allicin, inhibited Staphylococcus aureus growth in a dose-dependent manner and gave the peak area comparable to standard allicin at several concentrations. These results indicated that the purification methodology yielded highly purified allicin that is well suited for research purposes.
The yield of developed method was more than 70% in all purification steps. The higher yield in current procedure is due to using mild conditions during purification such as low temperature.
In conclusion, the results of this study demonstrated that our method could be feasibly used for purification of large quantities of allicin. The yield of this method can be further improved using the extracts from the garlic varieties with high levels of allicin or concentrating the starting material by removing the methanol fraction.