1. Background
Inflammation is the first response of the immune system to infection or irritation that helps the body to protect itself against an injurious stimulus. It can be evoked by a variety of noxious agents, (e.g. infections, antibodies or physical injuries). Inflammation may be acute or chronic, depending on the nature of the stimulus and the effectiveness of the initial reaction in eliminating the stimulus or the damaged tissues. Acute inflammation is rapid in onset (typically minutes) and is of short duration, chronic inflammation may follow acute inflammation or be insidious in onset. It is of longer duration and is associated with the presence of lymphocytes, macrophages, proliferation of blood vessels, fibrosis, and tissue destruction (1, 2). Nonsteroidal Anti-inflammatory Drugs (NSAIDs), opioids and corticosteroids are the group of drugs, which are used presently for management of pain and inflammatory conditions. The adverse drug reactions associated with NSAIDs are the gastrointestinal problems due to direct and indirect irritation of the gastrointestinal tract, the most common being gastrointestinal bleeding and peptic ulcers. Therefore, despite the currently available anti-inflammatory drugs, there is a need for safer alternatives. The use of natural remedies for the treatment of inflammatory and painful conditions has a long history in Ayurveda and other systems of traditional medicine (3-5).
Royal Jelly (RJ), a honey bee secretion used in the nutrition of larvae as well as adult queens, is secreted from the glands in the hypopharynx of worker bees and fed to all larvae in the colony (6). It is collected and sold as a dietary supplement, claiming to have various health benefits because of components like B-complex vitamins, such as pantothenic acid (vitamin B5) and vitamin B6 (pyridoxine) (7, 8).
Royal jelly has been reported as a possible immunomodulatory agent in Graves’ disease. It has also been reported to stimulate the growth of glial cells and neural stem cells in the brain (8). Preliminary evidences show that it has cholesterol-lowering, anti-inflammatory, wound-healing, and antibiotic effects. Researches also suggest that the 10-Hydroxy-2-Decenoic Acid (10-HDA) found in RJ may inhibit the vascularization of tumors (7, 9). Royal jelly is said to improve fertility in both men and women, in men by increasing the quality of their sperm, and in women by increasing the quality of their ovules (10, 11). It is also found to have antifungal activity against Candida spp. and Trichosporon spp. (8).
Studies suggest the hepatoprotective and antioxidant activities of the RJ against carbon tetrachloride (CCl4)- induced acute liver injury (12). Earlier biological investigations have shown that RJ has insulin-like activity and was found to be responsible for the lowering impact of honey on blood glucose levels (13).
2. Objectives
In the present study, the anti-inflammatory properties of RJ were investigated in formalin-induced rat paw edema.
3. Materials and Methods
3.1. Drugs
The RJ used in this study was purchased from Santa Cruz Biotechnology Inc, USA. Aspirin and formalin were obtained from Darou-pakhsh (Iran) and Merck (Germany), respectively.
3.2. Animals
Albino Wistar male rats (weighing between 150 - 200 g) were purchased from animal house of Ahvaz Jundishapur University of Medical Sciences. The animals were maintained under normal laboratory condition of humidity (50%), temperature (23 ± 2°C) and a 12-hour light/dark cycle, and allowed free access to food and water ad libithum. The animals were randomly allocated into 5 groups (n = 6). At first, the volume of intact rats paw was measured in all groups using a plethysmometer. The first group served as a negative control (normal saline 5 mL/kg i.p.) while the second, third and the forth groups received different doses (25, 50 and 100 mg/kg) of RJ and the fifth group received aspirin (300 mg/kg i.p.) as a positive control group, respectively. Edema was induced on the right hind paw of the rat by a subplantar injection of 0.1 mL of formalin (2.5 %) 30 minutes after drug administration. Swelling of formalin-injected foot was measured using a plethysmometer (Ugo Basile, Italy) hourly during the first and fifth hours after the formalin injection (14). The ability of anti-inflammatory agents to suppress paw inflammation was expressed as a percent inhibition of paw edema and calculated according to the following equation:

Where v1 = The animal paw volume before formalin injection and v2 = The paw volume after drugs and formalin injection at different time points.
3.3. Statistical Analysis
Data were shown as mean ± SD of different groups. The Data of this study were statistically analyzed using one-way ANOVA and Tukey tests. The P < 0.05 was considered statistically significant.
4. Results
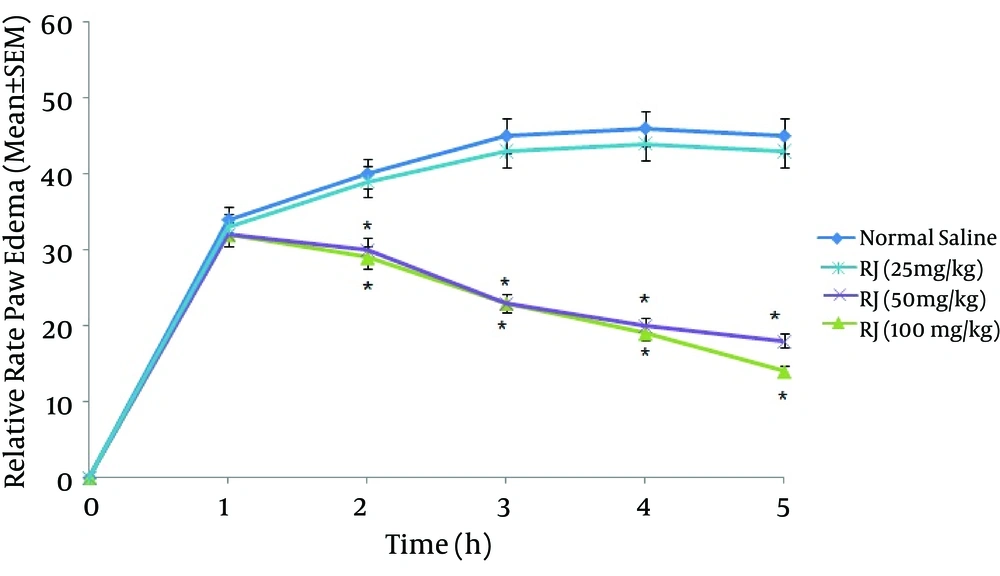
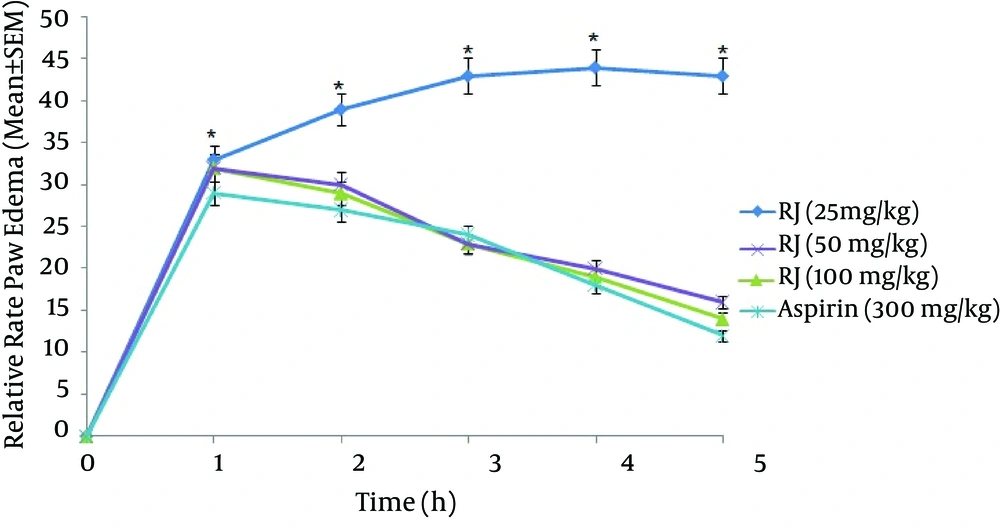
Royal jelly demonstrated a dose-dependent relationship in inhibiting formalin-induced inflammation. As shown in Figure 1 the RJ (50 and 100 mg/kg) significantly (P < 0.05) inhibited formalin-induced rat paw edema as compared to the control group. Also, it was shown that there was no significant difference between 3rd and 4th groups (50 and 100 mg/ kg) and the positive control group (aspirin 300 mg/kg) in inhibiting formalin-induced swelling (P > 0.05, Figure 2). Moreover, 25 mg/kg of RJ could not diminish paw swelling in response to formalin. Royal jelly at doses 50 and 100 mg/kg showed comparable effects to aspirin (300 mg/kg) to inhibit paw edema up to 60%.
5. Discussion
Tissue damage and injury are always associated with pain and inflammation. Formalin test is a biphasic response where first phase is the direct effect of formalin, which involves neurogenic pain. The second phase is involved in the inflammatory reactions mediated by prostaglandin, serotonin, histamine, bradikinin and cytokines, such as interleukin-1 beta, interleukin-6 tumor necrosis factor-alpha eicosanoids, and Nitric Oxide (15). In the present study, the formalin-induced paw edema model was used to investigate anti-inflammatory effects of RJ in experimental rats. The results demonstrated that anti-inflammatory effects of RJ was dose-dependent and the best anti-inflammatory effect was observed in the doses of 50 and 100 mg/kg. It was shown that 50 mg/kg RJ was as efficient as 300 mg/kg of aspirin in attenuating formalin-induced edema. Aspirin, a nonsteroidal anti-inflammatory agent, has been frequently used in experimental models as positive control to suppress inflammation. The results of the recent study confirm the findings of Karaca et al. which showed RJ inhibits colitis induced by acetic acid (16).
This effect can be attributed to the polyphenolic compounds in RJ that are responsible for the functional properties, such as antioxidant and anti-inflammatory capacity as showed by Kerem et al. (17), Almaraz-Abarca et al. (18), and Wu et al. (19). These compounds in RJ are collected by bees from the plants where they gather nectar (20). Royal jelly has been shown to exhibit DNA-protective and antitumor effects in experimental animals (21, 22).
Further mechanisms for protective effects of RJ have been proposed. Hamerlinck reported that RJ exhibited immunomodulatory properties by stimulating antibody production and immunocompetent cell proliferation in mice or by depressing humoral immune functions in rats (23). Another report indicated that the protection by RJ ingestion could be partly attributed to the presence of neopterin in RJ (23). The protection afforded by RJ against the immunosuppression produced by increased prostaglandin E2 levels has been demonstrated in studies in vivo and in vitro (21-24). El-Nekeety et al. found a potential protective effect of RJ against the toxic hazards of fumonisins (25). Kamakura et al. reported that RJ in the diet decreased the gene expression of Cytochrome P450 4A14 (CYP4A14) enzymes and detoxifying enzymes, which catalyse peroxidation of endogenous lipids, and increased the gene expression of glutathione S-transferase and glutathione peroxidase (26).
As a result, RJ was found to diminish the formalin-induced paw edema in a dose-dependent manner. Although it has been considered that it might exert the benefits through its antioxidant features, the exact mechanism of effect is yet unknown. In conclusion, it could be suggested that RJ provides considerable anti-inflammatory effects and the efforts to focus on the underlying mechanism/s of RJ may provide new opportunities for the development of new drugs.