1. Background
Chitosan (CS) is a natural biopolymer obtained by alkaline deacetylation of natural chitin, which is found in the shells of marine crustaceans. It is a high molecular weight polysaccharide containing β-1, 4 glycoside links and is composed of N-acetyl-glucosamine and glucosamine. This cationic biopolymer is remarkably used in pharmaceutical applications due to its unique properties such as non-toxicity, biocompatibility and biodegradability (1, 2). Hydrophilic nanoparticles (NPs) of CS have advantages since they can be used for the purposes of controlled drug release, therapeutic effect enhancement and targeted drug delivery (3). Since CS nanoparticles are positively charged and due to their mucoadhesive properties, they are widely used in colon and mucosal delivery, cancer therapy and target drug delivery (4). Diclofenac is a non-steroidal anti-inflammatory drug (NSAID) that is used via different routes such as oral, rectal, parenteral, topical and ocular administration (5). Diclofenac-loaded chitosan nanoparticles are expected to be potential carriers for ophthalmic drug delivery.
One of the most important problems of diclofenac ocular solutions, similar to other ophthalmic drops, are their short ocular residence time. Chitosan can enhance cornea contact time through its mucoadhesion and its ability to transiently open tight junctions (6). The later property and their ability to transport across gastrointestinal epithelial through adsorptive endocytosis as well as the capacity of CS nanoparticles to protect gastric mucosa from immediate contact of drugs (7, 8) makes this system a good candidate for diclofenac oral delivery. Among the many methods suggested for CS nanoparticles preparation, ionotropic gelation (polyelectrolyte complexation) has attracted much attention. This technique is a simple and mild preparation method in the aqueous environment (9). Chitosan NP formation is based on electrostatic interaction between protonated amine group of chitosan and negatively charged group of a polyanion (such as TPP) or an anionic polymer (10-12).
Previous studies demonstrated that formation of nanoparticles is only possible within specific and moderate concentrations of CS and TPP (13) therefore, it was suggested to investigate the appropriate CS/TPP ratio.
2. Objectives
The aim of the present study was to prepare and evaluate diclofenac-loaded chitosan nanoparticles based on the ionotropic gelation method, and investigate the optimum conditions in which the system shows maximum drug loading, minimum size and size distribution, and appropriate in vitro release profile to form an advantageous drug delivery system.
3. Materials and Methods
Low molecular weight chitosan (CS) was purchased from Primex, Iceland (deacetylation degree 95% and viscosity < 25 cps), which was obtained from fresh north Atlantic shrimp (Pandalus borealis) shells. Sodium tripolyphosphate (TPP) and Sodium diclofenac were supplied by Merck chemicals (Germany). Transcutol P was obtained from GatteFosse (France). All other materials and reagents were of the highest grade commercially available.
3.1. Preparation of Chitosan Nanoparticles
Chitosan nanoparticles were prepared on the basis of the ion-gelation method (10, 11). At the initial step, defined concentrations of TPP aqueous solution was prepared and added drop by drop to CS solution, which was prepared in 1% acetic acid (v/v); the solution was stirred at 400 - 600 rpm. Different CS/TPP ratios were prepared (1:1, 2:1, 3:1, 4:1, 5:1 and 10:1) to determine an appropriate formulation with optimum properties. Particle size distribution of the prepared colloidal suspensions was determined by the light scattering method using a particle-size analyzer (Malvern, Master sizer 2000, UK). Moreover, the effect of homogenization (at 20000 rpm for five minutes) and sonication (at 60 W) on mean particle size was evaluated.
3.2. Evaluation of pH Effect on Mean Particle Size
Different concentrations of CS solution (1, 2, 4 and 10 mg/mL) in 1% acetic acid (v/v) were prepared and defined amounts of TPP aqueous solution with a concentration of 1 mg/ml were added drop by drop to CS solutions, and stirred with a magnet stirrer at 400 - 600 rpm and room temperature to obtain a volume ratio of 4:1 CS/TPP. Next, a five-fold volume of phosphate buffer (pH 5.9, 7.4 or 8.4) was added drop by drop to the CS/TPP mixtures. Larger particles were removed by centrifugation (Hettich, EBA20, Germany) at 12000 rpm and 25°C for a 30 minutes period. The supernatant (containing CS/TPP nanoparticles) was sonicated at 100 W for five minutes in ultrasonic bath (ELMA, T710DH, and Germany). During the final step, the particle size of the samples was measured (14).
3.3. Preparation of Diclofenac-Loaded Chitosan Nanoparticles
A defined content of sodium diclofenac was dissolved in 1% Transcutol (as a solubilizing agent), and added to 0.1% CS solution in 1% acetic acid (v/v). In the next step, a certain volume of TPP aqueous solution with a concentration of 0.1% was added drop by drop to CS-drug mixture to obtain a volume ratio of 4:1 CS/TPP with regular stirring at 400 - 600 rpm and room temperature. The above mixture was homogenized at 20000 rpm for five minutes and sonicated at 60 W and 25°C for five minutes. It should be noted that defined amounts of drug concentration was added to CS solution to provide certain CS/Drug proportions (10/1, 5/1 and 4/1). Each sample was prepared in triplicate and evaluated.
3.4. Diclofenac-Loaded Chitosan Nanoparticles Characterizations
3.4.1. Size Determination of Nanoparticles
The sizes of diclofenac-loaded CS nanoparticles were determined using a particle size analyzer (Malvern, Mastersize 2000, UK). Particle size distribution is reported by a polydispersity index (PDI). Colloidal suspensions containing nanoparticles were poured in the nanosizer cell and placed in the analyzing chamber. Experiments were carried out at 25°C with detected angel of 90 degrees.
3.4.2. Nanoparticles Surface Charge Measurement
Zeta potential was measured by the zetasizer (Malvern, Zen3600, UK). For this purpose, the colloidal suspensions were diluted with distilled water and placed in the zetasizer chamber.
3.4.3. Drug Entrapment Evaluation
The prepared colloidal suspension containing diclofenac-loaded CS nanoparticles were centrifuged by application of the ultracentrifuge instrument (Sigma, 3K30, Germany) at 20000 rpm and 4°C for 20 minutes while drug loading was determined indirectly so that the amount of non-entrapped drug was determined in the supernatant solution using a UV spectrophotometer (Biochrom, WPA, UK) at 276 nm. Calibration curves were prepared using the supernatant of blank nanoparticles. Loading efficiency (LE) parameter was calculated according to the Equation 1 (15):
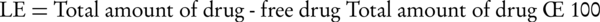
The precipitated gel was lyophilized at -55°C for 72 hours under vacuum using the freeze drier instrument (Criss, α2-4, Germany) and was used for calculating the loading capacity (LC) using the Equation 2 (15):
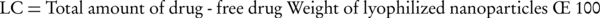
This parameter was used to determine drug release percentage.
3.4.4. Differential Scanning Calorimetry (DSC)
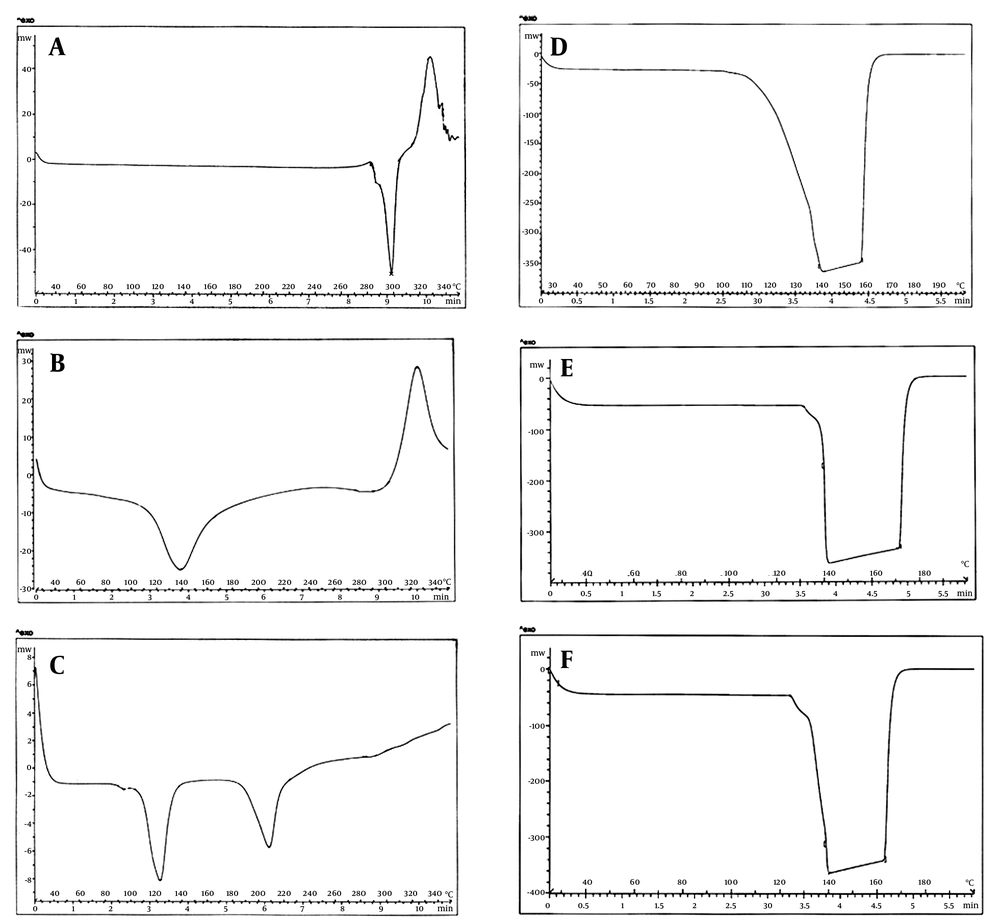
Differential scanning calorimetry evaluation was carried out using the DSC instrument (Mettler Tolledo, DSC1, Switzerland). Thermograms of sodium diclofenac, CS, TPP, and F1, F2, and F3 formulations (freeze dried) were recorded. Thermal behavior was studied by accurately weighed nanoparticle samples (10 - 20 mg), which were sealed individually in standard aluminum pans. The investigations were performed over a temperature range of 25 - 350°C, with heating rate of 10°C/minute (16).
3.4.5. Drug Release Studies
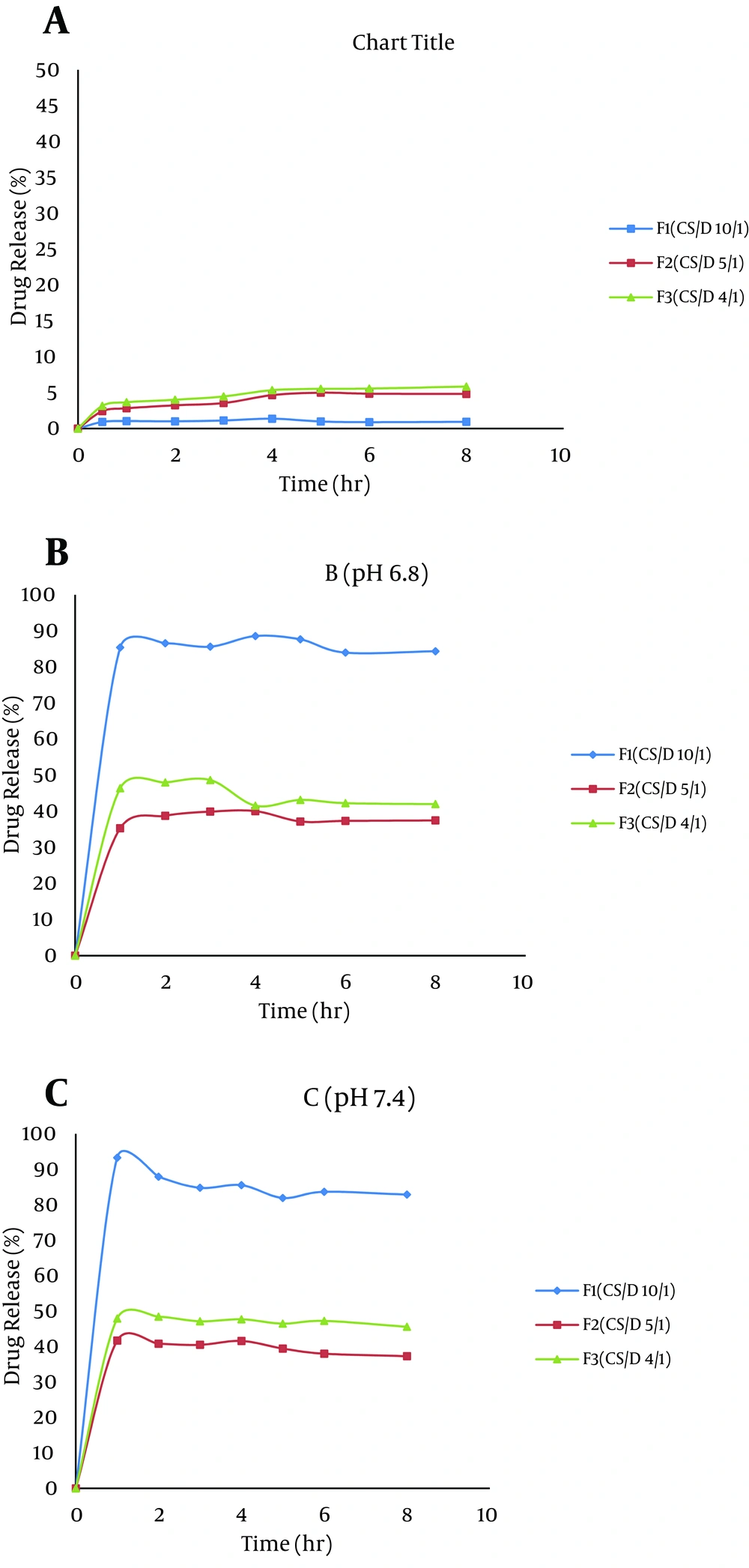
Drug release studies of freeze dried diclofenac-loaded CS nanoparticles were performed by a modified USP (United State Pharmacopeia) dissolution apparatus II (ERWEKA, DT-800, Germany) containing 30 mL of medium, preheated and maintained at 37 ± 0.5°C and a paddle stirrer set at 50 rpm. Different pH values including 1.2 and 6.8, to simulate stomach and intestine fluids, and 7.4, to simulate tears, were used as dissolution media. At predetermined time intervals, a sample was withdrawn and centrifuged. The content of diclofenac in the supernatant was determined by a UV spectrophotometer (Biochrom, WPA, UK) at a wavelength of 276 nm.
3.5. Statistical Analysis
In the present study, all experiments were carried out in triplicates and the results were expressed as mean ± standard deviations (SD). Statistical analysis of data was performed using one-way ANOVA and followed by Tukey’s test.
4. Results
4.1. Chitosan Nanoparticles Characterizations
The prepared different ratios of CS/TPP colloidal suspensions were checked in appearance. The suspensions with CS/TPP ratios 1:1, 2:1 and 3:1 showed undesirable turbidity. Among the other formulations, CS/TPP 4:1 nanoparticles showed the lowest mean particle size. The effect of homogenization and sonication on mean particle size of chitosan NP is shown in Table 1. The results demonstrate that these two processes significantly decreased mean particle size (P < 0.05).
| CS/TPP Ratio | Non-Homogenized Particle Size, nm | Homogenized and Sonicated Particle Size, nm |
|---|---|---|
| 10:1 | 205 ± 13 | 152.6 ± 7.6 |
| 5:1 | 99 ± 2.29 | 83.3 ± 9.45 |
| 4:1 | 78.4 ± 5.19 | 36.9 ± 7 |
4.2. Effect of pH on Particle Size of Chitosan NP
Table 2 shows a slight fall in particle size of chitosan NP by enhancement of pH value at different concentrations of CS (P < 0.05) but no explainable difference was observed between particle sizes of formulations prepared by various CS concentrations at each defined pH.
| Particle Size of CS/TPP Solutions at Different pH Values, nm | |||
|---|---|---|---|
| CS Solution Concentration, mg/mL | pH = 5.9 | pH = 7.4 | pH = 8.4 |
| 1 | 191 ± 4 | 125 ± 16 | 101 ± 9 |
| 2 | 279 ± 3 | 200.5 ± 1.5 | 86.6 ± 7.6 |
| 4 | 121.5 ± 12.9 | 103.5 ± 9.1 | 94 ± 8.6 |
| 10 | 124.5 ± 40.2 | 104.1 ± 4.3 | 101.1 ± 1.8 |
4.3. Diclofenac-Loaded Chitosan Nanoparticles Characterizations
The results of the experiments are presented in Table 3. Drug-loaded nanoparticles exhibited particle sizes ranging from 129 to 166 nm with a polydispersity index (PDI) under 0.4. A significant enhancement in CS/Drug proportion led to larger particle sizes. Transmission electron microscopy (TEM) revealed that the morphology ofnanoparticles in F1, F2, and F3 formulations were close to a spherical shape (Figure 1). As shown in Table 3, the zeta potential of all formulations were positive with values ranging from 19.05 to 21.25 mV. Drug loading efficiency of formulations was within a range of 14.48 - 65.43% (Table 3).
| Formulation Name | CS/Drug Ratio | LE | Size, nm | PDI | Zeta Potential, mV |
|---|---|---|---|---|---|
| F1 | 10/1 | 65.43 ± 7.03 | 166.35 ± 16.05 | 0.323 ± 0.03 | 19.05 ± 0.49 |
| F2 | 5/1 | 14.48 ± 6.72 | 129.15 ± 10.67 | 0.393 ± 0.018 | 21.25 ± 0.21 |
| F3 | 4/1 | 45.16 ± 14.9 | 148.35 ± 3.74 | 0.313 ± 0.002 | 19.95 ± 1.34 |
4.4. Evaluation of Differential Scanning Calorimetry
Differential scanning calorimetry of pure drug, CS, TPP, and F1, F2 and F3 formulations were performed and thermograms were obtained at a heating range of 25 - 300°C (Figure 2). The DSC thermogram of pure sodium diclofenac exhibited two phases of transition states at 298°C and 328°C, which could be observed before exposure to CS and TPP molecules. Chitosan powder showed an endothermic peak at 140°C and an exothermic peak at 325°C. Furthermore, thermogram of TPP also showed two endothermic peaks at 120°C and 210°C. In the DSC thermograms of F1, F2, and F3 formulations, the peaks of the single components were not visible and only a broad endothermic peak was observed within the range of 140 - 170°C.
4.5. Drug Release Studies
As it can be observed from Figure 3, the release of diclofenac was much lower in acidic conditions (pH 1.2) when compared to pH 6.8 and 7.4. The maximum drug release after eight hours from F1 at pH 1.2 was 0.94%. However, 93.3% of the drug was released from this formulation, at pH 7.4, during two hours.
5. Discussion
According to the ionotropic gelation method, in acidic medium, the -NH2 groups of CS are protonated and produce -NH3+, which in the presence of a polyanion such as TPP, form gel because of ionic interactions (11). This phenomenon results in reduction of CS aqueous solubility, and thus nanoparticles spontaneously form under mechanical stirring at room temperature (12).
The suspensions with CS/TPP ratios of the first drops of 1:1, 2:1 and 3:1 showed undesirable turbidity with the addition of TPP aqueous solution to CS acidic solution. This was possibly due to spontaneous formation of CS-TPP microparticles in the presence of high concentrations of TPP. Therefore, 10:1, 5:1 and 4:1 ratios of CS/TPP mixtures were prepared for determination of size and size distribution. The results indicated that by enhancement of CS/TPP ratio, larger nanoparticles could be obtained. Previous study showed that the size distribution of CS nanoparticles and their biological properties are remarkably influenced by CS/TPP ratio (17). In the present study, the optimum size distribution was obtained at 4:1 CS/TPP ratio; in other words this ratio produces more impact on nano-structures because of the high interactions of ions of opposite charges, therefore drug-loaded nanoparticles were prepared with a ratio of 4:1 CS/TPP.
The results of this study showed a negative correlation between pH and mean particle size of chitosan NPs. Previous studies reported that size distribution of CS nanoparticles were significantly affected by environmental changes such as pH and ionic strength of the solution (18). Tsai et al. studied the effect the pH of storage solutions on the size of CS/TPP nanoparticles. Their results indicated that the size of the nanoparticles decreased with increase of the solutions’ pH. They suggested that CS/TPP nanoparticles are metastable nano-gels, and their structures are easily changed with different environmental conditions such as pH and ionic strength of the solutions (18). Chitosan is a polycation, which expands at lower pH values due to higher protonation and electrostatic repulsive interaction between polymer chains, and produces nanoparticles with larger particle size. On the contrary, at higher pH values, this polymer intensively shrinks and forms nanoparticles with smaller sizes (14). However, higher levels of pH could be used to produce smaller sized particles unless the route of administration restricts range of applied pH in preparation.
Drug-loaded nanoparticles exhibited a small size (129 to 166 nm). When CS/D ratio was changed from 10:1 to 4:1, the particle size was decreased from 166 to 148 nm. The presence of high levels of drug during gel formation led to the production of gaps between CS and TPP and decrease in CS-TPP interaction; thus particles with larger sizes were produced. A low value of PDI (PDI < 0.4) for F1, F2 and F3 formulations possessed a narrow distribution of size. The PDI is a measure of the heterogeneity of sizes of molecules or particles in a mixture with values ranging from 0 to 1. It has been elucidated that PDI values close to zero represent homogeneous dispersion and those greater than 0.5 show high heterogeneity (19).
Zeta potential is an important physicochemical property of nanoparticles and can affect physical stability of particles and their mucoadhesive properties (20). It was elucidated that residual protonated amine groups after gel formation were responsible for the positive zeta potential. Previous studies have indicated that higher values of zeta potential result in production of more stable nanoparticles. The electrostatic repulsion between particles with the same electrical charge prevents the aggregation of the particles (20). In addition, long chain amino groups of CS absorb anionic groups of TPP and prevent aggregation by establishment of a thick electrical double-layer (21). Despite that no clear trend of increasing loading efficiency is observed by increasing CS/D ratio of formulations, yet there is a positive correlation between LE and particle size of different formulations (P < 0.05).
Differential scanning calorimetry thermogram of pure sodium diclofenac exhibited an endothermic peak at 298°C, which represents the fusion of solvated crystals while presence of an exothermic peak at 328°C was due to the oxidation reaction between DS and oxygen in air environment fusion (16). The endothermic peak in CS thermogram might be related to the glass transition temperature (Tg) of Cs and the exothermic peak at 325°C was probably due to degradation of the polymer (22-24). Thermograms of nanoparticle formulations were the same and showed only a broad endothermic peak around 140 - 170°C. The comparison between thermograms of TPP and the formulations demonstrates the disappearance of the endothermic peak of TPP at 210°C and association of another peak (at 120°C) with the wide peak in formulations thermograms. This can be due to the polyelectrolyte interaction between chitosan and TPP as a result of opposite charge interactions (25). Furthermore, the omission of endothermic and exothermic peaks of the drug in the formulations thermograms suggests that the drug exists in an amorphous or disordered crystalline phase as a molecular dispersion in the nanoparticle network (26-28).
The effect of pH on drug release was evaluated for three candidate formulations (F1, F2, and F3) containing nanoparticles. It is evident that the overall release of diclofenac in acidic pH was less than that at pH 6.8 and 7.4 due to low solubility of drug in acidic solution. Besides, in acidic medium, protonation of the amine groups of chitosan and presence of negative charges of polyanion (TPP) cause tightening of the network in the nanoparticle system. This effect results in less swelling thus retardation of drug release (25, 29, 30). Our findings are in line with the study of Naidu et al. (29) who reported that water uptake of diclofenac-loaded polyelectrolyte complex of chitosan and gum Kondagogu was maximal at pH 6.8 (0.1 phosphate buffer) when compared to acidic solution. In the present study, with increase in CS/D ratio in the nanoparticle formulations, the release of diclofenac was enhanced at pH 6.8 and 7.4. Chitosan is a hydrophilic polymer and can promote the entry of solution into the particles and greatly improves the solubility of diclofenac, thus accelerates its dissolution (29).
In the present study, a CS nanoparticulate system loaded with diclofenac was prepared and characterized based on the ionotropic gelation method. Slow drug release pattern in acidic medium and a rapid drug release at higher pH illustrates that CS nanoparticles could be further evaluated for the enteric delivery of diclofenac, to inhibit side effects of the drug on stomach tissue. However, despite the rapid drug release at pH 7.4, the prepared nanoparticle systems had high capacity for use in ophthalmic drug delivery. It should be noted that the maximum content of lachrymal fluids in each eye was 30 µL, which is much less than the applied volume in the dissolution test (30 mL), therefore it is expected for the release of drug from particulate systems in ocular cavity to have a much slower pattern. On the other hand, mucoadhesive properties of chitosan were able to prolong the retention time and bio-distribution of drug in the ocular system.