1. Background
Paraquat (PQ) is a non-selective dipyridyl herbicide with high toxicity that is used extensively. PQ can cause severe health issues in humans and animals (1). Exposure to PQ (methyl viologen), both accidental and voluntary, has caused thousands of human deaths. The ingestion of a small amount of this chemical compound (55 mg/kg) leads to the accumulation of PQ in the lungs, resulting in alveolitis, pulmonary edema, and the proliferation of bronchial epithelial cells; this may be followed by the proliferation of fibroblasts, the deposition of collagen, and fibrosis, which is the leading cause of decreased cases of PQ poisoning (2). Mitochondria, which are major sites for ATP production and for the regulation of energy expenditure, are associated with the production of reactive oxygen species (ROS) and apoptosis pathway signaling (3). Acute PQ exposure leads to mitochondrial dysfunction and toxicity in human and animal tissues (4). The cyclic oxidoreduction of PQ, which generates ROS, the hydroxyl free radical (HO•), and peroxinitrite (ONOO-), depletes cellular NADPH. This and lipid peroxidation (LPO) are the major mechanisms of PQ toxicity. Mitochondrial dysfunction and oxidative stress may play important roles in the pathological consequences of human exposure to PQ (5-8). Flavonoids, which are abundant antioxidants and are free radical scavengers, may be useful in fighting oxidative stress (9). Past studies have demonstrated the significant protective effect of flavonoids containing dietary sources and have suggested that health risks following exposure to PQ are reduced using flavonoids (10, 11). The potent antioxidant activity of naringenin (NG) (4’, 5, 7-trihydroxy-flavonone-7-rhamnoglucoside) makes it a candidate for use in counteracting the damage induced by oxidative stress (12). NG is a well-known flavonoid found in grapefruit, tomatoes, and citrus fruit (13). NG has a wide spectrum of pharmacological properties, including potential anticarcinogenic, antimutagenic, nephroprotective, hepatoprotective, and antiatherogenic activities (14). The antioxidant effect of NG is mainly attributed to the existence of a carbonyl group and hydroxyl groups (15).
2. Objectives
The aim of the current study is to identify the beneficial effects of NG on counteracting oxidative stress induced by PQ in mitochondria that are isolated from rat livers.
3. Materials and Methods
3.1. Chemicals
NG, PQ dichloride, dithiobis-2-nitrobenzoic acid (DTNB), reduced glutathione (GSH), 2’, 7’-dichlorofluorescin diacetate (DCF-DA), rhodamine 123 (Rh123), Coomassie blue G, ethylene glycol-bis (2-aminoethylether)-N,N,N’,N’-tetraacetic acid (EGTA), tetraethoxypropane (TEP), 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT), thiobarbituric acid (TBA), bovine serum albumin (BSA), D-mannitol, sucrose, and 2-[4-(2-hydroxyethyl) piperazin-1-yl] ethanesulfonic acid (HEPES) were purchased from the Sigma chemical company. Other chemicals used in the experiments were of analytical grades.
3.2. Animals
Male Wistar rats (200 - 250 g) were obtained from Ahvaz Jundishapur University of Medical Sciences. The rats were kept in polypropylene cages and were fed standard rat chow; the rats also had free access to water. The animals were kept at a controlled temperature (25 ± 2°C) and were subjected to a light cycle lasting 12 hours and a dark cycle lasting 12 hours. All studies conducted with the animals were completed according to the ethical standards and protocols approved by the committee of animal experimentation of Ahvaz Jundishapur University of Medical Sciences in Iran.
3.3. Preparation of Mitochondria
Mitochondria were prepared from Wistar rat livers using differential centrifugation. The removed livers were minced with small scissors in a cold medium containing 200 mM of d-mannitol, 70 mM of sucrose, 1 mM of EGTA, 0.1% (w/v) of BSA, and 10 mM of HEPES-KOH. This medium was prepared fresh, and the pH was adjusted to 7.4. The minced livers were gradually homogenized in a glass homogenizer and centrifuged at 600 g for 10 minutes at 4°C; this process was used to sediment the nuclei and the broken cell debris. Then, the supernatants were centrifuged at 10,000 g for 15 minutes. The inferior layer, which contained mitochondrial pellets, was re-suspended in the isolated medium and removed. The inferior layer then underwent centrifugation at 10,000 g for 10 minutes. The final pellets from the mitochondria were suspended in the mannitol solution. The protein concentrations were measured with a Coomassie blue protein-binding method, as used by Bradford (16).
3.4. Experimental Design
To determine the IC50 of PQ, the mitochondrial viability was determined using an MTT assay. Different concentrations of PQ (0, 2.5, 5, and 10 mM) were used to determine the IC50 of PQ. The suspensions of the mitochondria (at concentrations of 0.5 mg protein/ml) were incubated with various NG concentrations (100, 200, and 400 µM) for 30 minutes and were then treated with PQ at a concentration of IC50 at 37°C for another 30 minutes. The aim of centrifugation following NG exposure was to remove NG from the suspension medium, eliminating the possible interaction that may have occurred with PQ. The following factors were then investigated.
3.5. Mitochondrial Complex II Activity (Viability)
Mitochondrial complex II activity is mediated using a succinate dehydrogenase enzyme. The activity of mitochondrial complex II was assessed by measuring the reduction of MTT. One ml of the mitochondrial suspensions (at a density of 0.5 mg protein/ml) was centrifuged at 10,000 g for 1 minute. The pellets were re-suspended in 970 µL of the isolation medium and 500 µL of the 0.5 µM MTT. The pellets were then incubated at 37°C for 30 minutes. The purple formazan of the produced crystals was dissolved in 800 µL of DMSO, and the absorbency was measured at 570 nm using a spectrophotometer (UV-1650 PC, Shimadzu, Japan) (17).
3.6. Measurement of the GSH Content
Using DTNB as an indicator, the GSH content was determined using a spectrophotometric method in the isolated mitochondria (18). The mitochondrial suspensions (0.5 mg protein/mL) were added to 0.04% DTNB that was placed in 0.1 mol/L of phosphate buffers at a pH of 7.4. The yellow color that developed was measured at 412 nm using a spectrophotometer (UV-1650 PC, Shimadzu, Japan), and the GSH content was expressed as µg/mg of protein.
3.7. Mitochondrial Membrane Damage
The mitochondrial uptake of rhodamine 123, a cationic fluorescent dye, is widely used to determine the mitochondrial membrane potential (MMP). The mitochondrial suspensions, with densities of 0.5 mg protein/ml, were incubated using 1.5 µM of rhodamine 123 at 37°C for 10 minutes and were shaken gently. The fluorescence was measured using a Perkin Elmer LS-50B luminescence spectrophotometer. The excitation and emission wavelengths measured to 490 and 535 nm, respectively (19).
3.8. Mitochondrial ROS Formation
The measurement of the mitochondrial ROS was completed using the fluorescent probe DCF-DA. The isolated mitochondrial suspensions (0.5 mg protein/ml) were incubated with 1.6 µM of DCF-DA at 37°C for 10 minutes. The fluorescence was measured using the Perkin Elmer LS-50B luminescence spectrophotometer. The excitation and emission wavelengths amounted to 500 and 520 nm, respectively (20).
3.9. Measurement of LPO
LPO, as indicated by the formation of malondialdehyde (MDA), was measured according to the method outlined by Zhang et al. (21). The suspensions of the mitochondria, which were at a density of 0.5 mg protein/ml, were incubated in 0.25 mL of sulfuric acid (0.05 M) and 0.3 mL of 0.2% TBA. The tubes were placed in a boiling water bath for 30 minutes and were then placed in an ice bath. Following the addition of 0.4 mL of n-butanol, they were centrifuged at 3500 g for 10 minutes. The amount of MDA that formed was assessed by measuring the absorbance of the supernatant; this was completed at 532 nm using a spectrophotometer (UV-1650 PC, Shimadzu, Japan). TEP was used as the standard, and the MDA content was expressed as nmol/mg of protein.
3.10. Statistical Analysis
The treated experimental groups were compared using analysis of variance and significance, followed by a post hoc Tukey’s test. The IC50 of PQ was calculated using Minitab 16 statistical software. The data were presented as the mean ± SD (n = 3). Probability values of <0.05 were identified as statistically significant.
4. Results
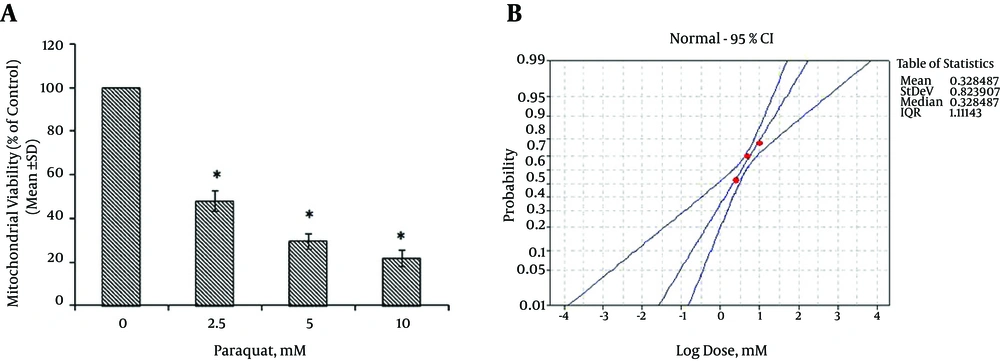
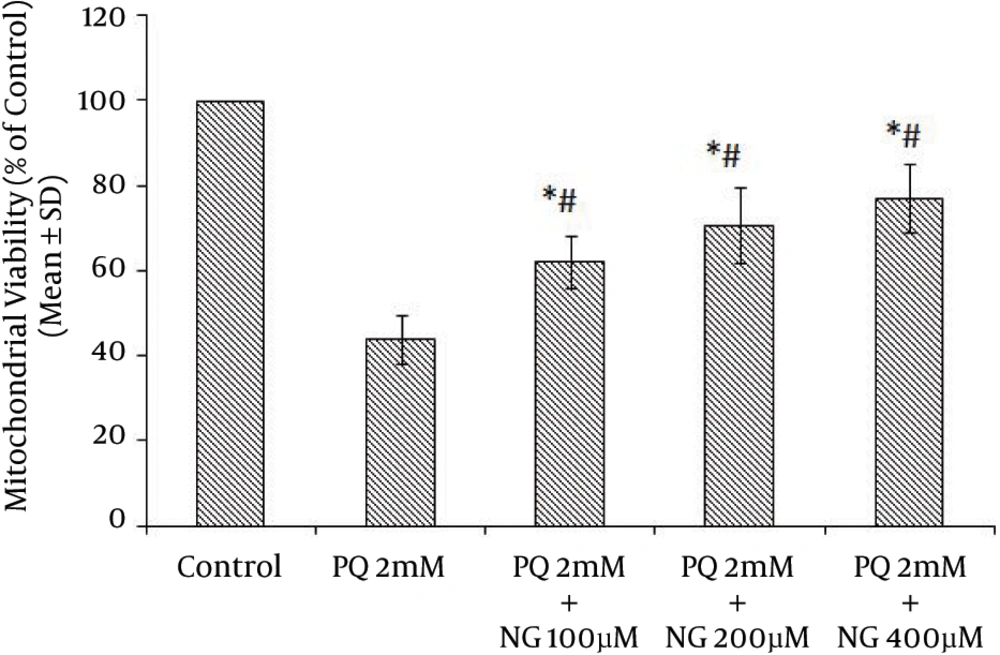
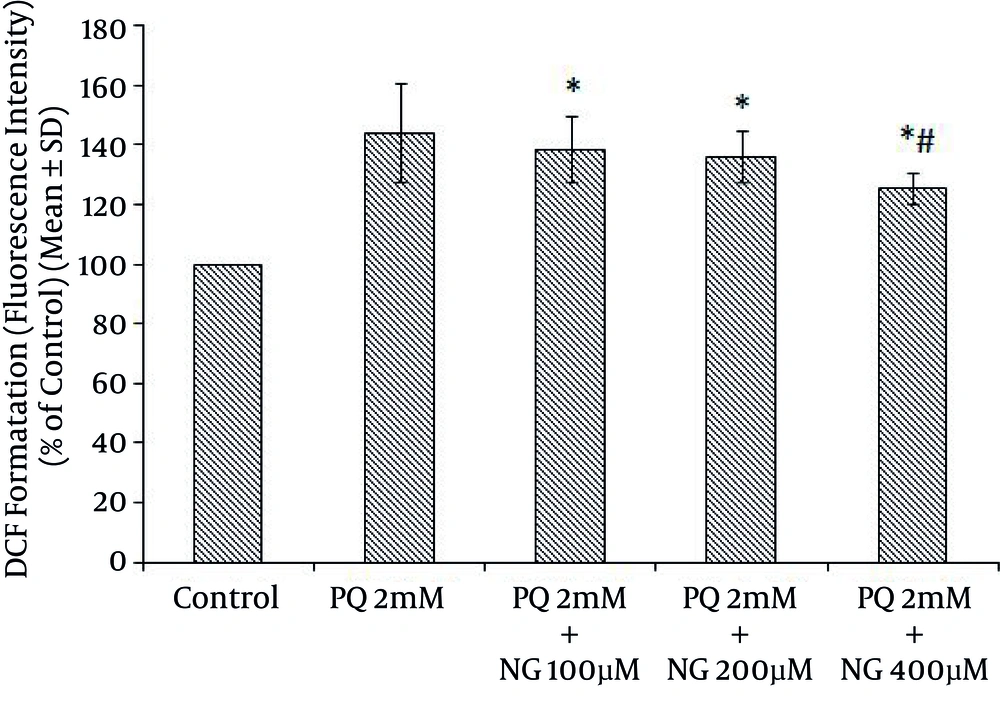
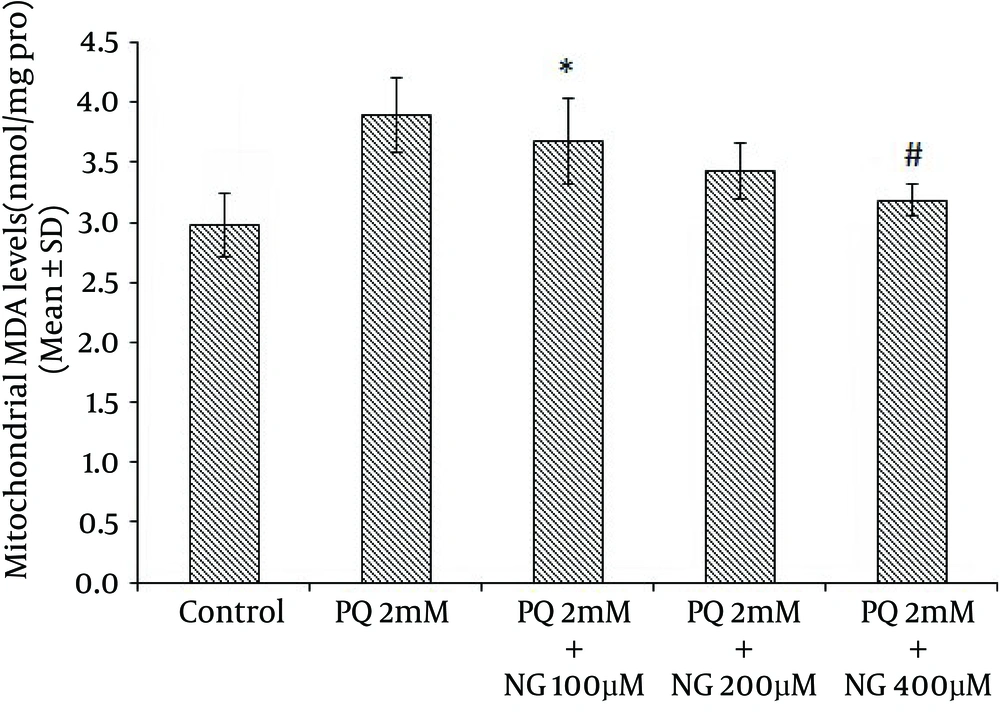
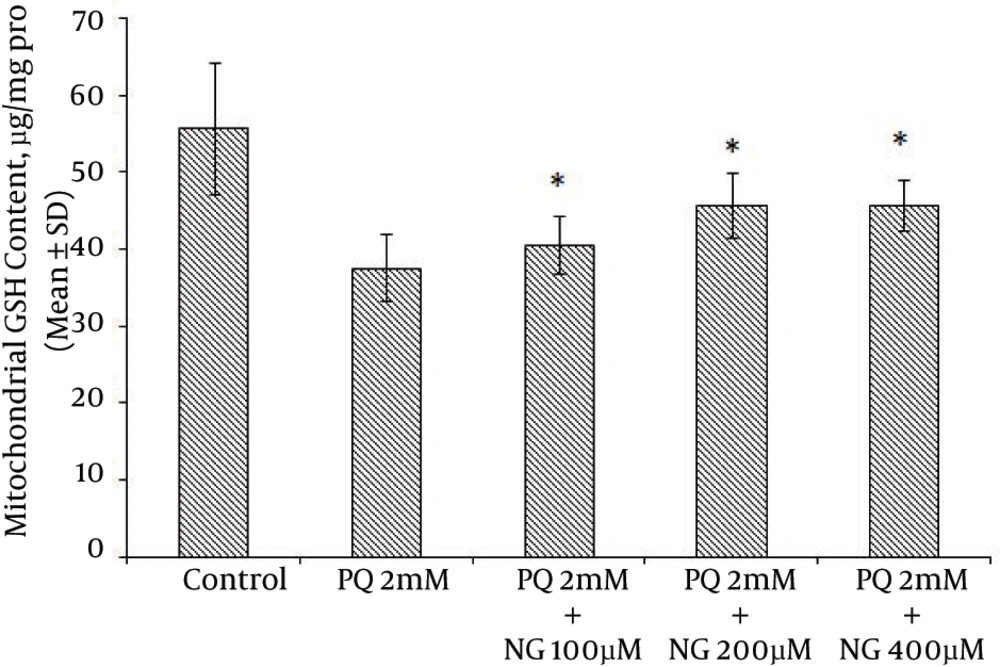
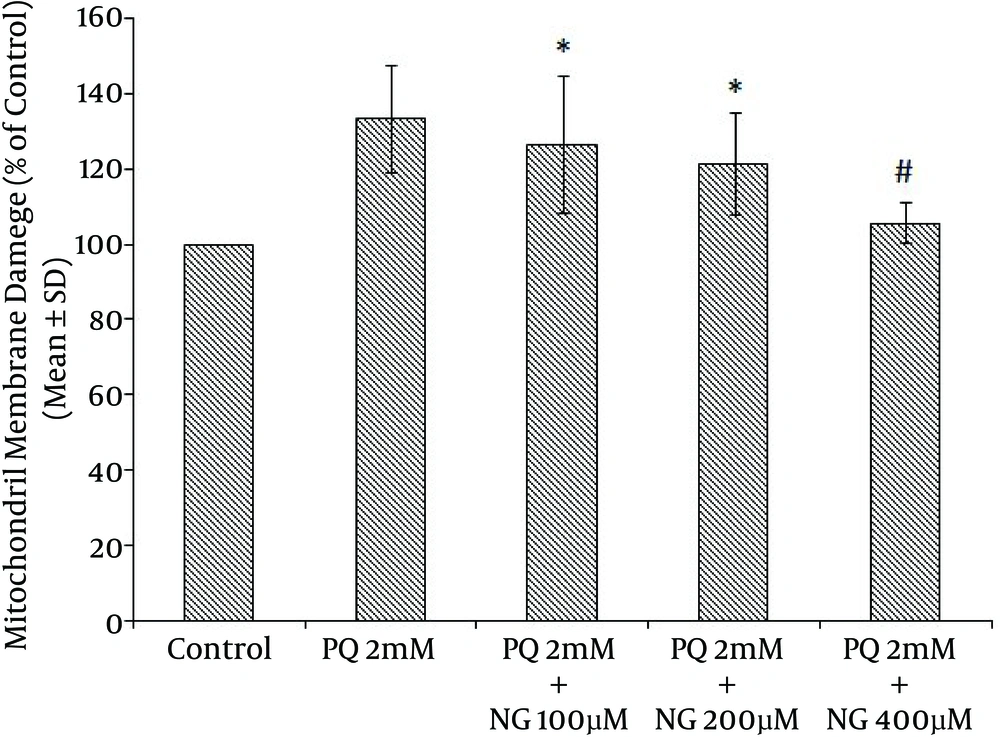
Based on the different concentrations of PQ in the mitochondrial suspensions, the IC50 of PQ was determined to be 2 mM (Figure 1). To determine the viability, the succinate dehydrogenase activity (mitochondrial complex II) was evaluated using an MTT test after the mitochondria were incubated for 30 minutes with 2 mM concentrations of PQ only and with a combination of different NG concentrations (100, 200, and 400 µM). Figure 2 shows the significant decrease in the mitochondrial metabolism of MTT in response to formazan at a concentration of 2 mM of PQ in comparison to the control group (P < 0.05). NG caused a significant concentration-dependent increase in the mitochondrial metabolism of MTT in response to formazan (P < 0.05). As shown in Figure 3, following PQ exposure (at a concentration of 2 mM), ROS generation increased significantly in the isolated liver mitochondria as compared to the control group (P < 0.05). However, ROS formation was significantly inhibited (P < 0.05) by NG pretreatment (200 and 400 µM). As shown in Figure 4, the levels of mitochondrial MDA as biomarkers of LPO were elevated in final LPO process. In the isolated liver mitochondria, MDA increased markedly after PQ exposure, and the administration of NG significantly prevented (P < 0.05) PQ-induced mitochondrial LPO. According to Figure 5, the GSH content of the PQ-treated mitochondria was significantly lower than that of the controls (P < 0.05). The group that was treated with NG (200 and 400 µM) experienced significant inhibition (P < 0.05) of PQ-induced mitochondrial GSH oxidation. Mitochondrial membrane damage is a sensitive indicator of the state of the mitochondrial inner membrane. Rhodamine 123 has been used to determine the disruptive effect of PQ on damage to the mitochondrial membrane. The results shown in Figure 6 suggest that, at a concentration of 2 mM, PQ significantly reduces mitochondrial membrane damage (P < 0.05). Pretreatment with NG (200 and 400 µM) significantly inhibits the prevention of mitochondrial membrane damage induced by PQ (P < 0.05).
5. Discussion
PQ is commonly used as a potent herbicide and is very toxic to humans and animals. It has been used as a putative agent in the development of oxidative stress. Many diseases, such as cardiovascular diseases, cancer, diabetes, cataracts, lung diseases, schizophrenia, and Parkinson’s disease, may be caused by the generation of ROS (22). ROS, hydroxyl free radicals such as HO•, and peroxynitrite (ONOO-) generated as a result of the metabolism of PQ by inducing mitochondrial toxicity were recommended as potential mechanisms for PQ mitochondrial toxicity (23). For this purpose, the ameliorative effects of NG on PQ-induced toxicity were shown using toxicity parameters, including the GSH level, mitochondrial complex II activity (MTT), mitochondrial membrane damage, mitochondrial ROS formation, and the measurement of LPO (MDA). Succinate dehydrogenase (succinate: ubiquinone oxidoreductase; mitochondrial complex II) plays a central role at high respiration rates. Therefore, the activity of this enzyme is a good marker for the mitochondrial oxidative capacity (24). Our results showed a significant reduction in complex II activity following PQ (2 mM), which contributed to PQ toxicity. Increasing mitochondrial viability using NG occurred at concentrations of 200 and 400 µM (Figure 2). The addition of NG to mitochondrial suspensions exposed to PQ produced a significant increase in complex II activity compared to the use of PQ (2 mM) alone. NG may reduce complex II by binding to this complex’s ubiquinone-binding sites (25). In normal physiological conditions, the ROS generated in the mitochondria are removed or scavenged by the antioxidant defense system. Several researchers have used antioxidants, such as vitamin E (26) and melatonin, as treatments for ROS production. It has been reported that the beneficial effect of NG on PQ toxicity is evident via the enhancement of the endogenous antioxidant system, which prevents lung fibrosis (27). One possible explanation for PQ-induced mitochondrial dysfunction is that the mitochondrial antioxidant system is ineffective at detoxifying the relevant ROS (28). NG has active flavonoids that can react with superoxide anion radicals or with hydroxyl radicals that act as radical scavengers (13). Our results showed a significant increase in ROS generation following PQ (2 mM), which contributes to PQ toxicity. In addition, the reduction in ROS generation with the use of NG occurred at concentrations of 200 and 400 µM (Figure 3). NG decreased the ROS levels that were generated by PQ (2 mM). LPO has been known to have a number of harmful effects, such as increased membrane tightness, osmotic crispiness, reduced cellular and subcellular components, decreased mitochondrial survival, and lipid fluidity (29). Our results suggest that PQ may support its effect by inhibiting membrane LPO that is mediated by the superoxide anion (Figure 4). Mitochondrial membrane damage is an electrochemical potential that consists of a transmembrane electrical potential and a proton gradient. MMP collapse is an early consequence of mitochondrial dysfunction (20). PQ significantly induced MMP collapse (P < 0.05), which was reversed by NG at concentrations of 200 and 400 µM (Figure 6). This indicates that PQ-induced MMP collapse is prevented by NG (P < 0.05) (Figure 6). Numerous enzymatic and nonenzymatic defense mechanisms, such as GSH peroxidase and total SH proteins, have main roles in regulating the biological effect of ROS. GSH is the major antioxidant for enzymatic defense mechanisms, with a low molecular weight in cells. Total SH protein levels are reduced during oxidative stress induced by toxicants (26). In the present study, GSH levels in the isolated mitochondria were measured. Among the agents that cause oxidative stress, PQ is a thiol(-SH)-oxidizing factor that results in the swift oxidation of GSH to GSSG. GSH depletion enhances the sensitivity of the mitochondrial structure to ROS-mediated damage (26). Our results indicate that treatment with NG ameliorates the decrease in total GSH levels resulting from PQ. Since GSH is a unique marker of oxidative stress, NG affects the GSH cascade (Figure 5) (26). The protective effects of NG, as shown by its impact on GSH content and LPO, suggest that exogenously administrated antioxidants such as NG may either restore the mitochondrial antioxidant status or improve mitochondrial functions, thus protecting these organelles from oxidant damage.