1. Background
Achillea millefolium (A. millefolium) or yarrow is a member of the Asteraceae family and is used frequently as a medicinal plant worldwide for the treatment of wounds, hemorrhages headaches, inflammation, spasmodic diseases, flatulence, and dyspepsia (1, 2). Recent research suggested that A. millefolium might serve as a complementary therapy for the treatment of nervous system-related disorders. One study reported that A. millefolium extract had anti-inflammatory effects in primary cultures of brain microglial cells and that it may have a role in the prevention and treatment of neurodegenerative disorders, such as Alzheimer’s disease and Parkinson’s disease (3). Another study indicated that aqueous extract of A. millefolium inhibited clinical scores and inflammation in an animal model of multiple sclerosis (4). In addition, in rodent studies, A. millefolium was reported to have anticonflict (5) and anxiolytic properties (6). Given the potential beneficial effects of A. millefolium extract in the prevention or treatment of central nervous system diseases and the increase in its use, understanding possible adverse impacts of this plant on the brain is important.
The whisker barrel cortex is a discrete region in the primary somatosensory cortex of rodents that is involved in the cortical representation of whiskers. Previous research demonstrated a one-to-one relationship between vibrissae (whiskers) and specific areas of the barrel cortex, with the neurons in each barrel showing greater responses to the principal whiskers (PWs) than to several adjacent whiskers (AWs) (7, 8). Cortical neuronal receptive fields have both excitatory and inhibitory components. These features make the whisker-barrel system a good model to study neuronal electrophysiological properties in the cortex.
2. Objectives
This study investigated the effects of A. millefolium extract on neuronal responses to natural stimuli using the rat whisker-barrel system as a model.
3. Methods
3.1. Animals
Twenty-one male Wistar rats were randomly divided into three groups, with seven animals in each group. They were kept in standard cages, under a 12/12- hour light-dark cycle and a temperature of 23 ± 2.0°C, with free access to food and water.
All the experimental procedures were carried out in accordance with the guidelines for the care and use of laboratory animals of Rafsanjan University of Medical Sciences and the European communities council directive of 24 November 1986 (86/609/EEC).
3.2. Plant Material
Leaves and flowers of A. millefolium were collected from cultivated plants in Isfahan botanical garden in March 2013. Dr. Valiollah Mozaffarian of the botany research division, research institute of forests and rangelands, Tehran, Iran identified the plant. A voucher specimen has been deposited in Isfahan botany herbarium (voucher specimen number: 9757).
3.3. Plant Extraction
The leaves and flowers of the plant were dried and powdered. The powder (4 g) was added to 200 ml of distilled water (80°C) and stirred for 15 min. The mixture was then filtered, and the solvent was evaporated under reduced pressure at 45°C using a rotary evaporator. The resultant extract (500 mg) was stored at 4°C. The extract was reconstituted by dissolving it in distilled water before use.
3.4. Animal Groups
The animals were randomly divided into a vehicle group and two treatment groups (n = 7 in each), which received 1.4 and 2.8 mg/kg of A. millefolium aqueous extract, respectively. These doses are similar to those proposed for human use in traditional medicine (9).
3.5. Anesthesia
The rats were anesthetized using urethane (1.5 g/kg, intraperitoneal). Using a stereotactic apparatus (Stoelting, U.S.), craniotomy (4 mm) of the right somatosensory cortex was performed, and a solution of 3% agar in saline was used to cover the cortex. The absence of spontaneous movement and overt whisking or whisker tremor were monitored to ensure a suitable depth of anesthesia. A sluggish hind limb withdrawal reflex was confirmed by firm pinching of the hind paw. Corneal and eyelid reflexes were present. Respiration was regular (80 - 100 breaths/min). Additional doses of urethane (10% induction dose) were used to maintain the level of anesthesia. The rectal temperature was maintained at 37°C using a servo-controlled heating blanket (Harvard Apparatus, Edenbridge, U.K.).
3.6. Extracellular Single-Unit Recording
Extracellular recordings were carried out using tungsten microelectrodes (FHC, Bowdoinham, ME, U.S.). The electrodes were oriented perpendicular to the cortical surface to measure extracellular neuronal activity at a depth of 500 - 800 µm from the surface of the skull microelectrodes were advanced with the aid of a micromanipulator (WPI Company). The data recorded from the tungsten microelectrodes were amplified and digitized by an electrophysiology recording system (Electromodule; Science Beam Inst., Tehran, Iran). Single neuron action potentials were isolated online using an amplitude window discriminator provided by eProbe software (recording and analyzing software solution, science Beam Inst.), and the waveforms and amplitudes of the signals were used to determine the accuracy of isolation. Inter-spike intervals were calculated in the first 50 ms after deflection of the spared PW, and additional criteria were used to ensure the precision of single unit isolation. At all the recording sites, the average inter-spike intervals were significantly greater than 1 ms (one-tailed t-test, all P < 0.05). Similar to a previous study (8), the spikes collected from depths of 500 - 800 µm were located, almost without exception, within the defined barrel territory, as evidenced by high cytochrome oxidase density in these regions.
3.7. Whisker Stimulation
Fine mechanical displacement was produced by two tiny speakers, which were triggered by a trapezoid-shaped waveform of an electrophysiology recording system (Electromodule, Science Beam Inst.). For whisker deflection, a thin glass tube with an internal diameter of 0.69 mm was attached to the center of each speaker, and a current was delivered by computer software. This current produced ramp and hold movements. It had an amplitude of 500 µm, onset and offset rise times of 5 ms, and a duration of 200 ms. The PWs and AWs were cut 10 mm from the base and placed inside the tubes (10).
3.8. Experimental Procedures
As explained elsewhere (11), two discrete responses follow whisker displacement: an “ON” response and an “OFF” response. The ON response is elicited when the stimulator begins to move, and the OFF response commences when the stimulator returns to its resting position (12). To study the responses of the barrel cortex neurons to combined stimulation of two vibrissae, a conditioning-test paradigm was used (13). During the conditioning paradigm, the AWs and PWs were deflected in succession at 0-, 5-, 10-, 20-, 30-, 50-, and 100-ms inter-stimulus intervals (ISIs). Each of the stimulation patterns (two single and seven combined whisker deflections) was repeated 50 times in a pseudo-random order. Each stimulation pattern lasted 1 second. Electromodule software was used both for the stimulus delivery and storage of the spike times. peristimulus time histograms (PSTHs) were produced in response to each of the nine deflection patterns. Spike units during the first 200 ms of each recording file (when there was no stimulation) were counted as spontaneous activity. The magnitudes of the ON and OFF responses were calculated by counting the number of spikes 5 - 35 ms after the onset of each movement. Whisker interactions were quantified using a condition-test ratio (CTR). The CTR index was calculated as the ratio of the average PW response when deflected after the AW (in the combined defection paradigm) to the response to PW deflection alone (14, 15). A ratio of less than 1 indicated response suppression, whereas a ratio of more than 1 indicated response facilitation.
The recordings were done at four time points: before the administration of the A. millefolium aqueous extract to obtain data on basic neuronal spontaneous activity and triggered responses and 0.5, 1, and 2 hours after oral administration of the extract.
3.9. Statistical Analysis
The recorded signals were imported to eSorter (Spike Sorting Software toolbox, www.ScienceBeam.com), and the peaks of the spike potentials and valley amplitudes were extracted and classified by k-means clustering offline. The classified spikes were further analyzed to detect ON and OFF responses using eSpike (spike analyzing software toolbox, www.ScienceBeam.com). The comparison between groups was done using a one-way ANOVA. For post-hoc comparisons, Tukey’s test was used. A P value less than 0.05 was considered significant. For comparing ON and OFF responses and the CTR at different times after A. millefolium administration, a repeated measures ANOVA was used.
4. Results
4.1. Effects of the A. millefolium Extract on Excitatory Receptive Fields
4.1.1. Response to PW Deflection
Figure 1 illustrates the modulation of the response of layer IV neurons to PW by the A. millefolium aqueous extract. The application of the different doses (1.4 and 2.8 mg/kg) of the extract at various times (0.5, 1, and 2 hours) did not affect the magnitude of neuronal ON and OFF responses to PW deflection (Table 1).
Population peristimulus time histograms (PSTHs) of ON responses to principal whisker (PW) deflection in the different groups (vehicle [n = 14 units], 1.4 mg/kg extract [n = 14 units], and 2.8 mg/kg extract [n = 14 units]). Each panel shows the accumulated responses to 50 deflections of the PWs. All the PSTHs are plotted on the same scale bar, where time 0 corresponds to the time of whisker deflection and time -10 shows the responses 10 ms before the start of deflection. The insets present typical examples of PSTHs (bin size = 1 ms), showing ON and OFF neuronal responses to 50 PW deflections. The whisker deflection paradigm is depicted in lines below each trace. NS; normal saline.
| Time of Recording | ON Response | OFF Response | ||||
|---|---|---|---|---|---|---|
| Groups | Groups | |||||
| Vehicle | 1.4 mg/kg | 2.8 mg/kg | Vehicle | 1.4 mg/kg | 2.8 mg/kg | |
| Before gavage (baseline) | 6.08 ± 0.46 | 6.62 ± 0.57 | 7.47 ± 0.44 | 5.39 ± 0.75 | 5.27 ± 0.73 | 5.86 ± 0.59 |
| 0.5 hour after gavage | 7.21 ± 0.62 | 7.37 ± 0.64 | 7.14 ± 0.88 | 5.59 ± 0.9 | 5.85 ± 0.78 | 6.49 ± 0.86 |
| 1 hour after gavage | 7.91 ± 0.78 | 6.72 ± 0.7 | 7.61 ± 0.85 | 6.34 ± 0.92 | 5.64 ± 0.8 | 6.52 ± 0.78 |
| 2 hours after gavage | 7.76 ± 0.84 | 6.79 ± 0.84 | 7.85 ± 0.78 | 7.02 ± 1.03 | 5.28 ± 0.8 | 6.15 ± 0.77 |
a1.4mg/kg and 2.8 mg/kg: groups that received 1.4 and 2.8 mg/kg/mL of the Achillea millefolium aqueous extract, respectively. The data represent the mean of neural spikes/bin size/50 stimulations. The bin size was set at 1 ms.
4.1.2. Response to AW Deflection
Neither dose (1.4 or 2.8 mg/kg) of the A. millefolium extract had any effect on the ON and OFF neuronal responses to AW deflection at any of the treatment times after acute oral administration (Table 2).
| Time of Recording | ON Response | OFF Response | ||||
|---|---|---|---|---|---|---|
| Groups | Groups | |||||
| Vehicle | 1.4 mg/kg | 2.8 mg/kg | Vehicle | 1.4 mg/kg | 2.8 mg/kg | |
| Before gavage (baseline) | 3.81 ± 0.75 | 3.85 ± 0.66 | 3.84 ± 0.79 | 3.21 ± 0.63 | 3.31 ± 0.68 | 4.24 ± 0.84 |
| 0.5 hour after gavage | 4.51 ± 0.8 | 4.34 ± 0.73 | 4.20 ± 0.8 | 3.57 ± 0.72 | 3.47 ± 0.71 | 4.05 ± 0.9 |
| 1 hour after gavage | 4.70 ± 0.8 | 4.49 ± 0.78 | 4.24 ± 0.77 | 4.52 ± 0.8 | 3.84 ± 0.73 | 4.11 ± 0.68 |
| 2 hours after gavage | 5.66 ± 0.93 | 4.33 ± 0.74 | 4.37 ± 0.85 | 4.54 ± 0.77 | 3.99 ± 0.7 | 3.54 ± 0.52 |
a1.4mg/kg and 2.8 mg/kg: groups that received 1.4 and 2.8 mg/kg/mL of the Achillea millefolium aqueous extract, respectively. The data represent the mean of neural spikes/bin size/50 stimulations. The bin size was set at 1 ms.
4.2. Effect of A. millefolium gavage on the temporal profile of the integration of responses
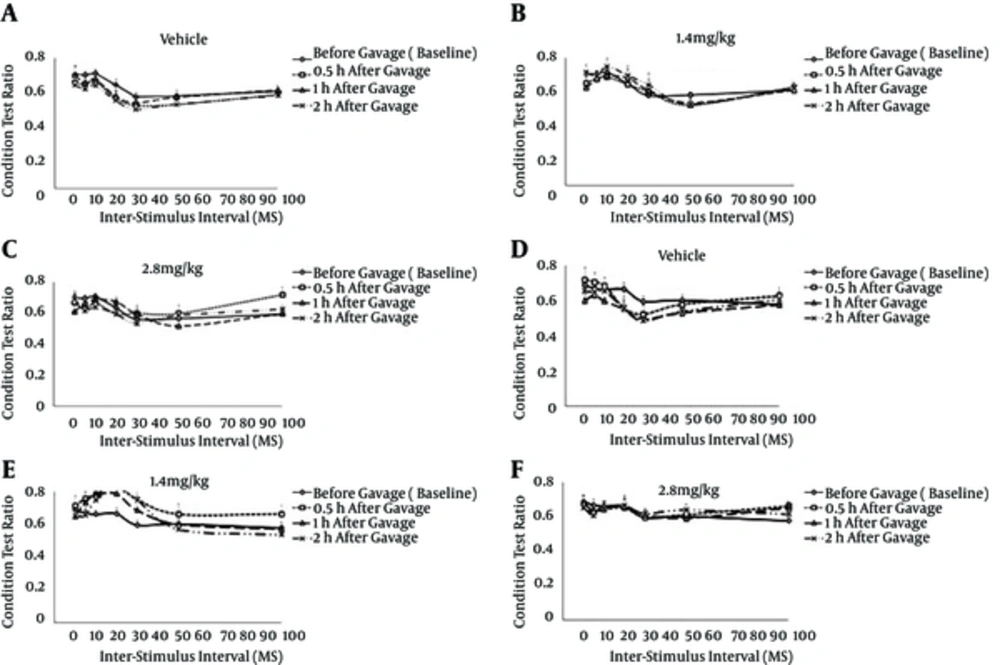
To investigate interactions between the vibrissae, a conditioning-test paradigm was used in which PW deflection was always preceded by AW deflection at ISIs of 0-, 5-, 10-, 20-, 30-, 50--, and 100 ms. As shown by the CTR ratios, acute oral administration (1.4 and 2.8 mg/kg doses) of the A. millefolium aqueous extract at different times (0.5, 1, and 2 hours) had no effect either on the ON or OFF responses (Figure 2).
The graphs plot the proportional change in the CTR as a function of the inter-stimulus interval. A CTR of > 1 indicates facilitation of the response to deflection of the first whisker, whereas a CTR of < 1 denotes suppression of the response to deflection of the second whisker. The error bars represent the S.E.M. The 1.4 and 2.8 mg/kg groups received 1.4 and 2.8 mg/kg/mL of the Achillea millefolium aqueous extract, respectively.
5. Discussion
Recently, many studies have focused on possible beneficial effects of A. millefolium on nervous system-related diseases. The use of A. millefolium as a treatment for such diseases raises important questions about possible side effects of this common plant on the nervous system. In our previous study, we reported that A. millefolium seemed to have no deleterious effects on tactile learning, as measured by a novel object-recognition test in rats (16). The focus of the present study was the effects of A. millefolium on neuronal responses to natural stimuli using the vibrissae pathway model in rats. The results demonstrated that oral gavage treatment with A. millefolium aqueous extract did not affect neuronal responses to the deflection of PWs and AWs. In addition, the treatment did not affect temporal aspects of response integration.
A. millefolium is one of the oldest-known herbal remedies and is used in many countries. Around 30 medicinal applications, including uses for wounds and skin problems, blood disorders, and digestive conditions, have been reported for A. millefolium (17). Its medicinal use as a general tonic has also been reported (17). The main form of A. millefolium is its aqueous extract. Experimental studies have used different doses of A. mellifolium extract (8 - 3000 mg/kg), but doses of 1.4 and 2.8 mg/kg are the most frequently employed (9). Thus, these doses were tested in the present study to enable comparisons with commonly used human doses.
A. millefolium-related toxicity is rare. Oral administration (10 g/kg) or intraperitoneal injections (3 g/kg) of A. millefolium aqueous extract caused no deaths in rats (18). Chronic administration of the extract also led to few adverse reactions in rats (18). In addition, administration of A. millefolium (2.8 g/kg/d for one week) to pregnant rats seemed to be well tolerated (19). In line with these results, the current study demonstrated that acute oral administration of aqueous A. millefolium extract had no deleterious effect on neuronal responses of the barrel cortex up to 2 hours after gavage.
Some reports have described toxicity-related side effects of A. millefolium, including contact allergic dermatitis after A. millefolium usage. However, according to one study, drying and processing A. millefolium seemed to decrease the concentration of sensitizing compounds (guaianolides) (20). In Drosophila, a study suggested that A. millefolium tea might have a weak genotoxic effect (21). Other studies indicated that high doses of both ethanol (22) and aqueous extracts (23) of A. millefolium interfered with spermatogenesis in rats.
5.1. Temporal Integration of Responses in the Barrel Cortex of Rats
During the exploration of their environment, rats use multiple whiskers to contact objects. The movements of the whiskers have a complex spatiotemporal pattern. The neuronal responses evoked by the stimulation of different whiskers are integrated in the barrel cortex, which plays an important role in processing sensory information on the external world (24). First, inputs from layer IV neurons are relayed to the superficial layers within a single column and then to the superficial layers of adjacent columns. Mapping the three-dimensional structural properties of objects depends on the pattern of activity across the cortical barrels (8, 25, 26). Previous studies showed that AWs evoked the inhibition in a time-dependent manner seemed to be necessary for the flow of inputs to superficial layers of the barrel cortex (8, 25). The results of the present study showed that oral gavage treatment with the A. millefolium aqueous extract did not affect inhibitory mechanisms evoked by AW stimulation in the IV layer. Thus, the type of activity that can pass through the IV layer to other barrel layers likely cannot be modulated by A. millefolium gavage. These findings are consistent with those of a behavioral study, which showed that the administration of A. millefolium aqueous extract did not affect tactile learning (16).
5.2. Facilitatory Interactions
The distribution of excitation within cortical receptive fields follows a columnar pattern (14). All driveable neurons respond maximally to one particular whisker called the PW. Other neighboring whiskers can also activate cells, but the strength of their excitation is usually less than that of the PWs. Thus, there are multiple whisker-receptive fields in the barrel cortex, with the largest and most numerous fields located in nongranular layers. These receptive fields are thought to be activated in part by convergence of excitatory inputs from single-whisker neurons in the relevant IV layer of the barrels (8, 13, 25). The results of this study demonstrated that gavage of A. millefolium aqueous extract did not affect the excitatory receptive fields in layer IV neurons.
5.3. Study Limitations
In this study, the data were obtained from neurons at a depth of 500 - 00 µm, which roughly corresponds to the location of layer IV neurons in the barrel cortex. However, most information processing occurs in other layers (24). Therefore, to better interpret the effect of the extract on neuronal responses of the barrel cortex, future studies should collect data from different depths in the barrel cortex.
In addition, this study did not distinguish between neurons in barrels and those in the septum in layer IV. Previous studies demonstrated that the receptive field of septal neurons involved multiple whiskers. They also showed that the deflection of evoked responses of PWs was as robust in septal neurons as in barrel neurons and that the latency of septal neuronal responses was similar to that of barrel neurons. Research also showed that septal neurons were excited by noise stimuli, irrespective of whether the stimuli were applied to PWs or AWs (27).
5.4. Conclusions
The results of this study demonstrated that acute oral administration of A. millefolium aqueous extract did not modulate excitatory and inhibitory receptive fields in the barrel cortex of rats. These results are consistent with those of a behavioral study, which showed that A. millefolium extract did not affect tactile learning (16).

![Population peristimulus time histograms (PSTHs) of ON responses to principal whisker (PW) deflection in the different groups (vehicle [n = 14 units], 1.4 mg/kg extract [n = 14 units], and 2.8 mg/kg extract [n = 14 units]). Each panel shows the accumulated responses to 50 deflections of the PWs. All the PSTHs are plotted on the same scale bar, where time 0 corresponds to the time of whisker deflection and time -10 shows the responses 10 ms before the start of deflection. The insets present typical examples of PSTHs (bin size = 1 ms), showing ON and OFF neuronal responses to 50 PW deflections. The whisker deflection paradigm is depicted in lines below each trace. NS; normal saline. Population peristimulus time histograms (PSTHs) of ON responses to principal whisker (PW) deflection in the different groups (vehicle [n = 14 units], 1.4 mg/kg extract [n = 14 units], and 2.8 mg/kg extract [n = 14 units]). Each panel shows the accumulated responses to 50 deflections of the PWs. All the PSTHs are plotted on the same scale bar, where time 0 corresponds to the time of whisker deflection and time -10 shows the responses 10 ms before the start of deflection. The insets present typical examples of PSTHs (bin size = 1 ms), showing ON and OFF neuronal responses to 50 PW deflections. The whisker deflection paradigm is depicted in lines below each trace. NS; normal saline.](https://services.brieflands.com/cdn/serve/3170b/c981911cbe795f6251d81b163e4213db27b948e0/jjnpp-In_Press-In_Press-37758-i001-preview-preview.webp)