1. Background
Pulmonary fibrosis is a common disease with high mortality because of damage to the lung parenchyma by inflammatory factors that ultimately lead to fibrosis (1-3). Chemotherapy with BLM and therapy with amiodarone as an anti-arrhythmic agent often result in lung injury with fibrosis (4, 5). In addition, environmental factors, such as exposure to asbestos and silica and toxins, such as paraquat, are usually associated with significant injury of lung and pulmonary fibrosis (6). Usually, inflammatory variable factors during lung injury, such as TGF-β, Macrophage Inflammatory Protein-1 (MIP-1), TNF-α, and absence of the PPAR-α receptors, do not respond to anti-inflammatory treatment (7-11). In addition, free radicals formation plays a key role in disease pathogenesis (12, 13). The main strategies for remedy of patients is improvement of parenchymal cells and maintains normal function by reducing free radicals and inhibition of inflammatory processes (14). Previous studies have reported that the ideal use of compound with antioxidant property, such as lycopene and curcumin, are effective on normalizing the level of anti-oxidant in amiodarone and BLM-induced inflammation (5, 15). There is little data regarding the role of the Peroxisome Proliferator-Activated Receptors Alpha (PPAR-α) in lung fibrosis (16). Furthermore, GEM as a fibric acid derivative lipid lowering agent and PPAR-α receptor agonist, has some effects other than in the treatment of hyperlipidemia (17-20). In previous studies, it has been well confirmed that GEM has antioxidant and anti-inflammatory properties through effects on TGF-B, TNF-α, and PPAR-α (21, 22).
2. Objectives
Taken together, GEM could be a good strategy for the prevention and treatment of pulmonary fibrosis. This study was conducted to examine the effects of GEM in an animal model of lung injury and fibrosis.
3. Methods
3.1. Animals
The male Sprague-Dawley rats weighting 180 to 200 grams were prepared from the research center and experimental animal house of Jundishapur University of Medical Sciences, Ahvaz. The animals were housed in stainless cages with room temperature of 21 to 24°C humidity of 45% to 55% and 12 hour light-dark cycle during the study time course. They were fed rodent standard food with free access to water. All procedures were approved by the animal care committee of the Jundishapur University of Medical Sciences (number: IR.AJUMS.REC.1394.659), Ahvaz, Iran.
3.2. Preparation of GEM
Gemfibrozil was obtained from Dr. Abidi Pharmaceutical manufacturing company (Tehran, Iran). The GEM solution was prepared by wetting of GEM in minimum amount of glycerin and carefully levigating in mortar by pestle with the addition of water. The GEM doses were prepared based on serial dilutions. Administration of GEM was done 3 days before induction of lung damage and fibrosis and continued for 1 or 3 weeks.
3.3. Experimental Design
In the study, the researchers evaluated both phases of lung damage consisting of inflammation (7th day) and fibrosis (21th day) after BLM, Intratracheally (IT). Accordingly, experiments were conducted in 2 different time periods, 1 and 3 weeks in duration. In each time period, thirty rats were divided to 5 groups (n = 6). The animals were pretreated by vehicle or GEM (25, 50, 100 mg/kg), orally. After 3 day, the groups received single normal saline or BLM (IT) for induction of lung fibrosis.
3.4. Induction of Pulmonary Fibrosis with Bleomycin
Anesthetized animals were laid on a slanted board and hanged through upper canine teeth. A single dose of 5 IU/kg of BLM was diluted in saline (1 mL/kg) and was instilled in the throat. Subsequently, swallowing and breathing were blocked to be completely aspirate BLM into the lung. To ensure of drug sector in all parts of the lung, the animals were laid down and after full consciousness, transferred to cage (23, 24).
3.5. Lung Indices
Lung index was calculated based on the ratio of lung weight to body weight in both inflammatory (1 week) and fibrotic (3 weeks) phases.
3.6. Macroscopic Analysis
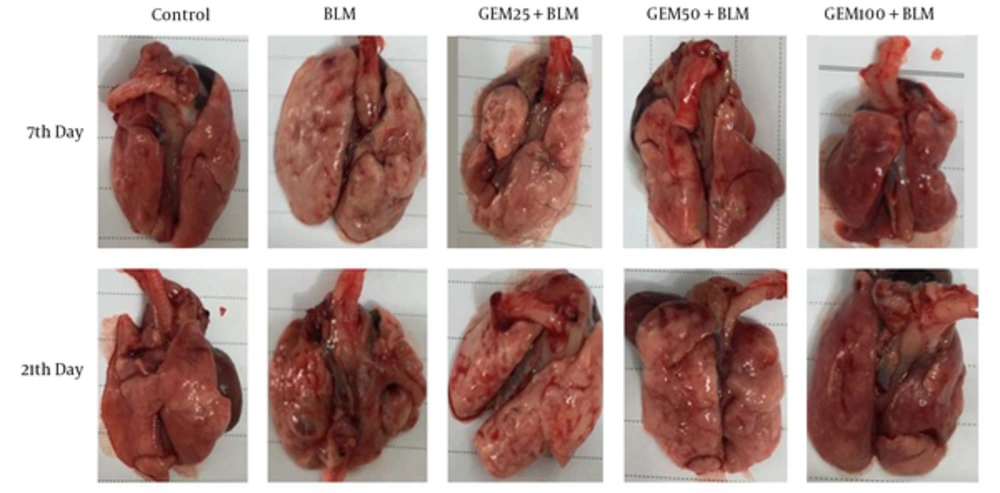
For macroscopic analysis, the lungs appearance was compared based on lung swelling, tissue discoloration, hemorrhagic lesions, and elasticity.
3.7. Measurement of Collagen
The paraffin-embedded tissue sections were deparaffinized and then impregnated with dye solution and dye extraction, including the Sirius Red/ Fast Green Collagen Staining kit (Chondrex, Inc.). Optical density of lung tissue samples was recorded and collagen content was measured through the following equation:
Collagen (µg/section) = (OD 540 value - (OD 605 value × 0.291))/0.0378
3.8. Histopathological Study
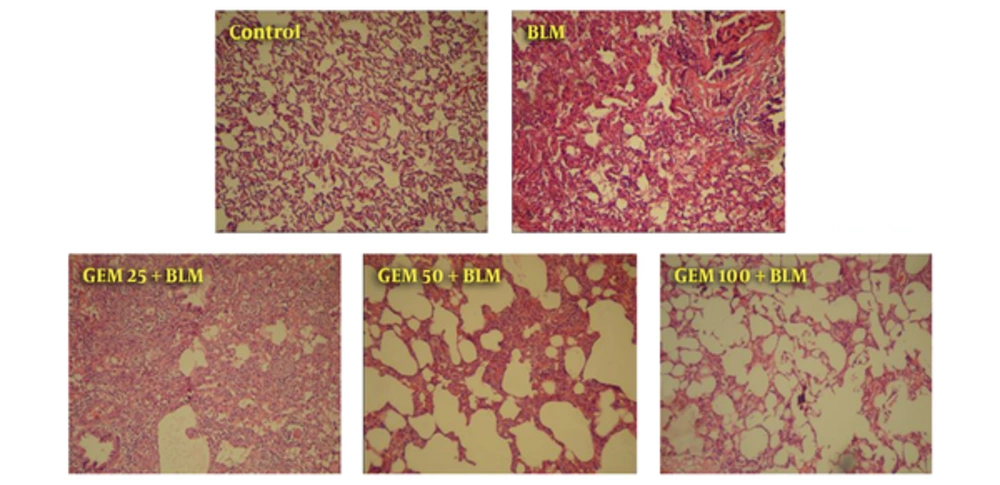
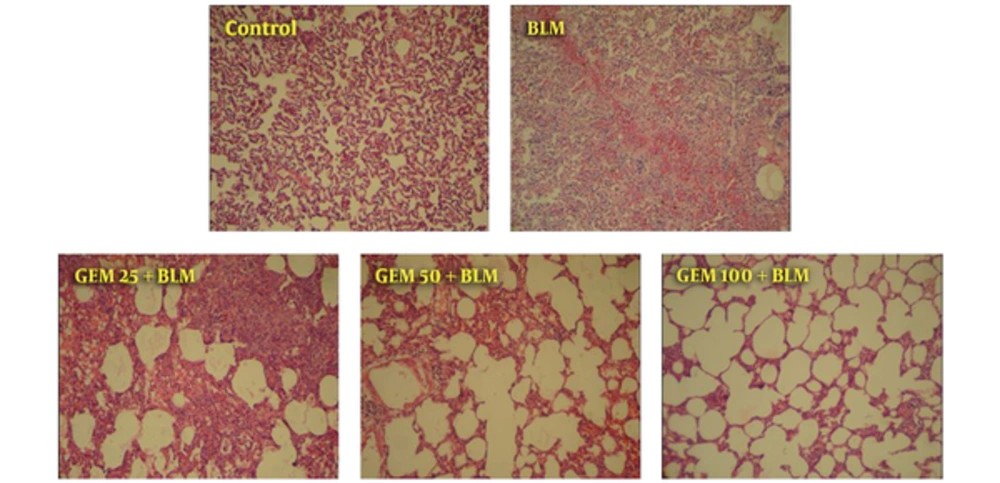
Histological studies were performed to evaluate changes caused by inflammation on the 7th day and fibrosis on the 21st day after BLM instillation. After fixation with 10% formalin and preparation of 5 µm tissue sections by rotary microtome, Hematoxylin and Eosin (H and E) staining was performed for evaluation of inflammation and fibrosis.
3.9. Statistical Analysis
All data are reported as mean ± SD. Statistical analysis was performed by the t-test and one-way analysis of variance (ANOVA) followed by LSD test, with the SPSS software. P values of < 0.05 were considered as statistical significance.
4. Results
4.1. Lung Index
The evaluation of lung index indicates that there was a slight effect in the first phase on the 7th day, so that only a dose of 50 mg/kg of GEM resulted in a significant difference (P < 0.05), while at doses of 25 and 100 mg/kg, reduction of lung index was not significant (Table 1). The results also showed that the level of lung index in the BLM untreated group during the second phase was increased, yet the lung index in the animals treated by GEM 100 mg/kg was reduced at the end of the second phase compared to the BLM group (P < 0.05).
| Groups, N = 6 | Lung Index Level, mg lung/g BW | |
|---|---|---|
| Inflammatory Phase (7th Day) | Fibrotic Phase (21th Day) | |
| Control (Vehicle + Saline) | 8.08 ± 0.59 | 6.02 ± 0.47 |
| Vehicle + BLM | 10.28 ± 1.5 | 11.93 ± 0.81 |
| GEM 25 mg/kg + BLM | 9.76 ± 0.82 | 11.26 ± 0.34b |
| GEM 50 mg/kg + BLM | 8.96 ± 0.87b | 9.68 ± 0.45b |
| GEM 100 mg/kg + BLM | 9.90 ± 1.4 | 9.69 ± 0.59b |
The Effect of Gemfibrozil on the Lung Index Level (Lung Weight to Body Weight Ratio) (Both Phase) in Rats with BLM-Induced Pulmonary Damagea
4.2. Macroscopic Assessment
Macroscopic observation showed that BLM administration led to brightness and loss of tissue color, hemorrhagic lesions, and loss of tissue elasticity. Administration of GEM produced increased tissue color and elasticity and decreased hemorrhagic damage (Figure 1).
4.3. Collagen Content
As shown in Table 2, when the animals were treated by different doses of GEM, significant differences in the level of collagen (both phase) was observed compared to the BLM group (P < 0.05). The comparison of mean values showed that treatment by GEM reduced collagen level on the 7th day and 21st day of the experimental periods. In addition, the researchers also found that there was a greater reduction in the group treated by GEM 100 mg/kg (approximately 33.5% and 61% in both phase, respectively). It was well known that these effects were dose-dependent (Table 2).
| Groups (N = 6) | Collagen Level, µg/tissue section | |
|---|---|---|
| Inflammatory Phase (7th Day) | Fibrotic Phase (21th Day) | |
| Control (Vehicle + Saline) | 5.20 ± 0.11 | 4.96 ± 0.10 |
| Vehicle + BLM | 11.23 ± 0.80 | 23.83 ± 0.22 |
| GEM 25 mg/kg + BLM | 9.70 ± 0.47b | 14.73 ± 0.42b |
| GEM 50 mg/kg + BLM | 8.40 ± 0.23b | 11.66 ± 0.13b |
| GEM 100 mg/kg + BLM | 7.46 ± 0.37b | 9.29 ± 0.16b |
The Effect of Gemfibrozil on the Collagen Level (Both Phase) in Rats with BLM-Induced Pulmonary Inflammation and Fibrosisa
4.4. Histopathological Observations
Histopathology evaluation of lung tissue on the 7th and 21st day showed that BLM resulted dramatic change, such as inflammatory cells infiltrates, decreased alveolar spaces, alveolar walls thickening, and increase of fibroblasts and widening of pulmonary interstitial. These findings represent inflammation and fibrosis. However, observations on the 7th day indicate inflammatory events with slightly fibrotic lesions and on the 21st day show the fibrosis without the presence of inflammatory cells (Figures 2 and 3). The current evaluation showed that treatment with GEM 25 mg/kg did not dramatically effect changes induced by BLM and tissue sections were similar to the BLM group. Although dose of 50 mg/kg of GEM led to an increase of alveoli space, the improvement was also clear. The best effect was found in the group treated by 100 mg/kg GEM, so that alveolar space increased and wall thickening of some lung parts was slight. After 21 days (second phase), the researchers found visible changes, such as increase in fibrotic cells, tissue necrosis resulted from BLM, along with deterioration of induced changes during the first phase. In the group treated by 25 mg/kg of GEM, the changes were improved yet still showed fibrosis. Data confirmed that the animals treated by 100 mg/kg of GEM had the most similarity with the control animals, howbeit, there were slight changes, yet improvement was perfectly obvious (Figures 2 and 3).
5. Discussion
Thus study presents new findings on the effects of GEM on pulmonary fibrosis. Bleomycin induces pulmonary fibrosis and tissue damage in animal models through oxidant-antioxidant imbalance and hydroxyl and superoxide radical production (12, 14, 25, 26). Accordingly, BLM is widely used for induction of pulmonary fibrosis (27, 28). Here, the study found that BLM resulted in dramatic changes, such as destruction of alveoli, thickening of lung wall, increment of lung interstitial fibroblast, and development of fibrosis. These changes start with inflammation that leads to the accumulation of collagen and ultimately fibrosis. It was shown that changes of collagen are a clear indication of fibrosis (29-31). The models of BLM or amiodarone-induced pulmonary fibrosis have determined increased hydroxyproline, which represents the destruction of tissue (32, 33). The current results confirmed the useful effect of GEM on pulmonary damage and fibrosis and it was clear that GEM at a dose of 100 mg/kg had a greater effect. Imbalance of oxidant-antioxidant agents occurs under conditions of pulmonary fibrosis, which is probably a major cause of tissue damage. One of the effects of free radicals increment is destruction of membrane lipids and MDA level. The measurement of MDA determines the amount of tissue damage (34-37). The protective mechanisms of vitamin E against amiodarone-induced pulmonary toxicity has been shown. Results have shown that vitamin E administration decreases the high level of TGF-β1 and hydroxyproline, which leads to reversal tissue destruction (33). In relation to antioxidant effects, the past results are similar to the current study. It has been reported that antioxidants, such as curcumin, could improve oxidative status in pulmonary fibrosis (38). In addition, it was shown that curcumin, as an antioxidant compound, was inhibited by the development pulmonary fibrosis, which is a good reason for its use (5). Green tea extract administration is effective in pulmonary fibrosis by reduction of MDA and hydroxyproline levels and endothelin expression (39). In an animal model of liver fibrosis, it has been recognized that decreased PPAR-α and increased TGF-β expressions was associated with development of hepatic fibrosis (40). Moreover, lycopene improved the pathological changes of lung tissue and MDA level, which is probably by inhibition of oxidative stress (15). Previous studies have shown that antioxidant and anti-inflammatory properties of GEM could be mediated by several pathways. It has been reported that GEM effects could be mediated through PPAR-α dependent and independent pathways and it has been emphasized that anti-oxidative, immunomodulatory, and anti-inflammatory activities of GEM are not PPAR-α dependent (20). It has been reported that GEM resulted in reduction of TNF-α and interleukin 6, and also decreased the expression of NADPH oxidase. In addition, it is known as an antioxidant and anti-inflammatory agent due to the inhibition of NF-κB activation and increase of PPAR-α activation (21, 22, 41-43). In conclusion, this study showed that GEM could alleviate inflammatory and fibrotic effects of BLM and these effects may be mediated through anti-oxidative properties, anti-inflammatory effects, and activation of PPAR-α receptors.
5.1. Conclusion
According to this line of antioxidant compounds in the current study, it could be suggested that GEM, by normalizing the levels of antioxidants, prevents tissue damage under conditions of BLM-induced pulmonary fibrosis. However, the researchers suggest further studies for understanding the exact mechanism of GEM and its possible side effects.