1. Background
Idiopathic pulmonary fibrosis (IPF) as a fibroproliferative lung disorder with unknown etiology is characterized by significant oxidative injury, massive collagen deposition, lung remodeling, and vigorous fibrotic lesions. The current treatment strategies cannot effectively ameliorate IPF. The lung failure is progressed and eventually, death occurs within 3 - 5 years after diagnosis (1).
A powerful and reproducible animal model of IPF is produced by bleomycin (BLM) as a cytotoxic antibiotic. The most prevalent mechanism suggested for the pathogenesis of pulmonary fibrosis (PF) is that bleomycin induces DNA-breakage and oxidative stress leading to apoptosis and obliteration of alveolar epithelial cells. These events lead to the recruitment of leukocytes, overproduction of free radical species, and abnormal secretion of inflammatory and fibrotic mediators (such as IL-1β, TNF-α, and TGF-β). Then, collagen deposition and lung remodeling are progressed by myofibroblast activation (2). The TGF-β signaling pathway and mitogen-activated protein kinases (MAPKs) signaling are involved in activating myofibroblasts (3). However, previous studies have shown that antioxidant and anti-inflammatory agents have protective effects against IPF (4).
Zingerone, an active non-volatile ingredient of ginger (Zingiber officinale), has been known as a multi-target agent, displaying various pharmacological effects. For instance, zingerone exhibits antioxidant activity equal to ascorbic acid activity and can attenuate oxidative injuries in the lung (5), liver (6), and kidney (7). The protective effects of zingerone against lung injury induced by lipopolysaccharide (LPS) have been reported by suppressing neutrophil influx and production of proinflammatory cytokines in mice. The protective effects of zingerone are attributed to the inhibition of the signaling pathways of nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinases (MAPKs) (8). In addition, zingerone reduces inflammatory and fibrotic responses in various experimental animal models of diseases accompanied by the decrease of TGF-β levels (6, 9, 10).
2. Objectives
Despite evidence of zingerone anti-fibrotic activity in different animal models, there is no study of its effect on pulmonary fibrosis. Therefore, the present study was conducted to evaluate the probable beneficial effects of zingerone against bleomycin-induced PF and underlying mechanisms.
3. Methods
3.1. Chemicals
Zingerone and bleomycin sulfate were purchased from Sigma-Aldrich (St. Louis, MO, USA) and Nippon Kayaku Co., Ltd., respectively. All other reagents were of analytical grade. A fresh concentration of drugs was prepared immediately before use.
3.2. Animal
Healthy male Sprague-Dawley rats (200 - 250 g) were obtained from the animal house and research center of Jundishapur University of Medical Sciences, Ahvaz, Iran. They were kept in standard conditions with free access to water and rodent laboratory chow ad libitum. The experimental design was approved by the Institutional Animal Ethics Committee of Jundishapur University of Medical Sciences (IR.AJUMS.REC.1395.150) and the experiments were conducted according to the NIH Guide for Care and Use of Laboratory Animals.
3.3. Induction of Pulmonary Fibrosis
The PF model was induced in accordance with previous studies (11). After the rats were anesthetized with intraperitoneal injection of ketamine (75 mg/kg) and xylazine (5 mg/kg), bleomycin (5 mg/kg body weight in 0.25 mL saline) was instilled intratracheally. The vehicle group received equal amounts of 0.9% saline instead of bleomycin.
3.4. Experimental Design
The optimal dose of bleomycin (5 mg/kg) was obtained from a preliminary study that led to proper pulmonary fibrosis without affecting survival. Thirty-six rats were randomly assigned to the following groups: I (saline + saline), II (bleomycin (BLM) + vehicle), III (BLM + zingerone 10 mg/kg), IV (BLM + zingerone 20 mg/kg), V (BLM + zingerone 40 mg/kg), VI (saline + zingerone 40 mg/kg). Zingerone was dissolved in saline and administrated by gavage for 21 consecutive days after intratracheal bleomycin administration. The route of administration, doses of zingerone, and the sample size were based on previous studies (8, 12). The body weight of the rats was evaluated weekly during the experiment. At the end of the experiments, the rats were euthanized and the whole lung was dissected out, washed with saline, and weighed. The lung index was calculated by the ratio of the lung wet weight (mg) to body weight (g).
3.5. Bronchoalveolar Fluid and Lung Sample Collection
By cannulating the trachea with a blunt needle, the airways were washed four times with 5mL of phosphate-buffered saline (PBS). Then, cell suspensions were centrifuged and the cell pellet was suspended by hypotonic lysis using 1% glacial acetic acid. Total leukocytes and differential cell numbers were counted by using hemocytometer and Wright-Giemsa staining (4). Besides, the left lung was used for histological examination and right lung tissue was homogenized in cold radioimmune precipitation assay (RIPA) lysis buffer to make a 10% homogenate (w/v). After centrifuging at 11180 g for 10 min at 4°C, the supernatants were maintained at -80°C for subsequent assessment. The total protein content of tissue homogenates was estimated by the Bradford method.
3.6. Hydroxyproline Measurement
The hydroxyproline content of lung tissue was measured in accordance with the method described by Edwards and O’Brien (13). The absorbance was read at 550 nm. The hydroxyproline content was calculated as mg/g lung tissue.
3.7. Estimation of Malondialdehyde and Glutathione
The malondialdehyde (MDA) content of lung tissue homogenates was determined in accordance with the TBA method (14). The reaction of thiobarbituric acid with MDA at high temperature and low pH leads to the formation of a red fluorescent complex. The absorbance was determined at 532 nm. The results were given in nmol/g tissue protein. The glutathione (GSH) level of lung tissue was measured by the method previously described by Ellman (15). The values were expressed as nmol/mg protein.
3.8. Determination of Superoxide Dismutase and Glutathione Peroxidase Activities
The glutathione peroxidase (GPx) and superoxide dismutase (SOD) activity was measured using a commercial RANSOD and RANSEL kits (Randox Labs, Crumlin, UK), according to the manufacturer’s instruction. The activities of GPx and SOD were expressed as IU/mg tissue protein.
3.9. Histopathological Studies
Buffered formaldehyde (10%) was used to fix the left lung tissue. Then, the tissue specimens were embedded in paraffin and were cut into 5-µm sections and stained with H&E (hematoxylin and eosin) and Masson’s trichrome. A single-blind examination was done by an experienced pathologist to score fibrosis in accordance with the modified numerical Ashcroft’s scale (0 - 8) (16).
3.10. Statistical Analysis
The statistical analysis was performed using PRISM version 5.01. The scores of fibrosis were analyzed by the Kruskal-Wallis test and other variables by the Two-way ANOVA or One-way ANOVA, followed by an appropriate post hoc test. Data were expressed as means ± SEM. P < 0.05 was considered as statistical significance.
4. Results
4.1. Effect of Zingerone on Body Weight and Lung Index
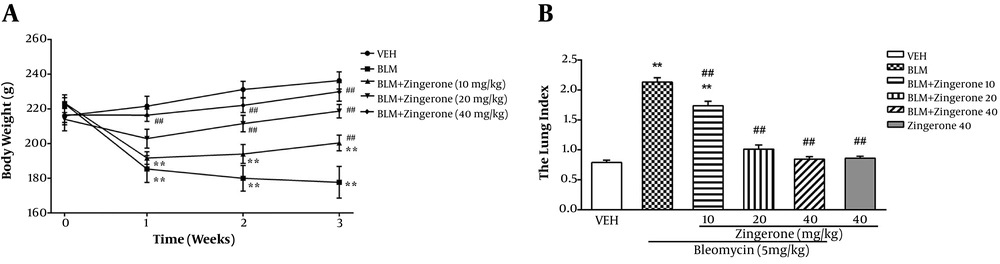
Bleomycin caused a significant body weight loss in exposed rats compared to the vehicle group at weeks 1, 2, and 3 (P < 0.0001). Zingerone (20 and 40 mg/kg) significantly increased weight gain compared to the bleomycin group during the experiments (group effect P < 0.0001, time effect P = 0.0002, and interaction effect P < 0.0001 in Two-way ANOVA) (Figure 1A).
Effect of zingerone (10 - 40 mg/kg/day, orally) on PF induced by bleomycin (5 mg/kg, intratracheally) on body weight at weeks 1, 2, and 3 A, (Two-way ANOVA followed by Dunnett’s test); and on lung index B, (One-way ANOVA followed by Tukey’s test) in the rat model of PF. Data are expressed as means ± SEM, (n = 6). ** P < 0.05 compared to the vehicle group; ## P < 0.05 compared to the BLM group.
Bleomycin significantly increased the normal lung index on day 21. Zingerone treatment (20 and 40 mg/kg) significantly decreased the lung index compared to the bleomycin group (P < 0.0001) (Figure 1B).
4.2. Effect of Zingerone on Bronchoalveolar Fluid Cells
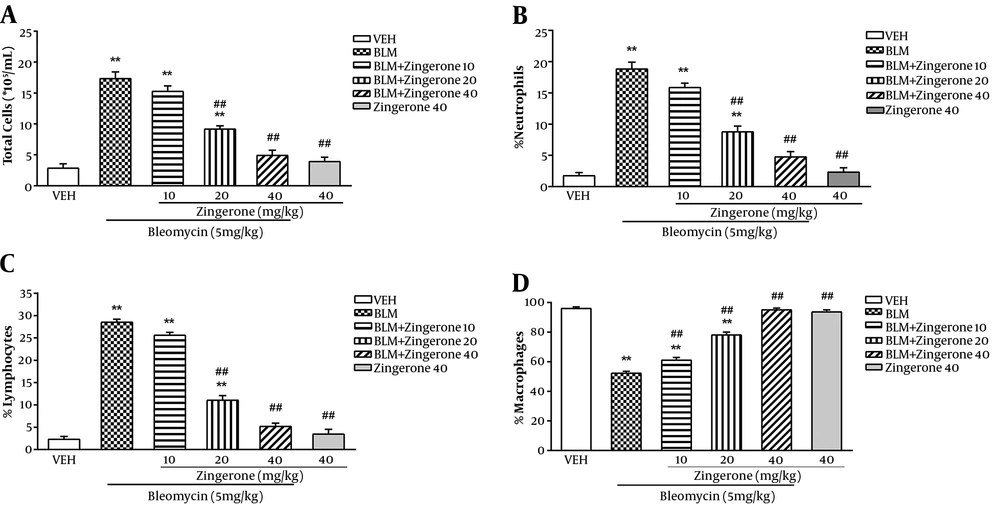
BLM significantly increased the normal total leukocyte count in BLAF (P < 0.01). Zingerone treatment (20 and 40 mg/kg) significantly reduced the total leukocyte number (P < 0.01) (Figure 2A). BLM significantly increased the normal neutrophil and lymphocyte proportions and significantly decreased the macrophages proportion (P < 0.01). Zingerone (20 and 40 mg/kg) significantly reversed the differential cell percentage changes in bronchoalveolar fluid (BALF) as compared to the BLM group (P < 0.01). The total cell number and differential percentage of leukocytes were not significantly different between the zingerone-treated group (10 mg/kg) and the BLM group (Figure 2B-D).
Effect of zingerone (10 - 40mg/kg/day, orally) on total cell count (A) percentages of neutrophils (B), lymphocytes (C) and macrophages (D) in BALF in bleomycin (5 mg/kg, intratracheally) rat model of PF. Data are expressed as means ± SEM, (n = 6). **P < 0.05 compared to the vehicle group; ##P < 0.05 compared to the BLM group. One-way ANOVA followed by Tukey’s test.
4.3. Effect of Zingerone on Oxidative Markers of Lung Tissue
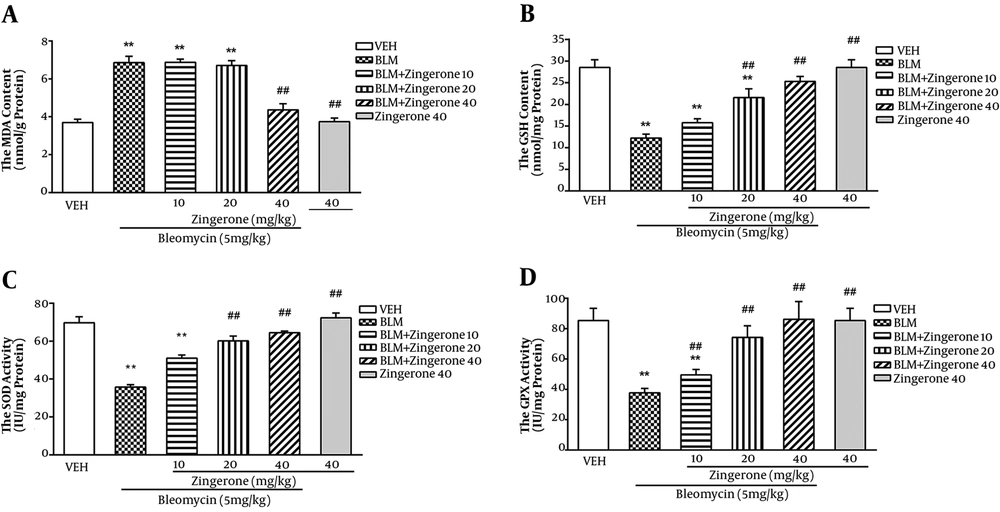
Bleomycin significantly enhanced the normal lung MDA level (P < 0.01) and only did zingerone (40 mg/kg) significantly reduce the MDA group compared to the BLM group (P < 0.01) (Figure 3A). Bleomycin significantly decreased the tissue GSH content and the activity of SOD and GPx as compared to the vehicle-treated group (P < 0.01). Meanwhile, zingerone (10 - 40 mg/kg) significantly enhanced the GSH concentration and activity of SOD and GPx as compared to the BLM-group (P < 0.01) (Figure 3B-D).
Effect of zingerone (10 - 40 mg/kg/day, orally) on lung tissue levels of MDA (4A), GSH (4B) and activities of SOD (4C) and GPx (4D) in bleomycin (5 mg/kg, intratracheally) rat model of PF. Data are expressed as means ± SEM, (n = 6). **P < 0.05 compared to the vehicle group; ## P < 0.05 compared to the BLM group. One-way ANOVA followed by Tukey’s test.
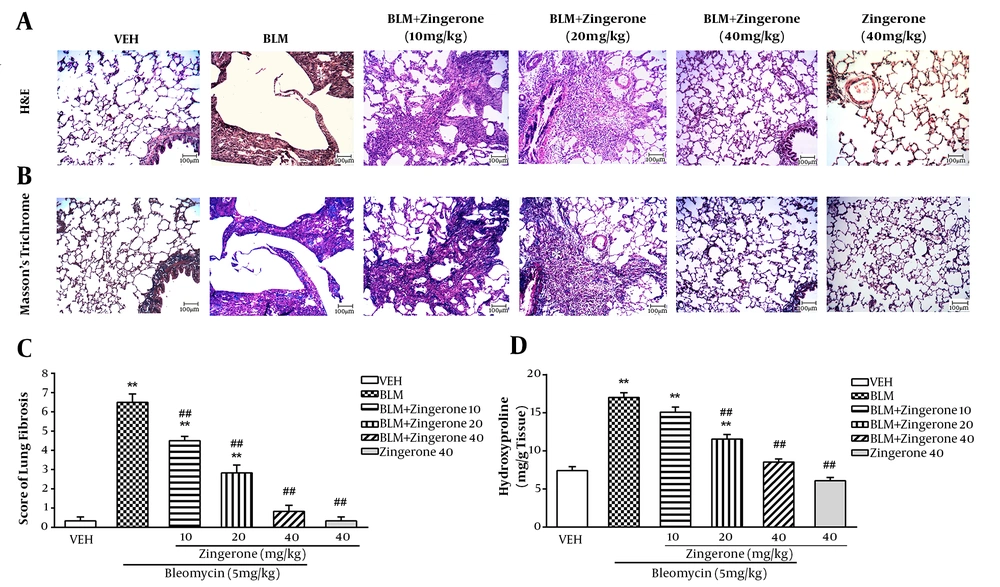
4.4. Effect of Zingerone on Histopathological Lesions and Lung Hydroxyproline
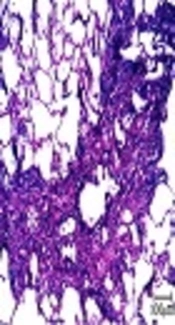
The evaluation of H&E and Masson's trichrome-stained sections revealed that the normal lung architecture was destroyed following bleomycin-instillation and a fibrotic pattern was observed (Figure 4A and B). BLM significantly increased the modified Ashcroft’s scale and hydroxyproline content (P < 0.01) (Figure 4C and D). Zingerone (10 - 40 mg/kg) dose-dependently decreased the fibrosis score as compared to the BLM group (P < 0.01). Zingerone (20 and 40 mg/kg) significantly reduced lung hydroxyproline as compared to the BLM group (P < 0.01), but zingerone (10 mg/kg) had no significant effect on hydroxyproline in BLM-instilled rats.
Effect of zingerone (10 - 40 mg/kg) on BLM-induced fibrosis shown in representative images of H&E (A) and Masson’s trichrome-staining (B) (scale bars: 100 µm, white star: Fibrotic foci) of rat lung sections on day 21. The dose-response of fibrosis changes (Kruskal-Wallis followed by Dunn’s test) (C) and hydroxyproline content (One-way ANOVA followed by Tukey’s test) (D) in different groups is shown. Data are expressed as means ± SEM, (n = 6). **P < 0.05 compared to the vehicle group; ## P < 0.05 compared to the BLM group.
5. Discussion
Idiopathic pulmonary fibrosis is a deadly and progressive lung disorder. Despite numerous studies, a few drugs are available to treat this disorder (17). The bleomycin-induced animal model of IPF is widely used to investigate new anti-fibrotic compounds (18). Several agents with potent antioxidant and anti-inflammatory activities exhibit protective effects against bleomycin-induced lung injury (4, 19). Zingerone, as an interesting active ingredient of ginger (Zingiber officinale), exhibits potent antioxidant, anti-inflammatory, and anti-fibrotic activities in various animal models of disease (5-8, 20). However, its effect on pulmonary fibrosis has not been reported. For the first time in the present study, the possible ameliorative effect of zingerone against PF induced by bleomycin was determined in rats.
Our findings showed that zingerone clearly attenuated fibrotic lesions in the bleomycin group. The body weight loss, lung index, and total and differential cell numbers of BLAF, histopathological scores of fibrosis, and lung hydroxyproline content were significantly reversed by zingerone in a dose-dependent manner. In addition, zingerone reversed the GPx and SOD activity, as well as GSH and MDA levels, near to normal values in the vehicle group. In addition, zingerone solely did not have a significant effect on the measured variables compared to the vehicle group.
The level of MDA, a lipid peroxidation by-product, mirrors the oxidative cell membrane damage that its level increases in the lung injury by bleomycin (19, 21). The tissue glutathione content and SOD and GPx enzymes as the most important antioxidant defenses play key roles in protection against oxidative stress caused by BLM (22, 23). In agreement with previous studies, our results showed that bleomycin markedly increased the levels of MDA in lung tissue. Meanwhile, SOD and GPx activities, as well as the GSH level, significantly reduced in bleomycin-instilled rats (24, 25). However, zingerone treatment significantly decreased the MDA level and improved the SOD and GPx activities, as well as, inhibited GSH depletion. The antioxidant effect of zingerone has been shown in other models of diseases including hepatic injury (6) and renal damage (7). Rao et al. showed that the oral administration of zingerone reduced the MDA content and elevated the SOD and GSH activity in gamma-radiated mice (26). Therefore, the ameliorative effect of zingerone against pulmonary fibrosis may be due to its potent antioxidant nature.
The influx of inflammatory cells in BALF as a lung inflammatory hallmark is exacerbated in pulmonary fibrosis. In addition, the percentage of lymphocytes and neutrophils increases while the macrophage proportion decreases in BALF in BLM groups (27, 28). The activated neutrophils and macrophage can produce toxic ROS and proteolytic factors, possibly worsening tissue damage (29). The same pattern of leukocytes infiltration into BALF was observed in the present study in bleomycin-instilled rats. However, zingerone treatment ameliorated these pathological changes in BALF. In agreement with our results, Xie et al. reported that zingerone suppressed neutrophil influx into BALF, alveolar hemorrhage, and pulmonary edema induced by lipopolysaccharide (8). Overall, our findings indicated that the protective effect of zingerone might be due to the attenuation of leukocytes recruitment into the lung.
The histopathological examination as a gold test is widely utilized to evaluate the efficiency of new anti-fibrotic drugs (30). Our findings support previous studies that bleomycin induces collagen deposition and lung fibrotic lesions (31). However, zingerone significantly reduced lung index and collagen deposition, as well as pulmonary fibrosis, in BLM-instilled rats. These results are consistent with other studies indicating that zingerone could improve inflammation and fibrosis via blocking the oxidative stress, as well as inhibiting the NF-ҡB and MAPKs activation, in various animal models such as hepatic and cardiac fibrosis (6, 10).
Taken together, zingerone exerts a protective effect against bleomycin-induced PF. Our findings suggested that zingerone exhibits antioxidant and anti-fibrotic activity and might be a novel therapeutic agent for the treatment of IPF. However, more investigations are needed to characterize the anti-fibrotic mechanism of zingerone.