1. Background
Breast cancer is the second reason for cancer-related death in women worldwide (1). In 2016, a total of 246,660 new cases of breast cancer and 14% of deaths due to breast cancer were reported in the US (2). Chemotherapy is considered as the most frequent systemic treatment for suppressing the tumor cell proliferation and subsequent disease progression and metastasis. However, chemotherapeutic agents not only kill tumor cells, but also deleteriously affect normal cells. Therefore, anti-cancer drug carriers characterized by targeted cancer therapy on cancer cells without affecting normal cells to maintain or improve the efficacy of drug delivery and reduce the burden of adverse reactions and side effects are urgently needed. To address this issue, a promising approach is using drug-delivery systems, of which nanosized drug-carrier systems have especially been interested (3). NPs, due to the different biological properties and applicability in a wide range of settings, provide a safe and effective platform for targeted-delivery systems of chemotherapeutic agents. These systems are classified into two main groups: polymeric nanoparticles and lipid nanoparticles (LNPs). LNPs, due to their natural and biological origins, are less toxic than polymeric NPs. Nanostructured lipid carriers (NLCs) are the second generation of LNPs after solid LNPs, which benefit from the advantages of both nanoemulsion and solid NPs. They can also be easily synthesized with oil-in-water nanoemulsion (4).
In targeted therapy, the development of magnetically guided drug delivery systems has widely considered (3), since it allows directing magnetic NPs to be concentrated at a target region by a simple external magnetic field. The characteristics, such as biocompatibility, non-toxicity, small size, magnetization, a high degree of saturation, and ease of appropriate surface modification with different polymers have made superparamagnetic iron oxide nanoparticles (SPIONs), especially those containing magnetite (Fe3O4), as the best choice for site-specific drug delivery systems (3).
Several studies have recently evaluated magnetic NPs loaded with different drugs (5, 6). For example, Cicha et al. (7) evaluated mitoxantrone-carrying SPIONs and the drug released from SPIONs. Mancarella et al. (8) developed a layer-by-layer functionalization of Fe3O4 NPs by coating them in dextran and poly-L-lysine. The results showed a high percentage of curcumin loaded Fe3O4 NPs for treating ovarian cancer (8).
Curcumin is a natural hydrophobic diphenol derived from the rhizomes of plant Curcuma longa. Numerous studies have confirmed the potential of curcumin in prevention and treatment of various cancer types, such as head and neck, melanoma, colon, breast, pancreatic, prostate, and ovarian cancers. The multiple molecular targeting of curcumin to selectively kill tumor cells with low intrinsic toxicity has been demonstrated in different studies (9-11). However, the properties, such as a high hydrophobicity, instability, and poor bioavailability limit its further clinical use (12). Therefore, there is a need for finding new strategies to improve the physicochemical properties and therapeutic efficacy of curcumin. Curcumin-loaded magnetic NLCs, along with oleic acid as a stabilizer, may provide a novel approach for use in the treatment of different cancers (13).
2. Objectives
The aim of the present study was to synthesize the SPIONs and combine them with NLC for the curcumin entrapment and evaluate their features in human breast carcinoma cell line. In our knowledge, this is the first study, which combined the features of NLC and SPIONs to targeted delivery of curcumin.
3. Methods
3.1. Material
Curcumin, FeCl3 and FeCl2 were purchased from the Merck Chemicals (Germany). Cetylpalmitate was received as a gift from the Alborz Bulk Pharmaceutical Co. Fish oil was purchased from the Daana Pharma Co. (Tehran, Iran). In addition, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide 17 (MTT) powder and Roswell Park Memorial Institute (1640) Medium were obtained from the ATOCell Co. (Hungry). Fetal bovine serum (FBS) and trypsin-EDTA 10× were obtained from the Biosera Co. (England). Dimethylsulfoxide (DMSO), ethanol, dichloromethane, Tween 80 and Span 60 were also obtained from the Merck Co. MCF-7 cells were purchased from the NCBI-National Cell Bank of the Pasteur Institute (Iran).
3.2. Synthesis of SPIONs
Chemical synthesis of SPIONs was performed by co-precipitation reaction of Fe3+/Fe2+in aqueous form (14). FeCl3 (0.5 g) and FeCl2 (0.29 g) with the molar proportion of 2:1 were dissolved in deionized water (45 mL) and maintained at 80°C temperatures in an inert nitrogen atmosphere of nitrogen and alkaline medium. Then, ammonia solution (5 mL) was gradually added into the mixture to obtain the final pH of 11. The oleic acid (5 - 6 drops) was added into the suspensions and stirred for 30 min. The black precipitated particles were magnetically separated and washed two times with deionized water and one time with acetone to remove the extra ammonium. The products were then dried at 60°C in an oven for 24 h.
3.3. Preparation of the NLC-SPIOINs Loaded with Curcumin (CUR-NLC-SPIONs)
CUR-NLC-SPIONs were prepared by homogenization technique, as described by Hsu et al. with some modification (15). Briefly, the lipid phase was produced by mixing dichloromethane (12 mL) and Fe3O4 NPs (5 mg) and subsequent ultrasonication at room temperature for 10 - 15 min to completely disperse the NPs. When Fe3O4 particles were completely dispersed, the clear and transparent solution changed to a black suspension. Then, curcumin, fish oil (liquid lipid, 200 mg) and cetylpalmitate (solid lipid, 250 mg) were added to this mixture. The aqueous phase consisting of Tween 80 (150 mg) and span 60 (150 mg) surfactants in deionized water was heated to the temperature of 70°C for 5 - 10 min on a stirrer. After that, the hot oil phase was gradually added to the aqueous phase under constant stirring at 10000 rpm for 5 - 6 min followed by further homogenization for 10 min at 13000 rpm. Finally, the resultant suspension was allowed to cool down at room temperature, leading to forming the CUR-NLC-SPIONs.
3.4. Characterization of the NPs
3.4.1. Particle Size and Distribution
The particle size, distribution, and zeta potential of the CUR-NLC-SPIONs were measured by dynamic light scattering (DLS) using a Zeta sizer device coupled with a He-Ne laser at a temperature of ± 25°C. The measurements were performed at a scattering angle of 173°. Prior to measurements, samples were diluted with deionized water to eliminate viscosity and generate appropriate scattering intensity, which confirmed by the absorbance at 633 nm. Three independent measurements in 3 consequent days were recorded for NPs.
3.4.2. Transmission Electron Microscopy (TEM)
The size and morphology of SPIONs and CUR-NLC-SPIONs were further studied by transmission electron microscopy (TEM) using the negative-staining method. A diluted sample of CUR-NLC-SPIONs was stained with phosphotungstic acid and applied on carbon-coated Cu grid and allowed to dry. Then, the grid was observed by TEM.
3.5. Stability of CUR-NLC-SPIONs
To determine the stability of each prepared formulation, the samples were stored at three different temperatures of 25°C, 4°C, and 45°C for 5 months (16). During this period, samples were withdrawn from the stock solution on days 1, 3, 7, 14, 21, and 30 and then every month up to 5 months to determine zeta potential, size, and distribution. For every formulation, three samples were used.
3.6. Entrapment Efficiency and Drug Loading Capacity
The drug loading capacity and entrapment efficiency of CUR-NLC-SPIONs were determined by measuring the amount of free drug in the dispersion medium using ultrafiltration technique (17). In this regard, 3 mL of the formulation was poured in the Amicon device and centrifuged for 20 min at 4000 rpm to separate the NPs. Next, the supernatant containing free unloaded drug was analyzed by UV spectrophotometer at 426 nm. Entrapment efficiency was calculated using the Equation 1. The standard calibration curve for curcumin was obtained by measuring different concentrations of curcumin in ethanol. The percent of drug loading was calculated using the reverse method using the Equations 1 and 2. The accuracy and precision (relative standard deviation, %) and sensitivity (limit of detection) of used tests were also calculated.
3.7. Drug Release Study
The in vitro measurements of drug release from CUR-NLC-SPIONs were carried out using dialysis sack and phosphate-buffered saline (PBS) containing 30% ethanol (pH = 7.4) as receptor compartment. Then, 1 mL of each formulation was poured in dialysis membrane immersed in 19 mL of PBS and the system was maintained at 37°C under continuous stirring. At predetermined time intervals, 2 mL of the sample was withdrawn from PBS and replaced with fresh PBS/ethanol solution. The drug concentration in the receptor compartment was measured at 426 nm using UV spectrophotometer. The standard calibration curve for curcumin was obtained by measuring different concentrations of curcumin in the receptor compartment medium. To ensure that constant drug release is not due to the membrane, curcumin alone, dispersed in the same concentration, was studied under the same condition for release. The released amount was calculated again by data obtained by UV spectrophotometer. Calculation of the concentration of the released curcumin was performed using a valid calibration curve.
3.8. Cell Viability Assay
The cytotoxicity of the various formulations, including free NLC-SPIONs and CUR-NLC-SPIONs were estimated using MTT assay (18). Briefly, MCF-7 cells were grown in the RPMI-1640 medium supplemented with 10% FBS and 1% penicillin/streptomycin and incubated in a humidified cell culture incubator with 95% O2 and 5% CO2 atmosphere. The confluent monolayer cells were harvested with trypsin/EDTA from T-75 cell culture flasks and seeded into 96-well microplates at a density of 104 cells/well and were exposed to the 10, 50, and 100 mg/mL of SPOIN-NLC and SPOIN-NLC-curcumin for 24, 48, 72, and 96 h. After passing the desired exposure time, cell viabilities were estimated using MTT assay. Briefly, 5 mg/mL of the MTT solution was added to the plates and incubated for 4 h. Thereafter, purple formazan crystals were dissolved with DMSO and the absorbance was read at 570 nm and 690 nm as the reference. Cell viability was reported as the percentage of viable cells compared with the control group.
4. Results
4.1. TEM Results for SPIONs and CUR-NLC-SPIONs
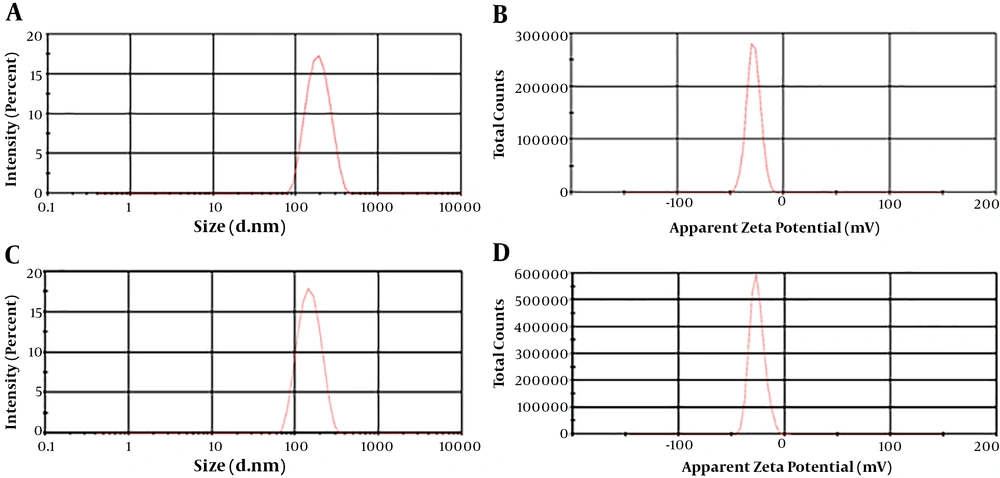
TEM was performed to evaluate the morphology and distribution of the SPIONSs, as well as CUR-NLC-SPIONs. As can be seen in Figure 1A, all the SPIONs showed a homogeneously spherical shape with a diameter of about 8 - 10 nm, which is consistent with the results of the other studies (19). Figure 2B illustrates the TEM image of CUR-NLC-SPIONs indicating the size of these particles at the range of 100 - 150 nm, which is consistent with DLS results (20, 21).
4.2. Particle Size, Distribution and Zeta Potential of SPIONs and CUR-NLC-SPIONs
DLS was carried out as a next method to determine the particle size and distribution of SPIONs and CUR-NLC-SPIONs, as shown in Figure 2. The mean size of SPIONs and CUR-NLC-SPIONs were obtained 209.6 ± 18.91 and 166.7 ± 14.20 nm, respectively. The results of CUR-NLC-SPIONs were consistent with the size range obtained by TEM with very slight differences. However, regarding SPIONs, a significant difference was found. PDI for SPIONs and CUR-NLC-SPIONs was found 0.22 ± 0.02 and 0.24 ± 0.14, respectively, which is in an acceptable range (22), showing the desired distribution for these particles (Figure 2A and C). Zeta potential is indicative of the particle surface charge and is a determinant parameter for instability of NPs. According to the results, the zeta potential was found -25.4 ± 2.77 mV and -27.6 ± 3.83 mV for CUR-NLS-SPIONs and SPIONs, respectively (Figure 2B and D). The negative charge of SPIONs is attributed to the formation of carboxylate ions of oleic acid decorated on the outer surface of MNPs (23). An increase in surface charge of SPIONs It is believed that the minimum zeta potential of ± 30 mV can provide an adequate stability for NPs (24). Therefore, considering the obtained zeta potential for both particles, a good stability was expected for them.
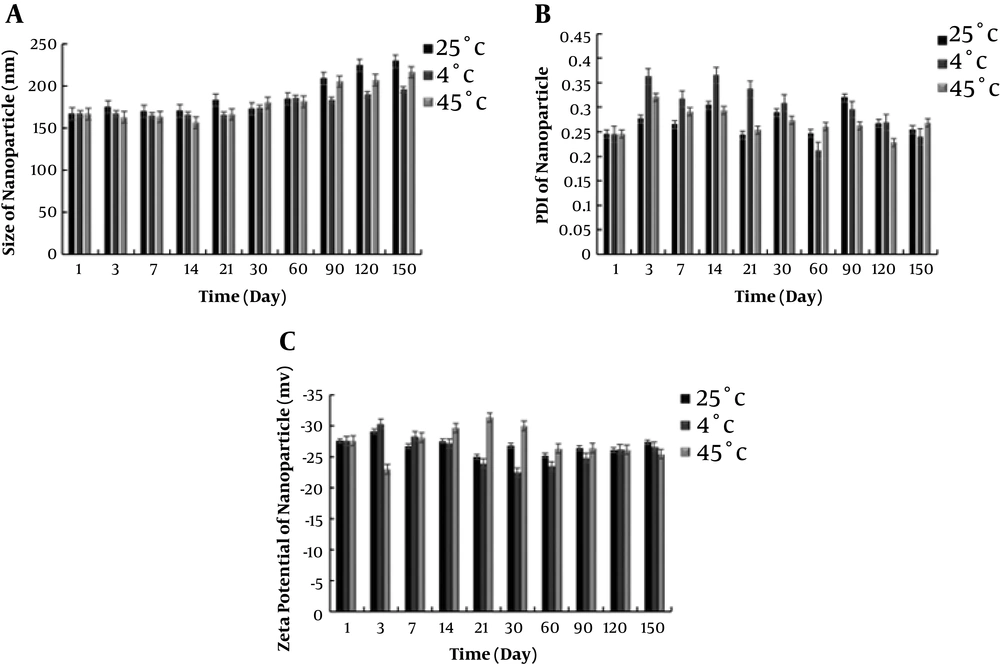
4.3. Physical Stability of CUR-NLC-SPIONs
To determine the stability of CUR-NLC-SPIONs, the samples were placed at three temperatures of 25°C, 4°C, and 45°C, and their stability, in terms of particle size, dispersion index, and zeta potential was evaluated in different time intervals up to 5 months. Following the changes in particle size is a suitable method to evaluate physical stability. As seen in Figure 3A, the average increase in particle size over a period of 5 months was less than twice the initial average particle size at each temperature. This finding indicated that the particles were not accumulated, and the changes in particle size may be due to the swelling of more surfactant absorptions on nanostructure surface. The obtained zeta potential results were in agreement with these results, confirming the stability of particles. The particle surface charge is one of the important parameters for evaluating physical stability. Higher physical repulsion between particles results in higher physical stability. To sum up, the CUR-NLC-SPIONs showed a high stability in the different conditions and according to Figure 3B, time and temperature did not exert a significant impact on NPs stability.
4.4. Curcumin Entrapment by Magnetic NLC
A standard curve was obtained by measuring the concentration of curcumin in water-tween or ethanol solution at the wavelength of 426 nm. It was used to calculate the amount of curcumin entrapped in or released by CUR-NLC-SPIONs. According to the calibration curve, the accuracy and precision (RSD%) and limit of detection for curcumin analysis were 4.54%, ± 105.1% and 0.192 µg/mL, respectively.
The amount of curcumin entrapped in CUR-NLC-SPIONs was obtained using Equations 1 and 2. The average for entrapment efficacy (EE) and DL were 99.95 ± 0.015 % and 3.76 ± 0.005, respectively. The high EE (more than 99%) for all three samples showed a suitable efficacy of prepared nanostructure for curcumin loading.
4.5. Curcumin Release Study
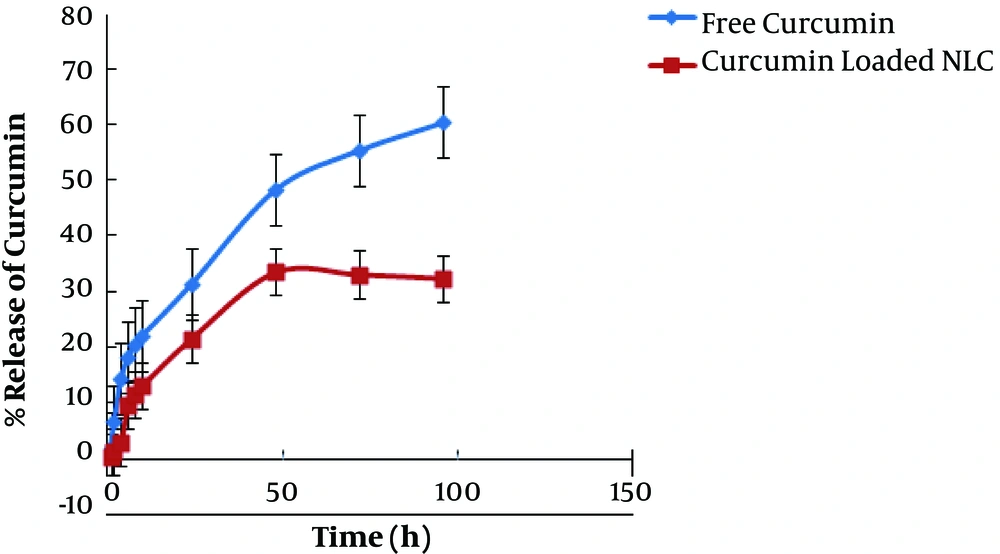
Figure 4 shows the in vitro curcumin release profile from CUR-NLC-SPIONs at a pH of 7.4. The drug release from CUR-NLC-SPIONs was compared with the release of free drug as the control. Curcumin was released from CUR-NLC-SPIONs in a more controlled release manner compared with the control, and following a controlled drug release for 48 h, the drug release reached a plateau. This finding confirmed the more effectiveness of CUR-NLC-SPIONs to provide sustained release than the control. Moreover, the maximum attainable drug release for free curcumin after 96 h was 60.40%, whereas it was 32.34% for CUR-NLC-SPIONs indicating higher solubility of the curcumin in nanoparticulate platform compared with the free drug.
4.6. MTT Assay
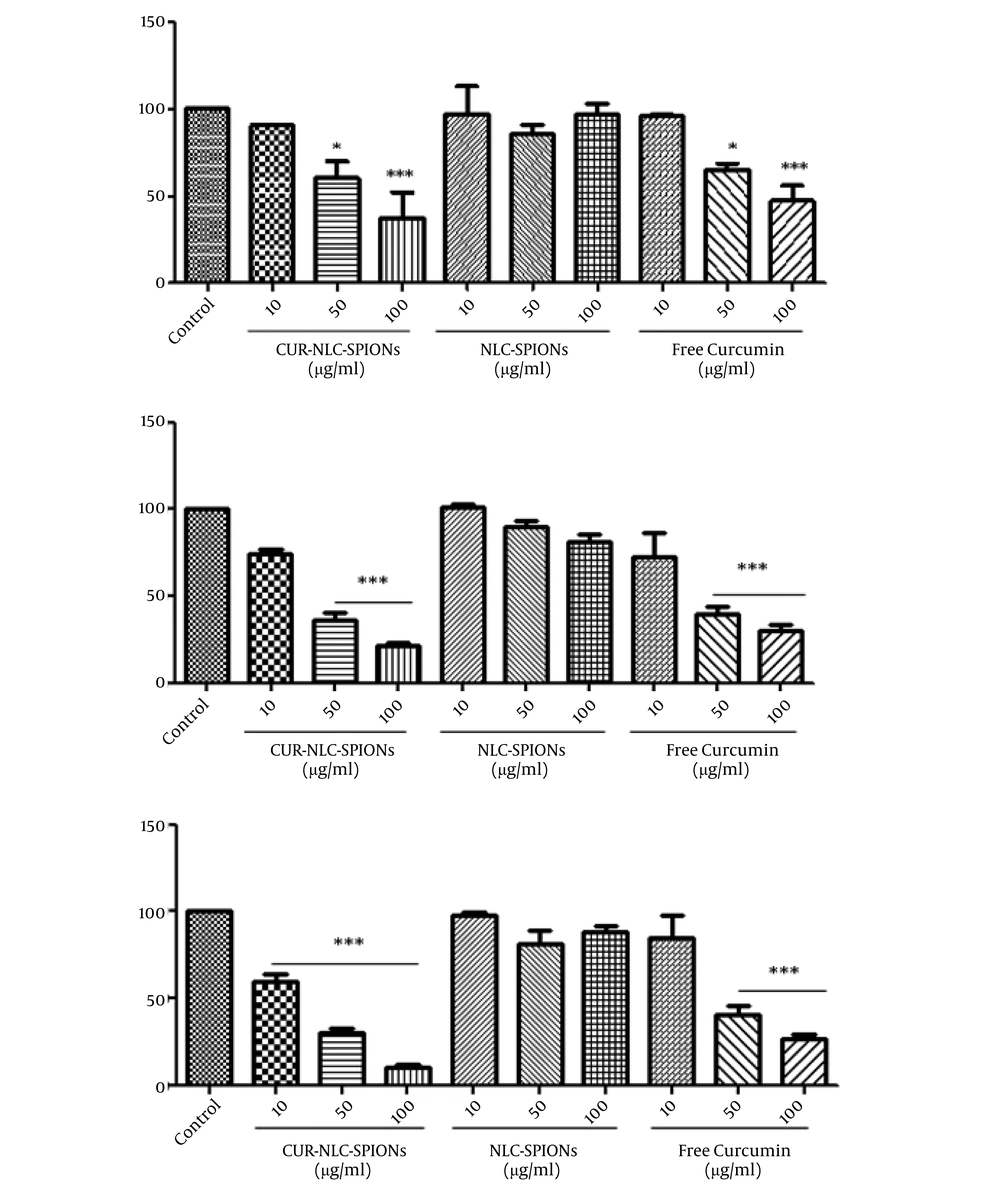
Figure 5 shows the results of MTT assay. The studied cells were treated with the increasing concentrations of CUR-NLC-SPIONs, NLC-SPIONs, and free curcumin for 48, 72, and 96 h. As shown, the cytotoxic effects of the curcumin were increased when loaded on NLC-SPIONs. The calculated IC50 of the CUR-NLC-SPIONs on MCF-7 cells after 48, 72, and 96 h exposure was approximately 42.1, 28.4, and 19.9 µg/mL, respectively. Compared with IC50 of free curcumin, CUR-NLC-SPIONs had lower IC50s on MCF-7 cells after 72 and 96 h, which can indicate a better solubility and penetration of curcumin with this newly designed nano-formulation. Our data also confirmed the safety of synthesized NLS-SPIONs, since they did not produce toxic effects at studied time periods.
The effect of CUR-NLC-SPIONs on MCF-7 cells. The cells were treated with desired concentrations of the samples and incubated for 48, 72, and 96 h at cell culture incubator. Cell viability was then assessed using MTT method AND presented as a ratio (%) to control group. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001.
5. Discussion
In this research, for the first time, we described a targeted curcumin delivery system using SPIONs with lipid nanostructured lipid carrier to improve the efficacy of breast cancer treatment. The selected anti-cancer drug in this study, curcumin (diferuloylmethane), has a potent antiproliferative effect against a variety of tumors in vitro through diverse biological properties (25-27). It also increases the antitumor effects of several chemotherapeutic drugs (28, 29) and has some advantages, including vegetable-based, very low side effects, cost-effectiveness, and accessibility (30). However, the therapeutic effects of this compound are limited due to lower aqueous solubility, chemical instability, rapid metabolism and clearance, and poor gastrointestinal absorption (31). To overcome these problems, a nanoparticulate system appears as an optimal solution. Therefore, in the present study, we used SPION incorporated with NLC for targeted delivery of curcumin to the MCF-7 cell line.
At the first, the SPIONs coated with oleic acid were synthesized with the co-precipitation method. The reaction was maintained under a nitrogen atmosphere to prevent the SPION oxidation. Oleic acid makes SPIONS to show high magnetization values resulting from diminution of surface spin disorder, and also high crystallinity of SPIONs (32). Hence, the NPs showed an appropriate stability and dispersity in organic solvents due to oleic acid. The size of these NPs was at the range of 8 - 10 nm shown by TEM. The spherical morphology of the NPs and their magnetic properties were also suitable.
In the next step, curcumin was loaded in the SPIONs incorporated with NLCs. Accordingly, solid and liquid lipids were used. Fish oil, as a liquid lipid, has some fatty acids, which their carboxylic groups are deionized after exposure to water, producing a slightly negative charge and resulting in negatively charged NPs that help the stability of them (33). Non-ionic surfactants, i.e. Tween 80 and span 60 were used, because their corresponding steric hindrance further increases the stability of the colloidal dispersion (34).
The successful synthesis of Cur-NLC-SPIONs was investigated by TEM and DLS. The size range obtained by DLS was 166.7 ± 14.20 nm. Depending on the shape, surface composition, and charge, the size range of NPs is varied (35). Very small-sized NPs (< 5 - 10 nm) are excreted renally (36), however, medium-sized NPs (30 - 150 nm) are widely distributed to the bone marrow, heart, kidney, and stomach (37). Therefore, the size of 166.7 ± 14.20 nm obtained for Cur-NLC-SPIONs in this study showed a desired potential to distribute in the body and affecting the targeted organ. The result of TEM and DLS for SPIONs was relatively different. This difference in particle size of TEM and DLS could be due to different causes. It can be explained by a high tendency of SPIONs to aggregate due to magnetic force, particularly in aqueous solution. Besides, the DLS measures the particle size in suspension form, whereas TEM measures size in dried form. Hence, DLS is a better indication of nanoparticle size, along with the PDI. Secondly, TEM provides the data for only a few hundred particles. However, in DLS technique, data is collected from many particles. And another reason can be the placement of SPIONs in the surface of NLC. The zeta potential obtained by DLS was -27.6 ± 3.83 mV. For an electrostatically stable nanostructure, a minimum zeta potential is ± 30 mV. Considering the obtained zeta potential of our designed nanostructure, it was suitable (24). PDI is a parameter to define the distribution of the size of NPs population with an acceptable range of 0 - 0.3 nm (38), which it was 0.24 ± 0.14 for the prepared NPs, indicating the suitable distribution of Cur-NLC-SPIONS.
The storage stability of nano-system was analyzed at three temperatures. The best way to determine the physical stability of NPs is measuring the changes in particle size and surface potentials (39). Surface potentials play an important role in NP stability due to electrostatic repulsion. A higher physical repulsion among particles results in higher physical stability (39). Among three temperatures of 25°C, 4°C and 45°C, the samples stored at 4°C had a greater stability in terms of size. An increase in average particle size over a period of 5 months was very low in two other temperatures. This indicates that particles have not accumulated and slight changes in particle size at 25°C and 45°C groups can be due to the swelling or absorption of the surfactant on the nanostructure surface. PDI and zeta potential of NPs were almost unchanged in three groups at different times and temperatures.
In the next step, the drug loading and in vitro drug release were evaluated. Various factors, such as the physical and chemical structure of the solid matrix, the solubility of the drug or its combination and mixing with melted lipid can affect loading capacity (40). DL% and EE% were determined 3.76% ± 0.005 and 99.95% ± 0.015, respectively, indicating an adequate curcumin loading in NPs.
The in vitro release of curcumin from NPs at PH = 7.4 was compared with free drug release and it was observed that the drug release from LNPs had two phases with a slower and gradual rate of releasing in the second phase. The more release rate of drug at the first phase is due to the drug absorbed on the surface of Cur-NLC-SPIONs. This controlled and gradual release of curcumin could result in more lasting effects and higher efficiency in targeted area in vivo. Besides, the free drug can reach toxic doses and due to poor bioavailability and solubility is removed faster from the body and naturally does not have the desired effect. In the present study, gradual curcumin release from designed formulation was obtained, which could result in nontoxic effects and delayed removal from the body. In other words, the half-life of the curcumin could be increased and the need for repeated doses would be eliminated with reduced possible side effects. Our data obtained by cell viability assay showed the stronger cytotoxic effect on MCF-7 cells in a time- and dose-dependent manner, indicating the improvement of pharmacokinetic properties of the curcumin.
5.1. Conclusions
The results of this study showed that the CUR-NLC-SPIONs are suitable carriers as a nano-formulation for targeted therapy of cancer cells by curcumin.