The
Acinetobacter genus is a major cause of nosocomial infections. In recent years, the number of
Acinetobacter isolates from blood cultures has increased (
11). In the present study, 67% of
A. baumannii strains were isolated from blood samples, which is similar to the reports of a study performed in central of Iran (
11).
Antibiotic resistance patterns among hospital pathogens may vary widely in different parts of a country or from one country to another. According to previous studies, first-line antibiotics for the treatment of
A. baumannii infection include amikacin, carbapenems (imipenem, meropenem and doripenem), ceftazidime and quinolones (
12). In addition, most pathogens have become almost completely resistant to some of the new antibiotics, such as broad-spectrum cephalosporins (cefotaxime and ceftazidime). Imipenem is recognized as the most effective drug against the infections caused by
Acinetobacter spp., but recent evidence has indicated the spread of imipenem-resistant strains. The highest rate of resistance to imipenem among
Acinetobacter spp. has been observed in
A. baumannii. The widespread emergence of resistance to imipenem is a serious threat to the future of healthcare. The extent of antimicrobial resistance varies across countries and is influenced by environmental factors and the application of different antimicrobials (
13,
14).
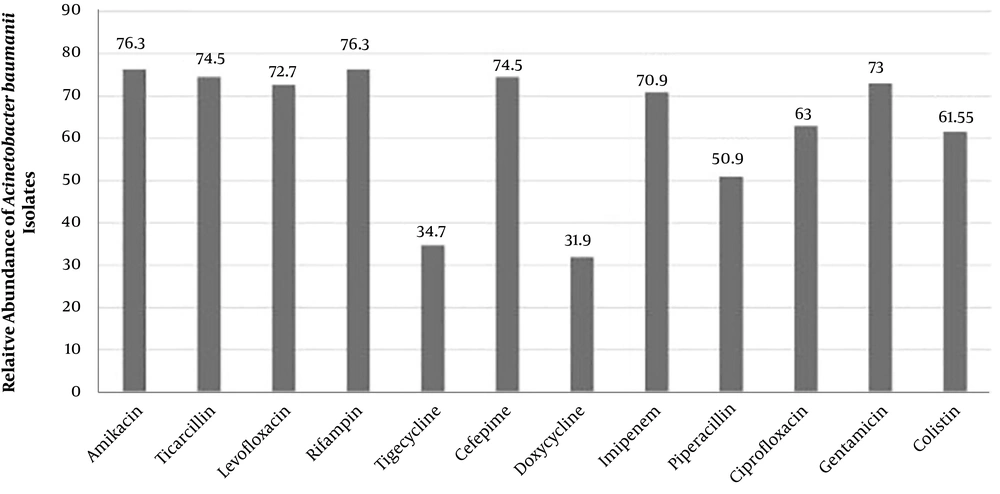
In this study, A. baumannii isolates were resistant to all the tested antibiotics, and the highest rate of resistance was observed against amikacin and rifampin (76.3%). Following these two antibiotics, the highest resistance rates were against ticarcillin and cefepime (74.5%), gentamicin (73%), levofloxacin (72.7%) and imipenem (70.9%). The lowest resistance rates were against tigecycline (34.7%) and doxycycline (31.9%).
Similar to our findings in a previous study performed in Iran, more than 60% and 78% of the MDR bacteria were resistance to amikacin (
11) and imipenem (
15), while in another study the rates of imipenem and amikacin resistance were 58% and 40% among
Acinetobacter strains, respectively (
16).
In other studies, the rates of resistance to amikacin among
Acinetobacter spp. were 47.2% (
17) and 85.2% (
12). This difference could be due to the extensive use of antibiotics in different hospital wards and the uncontrolled and arbitrary use of these drugs in our study area. Furthermore, the quality of the antibiotic disks and the techniques used for determining antibiotic susceptibility in microbiological laboratories could have affected the results. The emergence of MDR
A. baumannii strains has become a growing global health problem (
18,
19). In this regard, previous studies in Iran reported the frequency of MDR
A. baumannii strains to be approximately 80% (
11).
The high prevalence of MDR
Acinetobacter strains has been also found in other countries (
20,
21). Inconsistencies between findings of studies may be attributed to the differences in the type of studied clinical samples, time of the study and the therapeutic strategies, including the use of broad-spectrum antibiotics in initial therapy. Colistin is known as an effective antibiotic against MDR
A. baumannii strains because of its broad-spectrum antimicrobial activity against Gram-negative bacteria. However, an increase in the prevalence of colistin-resistant isolates has been noted in recent years (
18).
Given the results obtained from our study, the use of different antimicrobial compounds seems sensible for controlling such resistances. One of these compounds is the
P. harmala extract that contains various alkaloids with antimicrobial effects and no side effects (
22,
23). A previous study also demonstrated the favorable antimicrobial activity of this extract against various species of bacteria, particularly drug-resistant ones (
24).
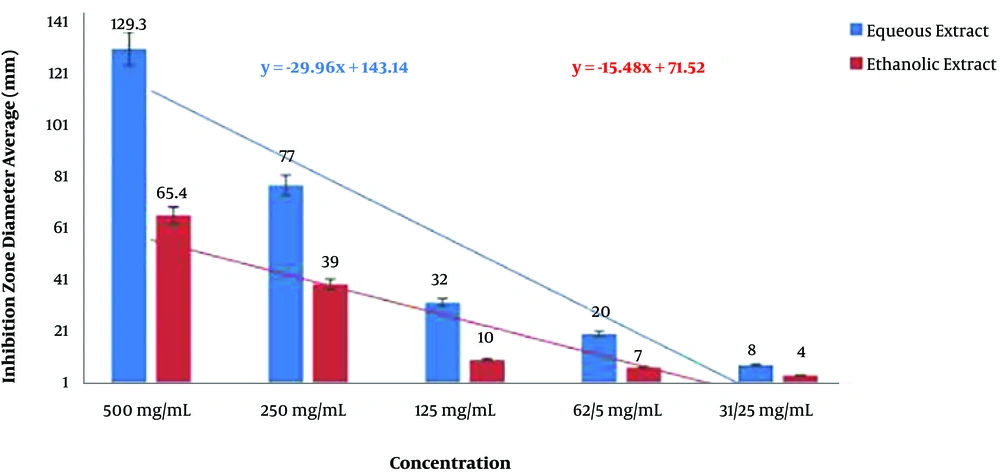
In our study, 87.3% and 50% of the
Acinetobacter isolates were susceptible to the aqueous and ethanolic extracts of
P. harmala, respectively. In other words, the aqueous extract of
P. harmala exhibited significantly higher antimicrobial activity against
Acinetobacter isolates compared to the ethanolic extract. Contrary to this finding, researchers have reported that the aqueous extract of
P. harmala is significantly more effective against
Lactobacillus and
Candida spp. compared to the aqueous extract (
25).
Our results also showed that the type of solvent had a significant impact on the extraction of active compounds from the plant. Polar solvents such as ethanol and water are thought to be more suitable for the extraction of bioactive secondary metabolites of plants. The antibacterial activity of the ethanolic extract of
P. harmala can be attributed to the presence of alkaloids such as harmine and harmaline. The abundance of these compounds in the aqueous extract may also explain the higher antimicrobial activity (
23).
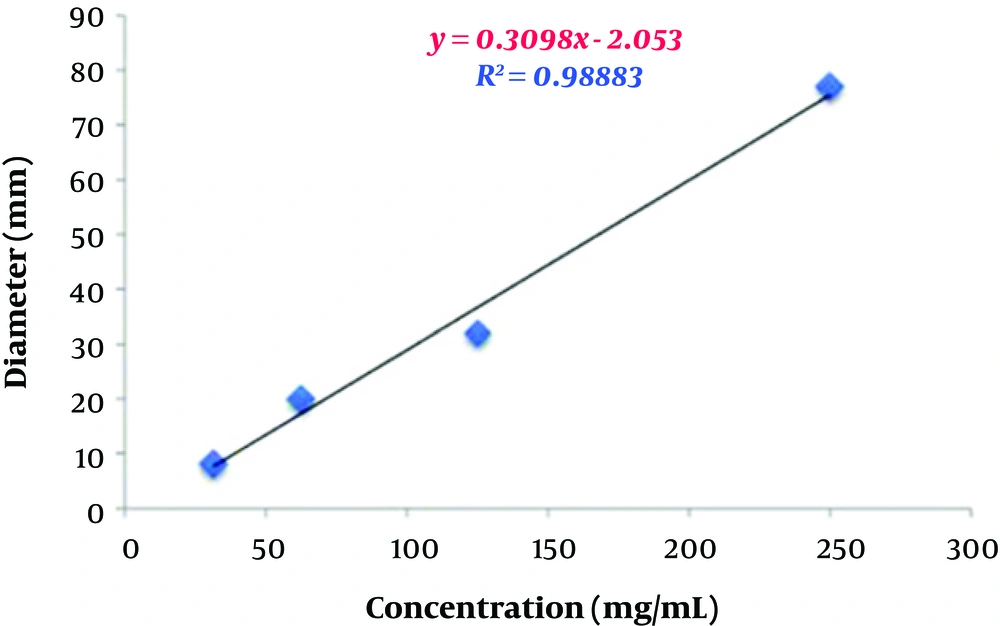
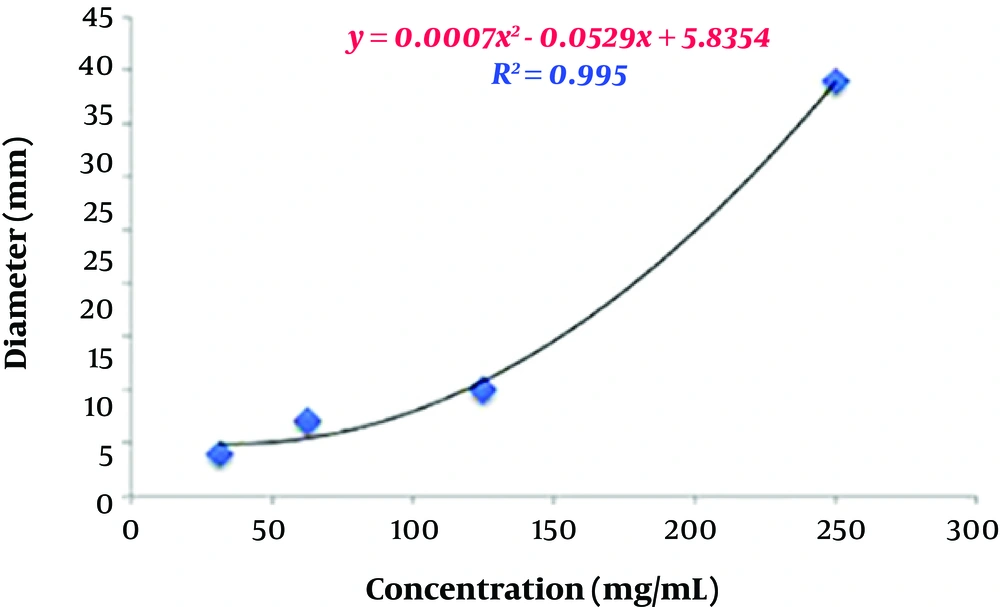
In the microdilution method, the antibacterial activity of the aqueous extract increased in a dose-dependent manner, such that 2048 μg/mL of the extract was able to inhibit the growth of almost all resistant Acinetobacter isolates.
In the present study, the number of susceptible isolates to the aqueous extract was greater than that of resistant ones, which indicates the significant inhibitory effect of the aqueous extract of P. harmala against A. baumannii isolates.
5.1. Conclusions
We observed that the prevalence of MDR A. baumannii isolates is high in blood samples of patients with sepsis, which highlights the importance of this pathogen in developing nosocomial infections and a need for programs to control the spread of this pathogen.
The aqueous extract of P. harmala has favorable antimicrobial activity against antibiotic-resistant A. baumannii isolates. This extract has low toxicity and great antimicrobial activity, making it a cost-effective alternative to chemical antibiotics for the treatment of infections caused by resistant Acinetobacter strains. Based on the results of GC-MS analysis, the higher antibacterial activity of the aqueous extract compared to the ethanolic extract could be related to the increased concentration of vasicine/peganine and 8-hydroxy deoxy peganine in the aqueous extract, which might be due to their high water solubility. Therefore, it is recommended to conduct further research on the pharmaceutical potential and antibacterial activity of these compounds.