1. Background
In recent years, natural antioxidants have been noticeable. One of the reasons for the interest in using natural antioxidants is the suspected immunity and the safety of synthetic antioxidants. These compounds exhibit a wide range of biological effects, including anti-inflammatory, anti-aging, antimicrobial, and anti-cancer. Considering their important health effects, the efficient extraction methods of natural antioxidants from their main resources are drawing great attention in food science and nutrition (1).
In the past, antioxidants were often extracted from plants, but recently some researchers have reported the antioxidant properties of lactic acid bacteria, which could promise a new source of these substances (1, 2). Probiotic bacteria, which often belong to the Lactobacillus and Bifidobacterium species, have antioxidant effects on the host (3, 4). Production of various non-enzymatic antioxidants, such as beta-carotene (5), glutathione (6), folic acid (7), peptides (3), polysaccharides (8), and enzymatic antioxidants such as superoxide dismutase and catalase (4) by probiotics, were frequently reported. In this research, the production of antioxidant substances by isolated Lactobacillus probiotic was investigated.
Wang et al. reported that antioxidant activity in bacteria started at the beginning of the exponential growth phase, and the maximum production of antioxidants is in the stationary phase (9, 10). So, Lactobacillus cultures in the stationary growth phase were harvested for studying antioxidant production. Oxidative stress and the lack of essential nutrients, which were caused by aerobic fermentation and minimal growth medium, limit the growth of bacteria; therefore, they enter the stationary growth phase (11). In this study, using the minimal medium and aeration, it was assumed that the earlier and higher production of antioxidants would be genetically induced.
The effective extraction and proper assessment of antioxidants from natural sources are crucial to explore the potential antioxidant sources and promote their applications in pharmaceuticals, functional foods, and food additives.
2. Objectives
In the current study, the methanol extraction followed by thin-layer chromatography (TLC) as a fractionation tool used to assess extracellular antioxidants derived from lactobacilli and the antibacterial properties of antioxidants fractions were also explored.
3. Methods
3.1. Bacterial Strains and Culture Media
Kefir samples were diluted in Ringer solution at 1:9 dilution and cultured in plates and tubes containing de Man, Rogosa, and Sharpe (MRS) medium. The samples were incubated under different conditions (temperatures 15, 30, and 45°C) for 48 to 72 hours under anaerobic conditions. Finally, for biochemical and identification tests, the purified colonies were frozen in an MRS broth containing 30% glycerol. In order to study the production of antioxidant and antibacterial agents, MRS broth was used, but to increase the potential production of antioxidants, plantarum minimal medium (PMM5) (12) was used.
3.2. Supernatant Preparation for Antibacterial and Antioxidant Tests
The cultured MRS broth was centrifuged at 4000 rpm for 15 minutes and passed through a sterile syringe filter with a pore size of 22 µm. After freezing at -20°C, the filtered materials were freeze-dried and extracted with methanol solvent (13). Different concentrations (10, 20, 40, 80, and 120 µg/mL) of crude extracts were prepared as dilution series of samples.
3.3. Determination of Antioxidant Activity Using the DPPH Method
One of the most widely used methods for measuring antioxidant activity is stable DPPH free radicals (14). A 40 µg/mL solution of DPPH reagent was made in methanol and stored in a dark container in a refrigerator. For the DPPH measurement, control (methanol only) and 5 dilution series of samples (250 µL) were added to the wells of a 96-well plate. DPPH solution (63 µL) was added to all of the wells. Freeze-dried MRS and PMM5 broths were used as blanks. The plate was kept in the dark place for 40 min, and an ELISA reader read the absorbance at 517 nm. The DPPH inhibition was calculated using the following formula (15). Dilution series of a synthetic antioxidant, butylated hydroxytoluene (BHT) was used as a standard in each set of the experiment for confirming the experiment conditions and DPPH function (Figure 1).
DPPH scavenging activity (%) = [(λ517 blank - λ517 sample)/λ517 blank] × 100
Schematic drawing of 96-wells plate in DPPH assay. This plate in any experiment of DPPH is as follows: Control: Methanol (187 µL) + DPPH solution (63 µL) = 250 µL. Blank: Extract (1.25, 2.5, 5, 10 or 20 µL) + x µL methanol (final volume of 250 µL). Sample: Extract (1.25, 2.5, 5, 10 or 20 µL) + DPPH solution (63 µL) + Methanol (final volume of 250 µL). BHT: BHT solution (1.25, 2.5, 5, 10 or 20 µL) + DPPH solution (63 µL) + methanol (final volume of 250 µL).
Inhibitory concentration (IC50) is used to evaluate the function of drugs and compare the efficiency of samples with each other, which is equal to the amount of drug that causes 50% inhibition of DPPH activity (15). For this purpose, the DPPH scavenging activity of the supernatant dilution series was plotted in excel software. Using each plot equation, IC50 (x in the equation) calculated while (y) equals 50. Lower IC50 values indicate stronger antioxidant activity.
3.4. Determination of Antibacterial Activity
Antibacterial activity of the extracts measured by the broth microdilution susceptibility tests. E. coli ATCC 11303 and S. aureus ATCC 6538 were used as indicator microorganisms, which were cultured in Muller Hinton broth (MHB). Serial dilutions of the freeze-dried specimens (0.5 - 256 µg/mL) in MHB with an equal volume of overnight bacterial suspension was added to each well of 96-well plate in a way that the final count of bacterial cells was about 5 × 105 CFU/mL (16). MIC was recorded 16 to 20 hours after incubation at 35 ± 2°C. The results were recorded as the lowest concentration of specimen capable of inhibiting the visible growth of microorganisms. Minimum bactericidal concentration (MBC) was determined by culturing 100 µL of each well of MIC test on an MHA plate. The results were expressed 24 hours after incubation at the appropriate temperature at the lowest concentration capable of killing 99.9% of the indicator microorganisms. All experiments were repeated three times, and chloramphenicol was used as standard. Freeze-dried MRS and PMM5 broths were used as a control to prove their inactivity in each test.
3.5. Separation of Active Fraction Containing Antioxidants by TLC and Bioautography
The developed chromatogram is sprayed or immersed in 0.2% DPPH solution in methanol, and antioxidant fraction appears as a yellow spot on TLC that its RF (maintenance coefficient) can be determined. RF is defined as the ratio of stain motion to the solvent motion. The RF value is calculated by the formula where Zi; the moving distance of the material (mm), Zf; is the distance of solvent movement. RF = Zi/Zf’ (17).
After fine adjustment of the solvent systems and determination of RF, silica gel containing antioxidant fractions were isolated from several TLC plates and placed in methanol to separate antioxidants material from silica gel and dissolve in methanol solvent. Then, silica gel was separated from the fraction by centrifugation at 12000 rpm for 15 minutes and a filtration set. The solvent containing the antioxidant fraction was placed in a glass (with a defined weight), and the solvent was evaporated using a rotary machine with a temperature of less than 40°C (17). Then, fractions with concentrations of 1000, 500, 250, 125, 60, 30, 15, 7, 3, and 1.5 were prepared, and MIC of each fraction was determined using the microdilution susceptibility tests that were described previously (16).
3.6. Study of Isolated Lactobacillus Probiotic Potential
Probiotic potential of isolated Lactobacillus evaluated by resistance to pH and bile acid. The ability of the isolates to survive and grow at different pHs (3 and 5.5) was determined by bacterial growth (A600 initial = 0.2) in MRS broth, which its pH adjusted using hydrochloric acid at 37°C for 24 h followed by lawn culture method for the next 24 h (18). Resistance to bile acids was determined by inoculating 5 µL of one overnight culture of each isolate (A600 ~ 0.1) on an MRS agar plate containing a final concentration of 0.15, 0.3, 0.5 bile acid. These plates were incubated at 37°C under anaerobic conditions, and the growth and non-growth of the isolates were evaluated after 72 hours (19).
3.7. Screening Antioxidant-Producing Isolates Based on Resistance to Active Oxygen Species (ROS)
Resistance of isolates to active oxygen species was performed by three resistance tests against hydrogen peroxide (20), hydroxyl radical (21), and superoxide anions (20).
Biochemical identification of antibacterial-producing strain: growth ability of strain in MRS broth at 10°C for 7 days at 42°C for 24 - 48 h, growth in 2, 4 and 8 % NaCl, oxidase, mobility test, production of H2S, production of gas from glucose, arginine hydrolysis, esculin and gelatin, nitrate recovery test, citrate, citrus growth, and fermentation of various sugars were investigated (22).
Genetic identification of the antibacterial-producing strain: Lactobacillus strain was cultured in 5 ml of MRS broth at night, and DNA was extracted using a DNA extraction kit of gram-positive bacteria (pioneer gene transfer, Iran) according to the manufacturer’s instructions. For proliferation of V2-V3 regions of 16S-23S intergenic region (ITS), the following primers that were used in several studies in different years were selected (23, 24): 16-1A (5’-GAATCGCTAGTAATCG-3’) and 23-1B (5’-GGGTTCCCCCATTCGGA-3’) (Pioneer, Korea). A PCR reaction solution of 5 µL PCR buffer, 0.2 mM primer, 0.2 mM dNTPs, magnesium chloride 0.5 mM, U5 Taq-polymerase enzyme (CinnaGen, Iran), and 200 ng of extracted DNA were used, and distilled sterilized water was added to a final solution volume of 50 µL. The PCR program used for initial denaturation for 2 minutes at 94°C and 30 cycles, including 1 minute at 95°C, 30 seconds at 55°C, and 1 min at 72°C, and final proliferation at 72°C for 5 min. The PCR products were purified using a GeneJET kit (Thermo Fisher Scientific, US) and were sent to Pioneer, Korea, to determine sequence from two directions. The bacterial sequence was investigated using the BLAST program on the NCBI website. The phylogenetic tree of the Lactobacillus strain was then drawn using the MEGA 7.0.14 software program by Neighbor-joining (NJ) (25).
3.8. Relative Identification of Bioactive Fractions with Some of the Reagents
Some reagents that were used for identification of the nature of antioxidant-antibacterial fraction were ninhydrin, alkaline, iron chloride, Benedict, Folin, chloroform, and concentrated acid (26). All materials were purchased from Merck, Germany.
3.9. Statistical Analysis
Data were presented as the mean ± SD of three measurements. Differences in mean values between the IC50 values of groups were analyzed by one-way analysis of variance (ANOVA). All tests were considered statistically significant at P ⟨ 0.05.
4. Results and Discussion
In the period from 2006 to 2011, 2250 outbreaks of food-borne diseases were reported in Iran. Statistical analysis showed that the number of these individuals increased from 0.07 (2006) to 1.38 (2011) in 100,000 people (27). The use of antibiotics led to an excessive increase in some pathogenic bacteria that are not only resistant to antibiotics but also are resistant to certain food preservation and processing methods. In addition, due to increased consumer awareness about the negative effects of artificial preservatives on human health and the benefits of natural additives, researchers have been more inclined to produce and apply natural products in foods. These factors led the food industry to search for natural preservatives that can increase the health and quality of food (28). One of the best methods for preventing and controlling pathogenic bacteria is the use of lactic acid bacteria (29).
Some studies on the health benefits of probiotics were published, and some researchers reported antibacterial activity of probiotics against pathogens (30, 31). Turgay and Erbilir (31) showed that isolates of L. casei and L. bulgaricus exhibited weak antibacterial activity against E. coli and S. aureus, which was in contrast to our results.
Among lactobacilli that were isolated from kefir, Lactobacillus K1C strain showed a wide antibacterial activity against E. coli ATCC 11303 and S. aureus ATCC 6538. This strain has a white colony of 2 to 3 mm in size. Under the microscope, long bacilli were seen with the pair and the chain arrangements. Growth inhibition due to superoxide anions produced by Paraquat was not observed in the case of the K1C strain, and it showed a weak growth at 42°C. It grows at different sodium chloride concentrations and 1% of some tasted sugars (Mannitol, sorbitol, sorbose, sucrose, glucose, galactose, melesitose, trehalose). The probiotic properties of this strain were shown in Figures 2 and 3.
A, Survival of Lactobacillus strain K1C in the presence of hydrogen peroxide. The cultures of the isolates were suspended at 107 CFU/mL in saline phosphate buffer and heated with 0.4 Mm hydrogen peroxide; B, survival of Lactobacillus strain K1C in the presence of radical hydroxyl. The strains were suspended at 107 CFU/mL in a salt buffer and heated by a solution containing THA of 10 mM in saline phosphate buffer and copper sulfate 0.01 mM. Hydroxyl radicals are generated by the reaction of Fenton and by adding hydrogen peroxide 1 mM in solution. These experiments were repeated for three times.
The antioxidant traits increased significantly in minimum culture media and anaerobic condition (492.1 ± 0.25 µg/mL) compared to the similar condition in MRS broth (880.96 ± 0.05 µg/mL). The highest antioxidant production was observed in the stationary growth phase of the aerobically fermented minimum medium (266.82 ± 0.17 µg/mL).
The growth phase of lactobacilli during the fermentation process could have effects on antibacterial and antioxidant production. Based on the results (Figure 4), the maximum antibacterial activity was observed in the middle of the logarithmic stage until the beginning of the stationary growth phase. The maximum antioxidant activity in the stationary phase is in agreement with the previous reports by Wang et al. (10). Kuliisar et al. (20) stated that more aeration gave more free radicals, which would increase the antioxidant properties of lactobacilli. According to the results, antioxidant activity was elevated at aerobic conditions, but antibacterial activity was decreased by aeration that was approximately in agreement with the results of Abbasiliasi et al. (32).
The results showed that the antibacterial activity of Lactobacillus strain K1C increased in a minimal medium, but aerobic conditions have no effect on the production of antibacterial agents (Figure 5). Antibacterial activity (MIC) of cellular free methanol extract of MRS medium fermented with Lactobacillus casei strain K1C against E. coli ATCC 11303 and S. aureus ATCC 6538 were 32 and 128 mg/µL, respectively. The antibacterial activity of cellular free methanol extract of PMM5 medium fermented with Lactobacillus casei strain K1C towards E. coli ATCC 11303 and S. aureus ATCC 6538 were 25.32 and 50.64 mg/µL.
A, MIC of the methanol extract of MMP5 supernatant cultured by L. casei strain K1C under anaerobic conditions for 48 hours against S. aureus ATCC 6538 (MIC 25.33 mg/mL) and E. coli ATCC 11303 (MIC 50.64 mg/mL); B, MIC of the methanol extract of MMP5 supernatant cultured by L. casei strain K1C under aerobic conditions for 48 hours towards S. aureus ATCC 6538 (MIC 261.76 mg/mL) and E. coli ATCC 11303 (MIC 130.87 mg/mL).
After fractionation of extracellular methanol extract of a minimal medium by TLC and using of TLC-DPPH bioautography method, only 3 antioxidant fractions were isolated that their Rf were 0.03, 0.20, and 0.34 by a solvent system of methanol: chloroform (2:3). Fraction number 1 with an Rf value of 0.03 showed antibacterial activity. The MIC of this fraction was evaluated against some pathogenic bacteria such as S. aureus (1,000 µg/mL), E. coli (500 µg/mL), Sal. Typhi (31.25 µg/mL), S. pyogenes (250 µg/mL), Y. entrolitica (1,000 µg/mL), S. epidermidis (500 µg/mL), B. licheniformis (250 µg/mL), and K. pneumonia (250 µg/mL).
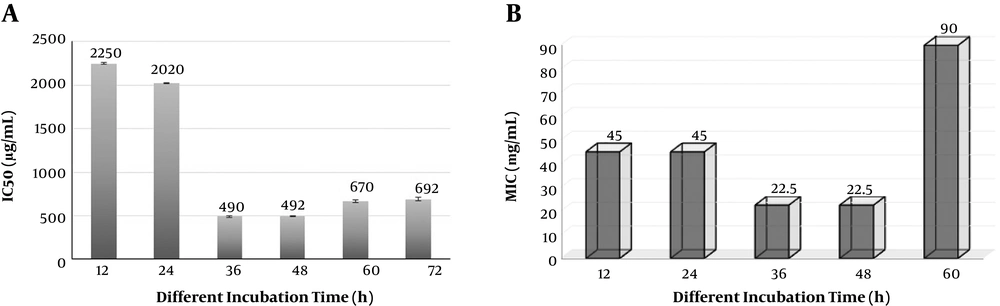
For determination of antioxidant and antibacterial production at different times of growth phase, MIC and DPPH assay of cell-free supernatants of L. casei strain K1C were monitored at different hours that its results presented in Figure 6.
Determination of antioxidant and antibacterial production in PMM5 medium at different times of growth phase: A, IC50 of L. casei strain K1C supernatants harvested at different hours and determined by DPPH assay; B, MIC of L. casei strain K1C supernatants harvested at different hours and monitored by broth microdilution susceptibility method against E. coli ATCC 11303. Note: Lower IC50 and MIC values indicate stronger antioxidant and antibacterial activities.
As expected, with the relative purification of fractions by TLC, the antioxidant and antibacterial activity of the fractions increased compared to the crude methanol extract of the same supernatant, which indicates that the purification and separation steps are carried out carefully, and there is an effective ingredient within the separated fraction. MIC of fraction number 1 derived from aerobically fermented PMM5 by L. casei strain K1C is 1,000 µg/mL against S. aureus and 500 µg/mL towards E. coli, whereas the crude methanol extract of the same supernatant has a MIC of 25.32 mg/mL against both indicator bacteria.
According to the biochemical tests for identification of fraction number 1 with Rf value of 0.03, the fraction has amine bonds and includes phenolic compounds, but no sugar and flavonoids compounds were detected.
According to the phylogenetic tree of L. casei K1C strain (Figure 7), K1C isolate showed the most similarity (92%) to the probiotic bacteria known as L. casei strain Shirota, which were recorded under the access number KU954559 in the GenBank database.
Phylogenetic tree based on nucleotide sequence of variable regions V2-V3 of 16S rRNA gene and 16S-23S ITS of Lactobacillus strain K1C, which is drawn using the neighbor-joining (NJ) method of MEGA software version 7.0.14. The scale line represents an alternative 0.01 for each nucleotide position. Bootstrap has been shown in 1000 splashes per cent in all splits.
4.1. Conclusions
There is no fractionation report on Lactobacillus supernatant by TLC, and this is the first published data about this procedure and the proper solvent system that was methanol: chloroform (2:3). Based on the results, the supernatant extract of Lactobacillus strain K1C exhibits strong antibacterial activity against pathogenic bacteria; therefore, it could be useful as a starter culture to equip antioxidants in food or as a natural preservative to the packaging food. This strain has the highest genetic similarity with L. casei strain Shirota, which is considered to be food additives authorized by the food industries (33). However, its impact on the microbiota and sensory characteristics of the food product should be investigated.