Dear Editor,
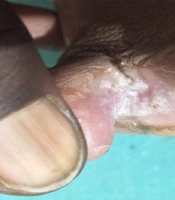
Chronic intertrigo is a common and recurring interdigital dermatosis of the toe and finger web space (1). Several topical medications have been prescribed for this problem. However, these topical medications and physical barriers are ineffective in some patients due to the hydration and maceration of the space. To keep the space dry, some techniques have been used to separate the toe web space. Moreover, the routine separation of the toes for 6 weeks or more is a little cumbersome (2-4). Cryosurgery and chemical cautery (as personal experience) are effective treatment modalities for interdigital dermatoses, but the availability of cryogen is a major concern (5). As a result, chronic intertrigo of toe web space represents a therapeutic challenge for dermatologists. To solve these problems, we used cyanoacrylate glue which is US FDA-approved cyanoacrylate glue with suitable washing-off resistance and control over transepidermal water loss from a macerated or abraded area (6).
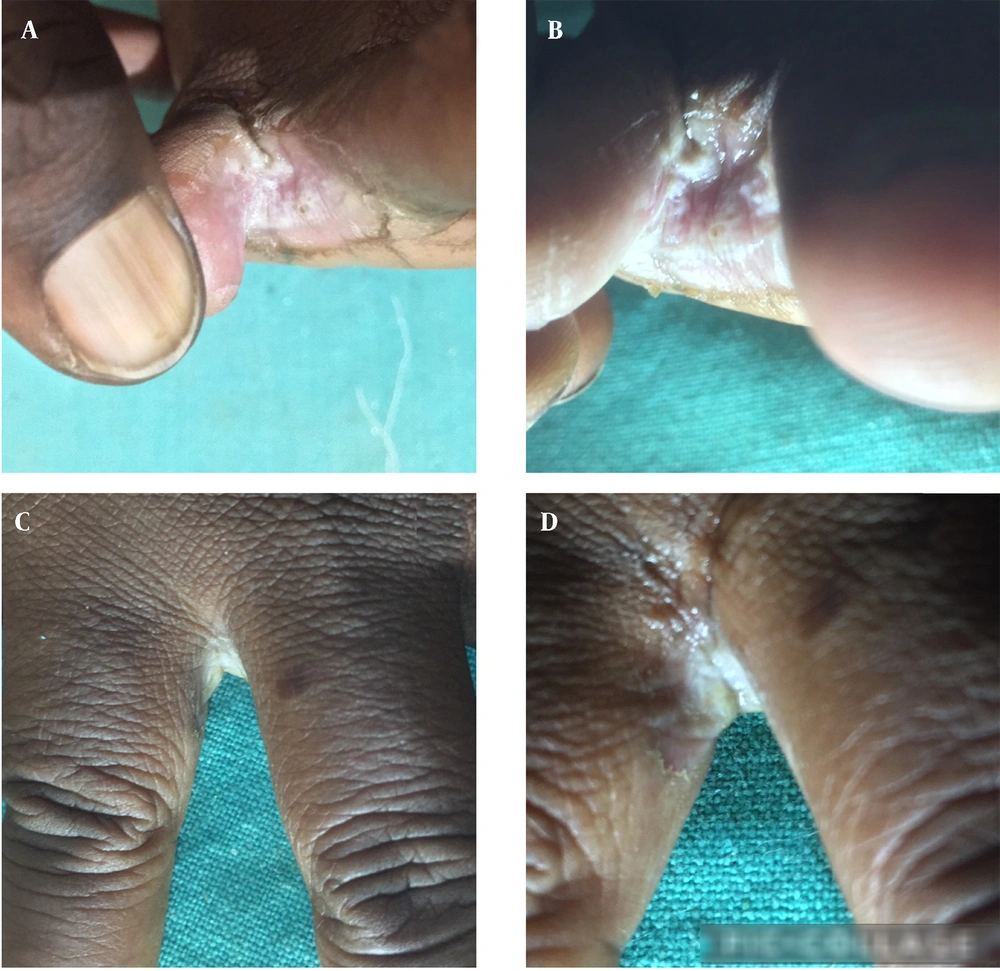
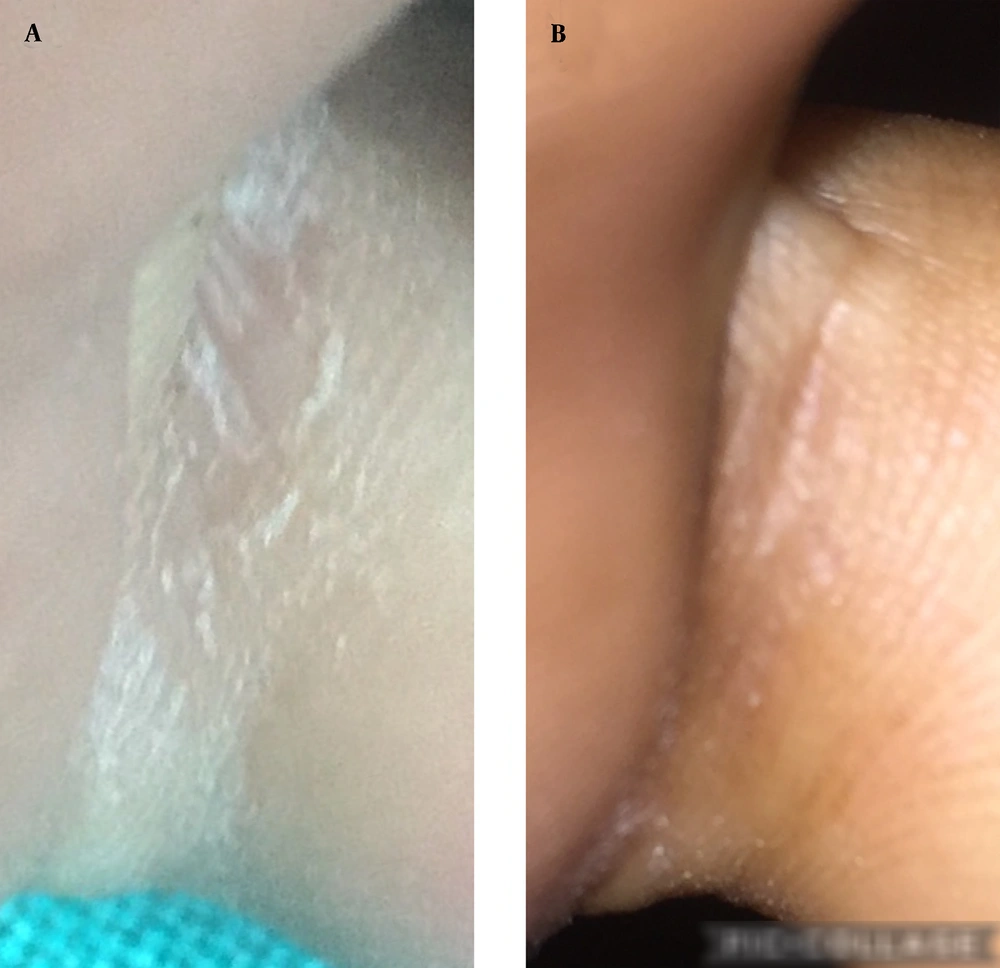
Keeping the toe web space dry and sealing the space from external causative factors are two major factors in treating toe intertrigo. Keeping this in mind, we have used cyanoacrylate glue (thermal, dehydrating, anti-infective, sealing properties with better washing-off resistance and trans-epidermal water loss (TEWL) than physical barrier creams) for treating interdigital dermatoses as a simple and cost-effective method of treatment. The cost of glue is 0.5 US dollars for 20 mL. Under the aseptic condition, the glue is applied correctly into macerated interdigital space (to seal the space) using a cotton bud (Figure 1A-D). The procedure was repeated at weekly intervals for 4 weeks in most cases. The glue must be applied twice or thrice a week in patients with water work. For patients with severe intertrigo, in the first visit, glue can be applied in the intertriginous toe space after putting twigs of cotton. This may cause a burning sensation that can be controlled by spraying water to avoid chemical burn injury. No topical medications were given except liquid paraffin and general precautions to keep the space dry. Patients were followed up weekly for 6 weeks. The toe web space became dry and free from maceration and interdigital space healed in 4 - 6 weeks without significant complications (Figure 2A and B). Three patients with chronic toe intertrigo were treated successfully by this method. Therefore, cyanoacrylate glue could be a good, cost-effective agent for keeping the toe space dry, clean, macerated, and infection-free. It can be an excellent topical option for treating the chronic intertrigo of toe web space. However, the role of chemical sealing with cyanoacrylate glue should be evaluated by a case-control study on a large number of patients.
Keypoints of message: (1) cyanoacrylate is a monomer that, when combined with moisture, generates a polymer coating that protects the skin against wetness, maceration, infection, and increased trans-epidermal water loss; (2) this adhesive material has been found statistically to perform better in wash-off resistance and TEWL from abrasion than physical barriers, such as petrolatum, silicone, and zinc; (3) glue can be administered in the intertriginous region after inserting a cotton twig in patients with severe intertrigo. It causes a burning feeling, which may be managed by spraying water; (4) typically, one application per week is adequate to treat the lesions. Water workers might be applied every two days or every day in some circumstances; (5) it is less allergic and safe to use on the skin because it is FDA authorized in the United States.