1. Background
The current epidemic of the COVID-19 presents an unprecedented and severe global health problem with high hospitalization and mortality rates. In more than 15% of the patients, the disease may progress to acute respiratory distress syndrome (1). The global prevalence of this disease undoubtedly has a tremendous impact on various medical and socio-economic issues. The care of health care systems, the overburdening of hospital capacity, the inability of all patients to access respiratory support, and the lack of personal protective equipment, even in developed countries with high-level medical care, have become a threat. Considering the critical care aspects of the pandemic, the emphasis is primarily on respiratory support, ARDS treatment, and the prevention and treatment of multiple organ failure (2). Although the main target of the virus is the respiratory system, but also, other systems of the body may be involved. Therefore, different and varied symptoms may occur. Coagulation and thromboembolic events are the complications of this disease and are considered poor prognostic factors. Coagulopathy can be one of the leading causes of death in COVID-19 patients. Some studies have also suggested that coagulation is associated with the severity of the COVID-19 symptoms (3). The results of published studies show that COVID-19 can also affect the cardiovascular system. Population analysis in China, Italy, and the United States shows that among the diseases associated with the severe and fatal course of the COVID-19, cardiovascular disease is one of the most common so that COVID-19 is associated with higher mortality in the presence of cardiovascular disease (2, 4). Given a large number of articles, preprints, and data recently published from unofficial sources, it is very important to be based on known knowledge. Although most of the recommendations are weak and based on mediocre evidence, they do offer some basic knowledge that can enhance our insight into this new disease (2, 5).
Little is known about the hemodynamic consequences of the COVID-19 disease and the hemodynamic management of patients in need of admission to the intensive care unit (6). These patients have several reasons for hemodynamic instability. First, they may have hypovolemia due to fever and fluid restriction, which is recommended from hospitalization to limit the development of pulmonary edema. Like any patient with systemic inflammation, they may have some degree of vasodilation. Circulating cytokines may cause ventricular dysfunction (6-8). Some studies have suggested that myocarditis may be the cause of systolic and diastolic dysfunction (6-11).
On the other hand, in the current situation, the necessity of some surgeries is undeniable. Inevitably, unavoidable changes occur during surgery and anesthesia in the COVID-19 patients. Hemodynamic instability during anesthesia has adverse effects on blood circulation and the process of oxygenation to body tissues, especially vital organs such as the brain, kidneys, and liver. Each anesthetic causes specific hemodynamic changes (12). Also, pressure stimulations following laryngoscopy and endotracheal intubation, intraoperative bleeding, and loss of circulating fluids can cause hemodynamic changes (13).
Given that the COVID-19 can cause hemodynamic changes, anesthesia also affects hemodynamic factors; therefore, the concurrence of the COVID-19 and its pathogenic changes with general anesthesia may increase the risk of anesthesia. Therefore, the question arose in the minds of researchers whether the simultaneity of these two events can cause significant hemodynamic changes in the patient and increase the risk of anesthesia.
2. Objectives
According to our best searches in English and Persian sources, no study has been found on hemodynamic changes in patients with the COVID-19 under general anesthesia; therefore, the aim of this study was to evaluate this issue.
3. Method
The aim of this cross-sectional analytical study of two groups was to compare the hemodynamic status of non-COVID-19 and the COVID-19 patients during anesthesia in Allameh Bohlool Gonabadi hospital in 2020. The study population comprised of non-COVID-19 patients and those with the COVID-19 who were candidates for emergency and urgency surgery under general anesthesia in hospital. Non-COVID-19 patients included those who were candidates for surgery and did not have symptoms of the disease and on whom PCR was negative. Definitive patients included those candidates for surgery whose PCR test was positive and had mild to moderate disease according to the physician diagnosis and based on guidelines for diagnosis and treatment of the COVID-19 of the Ministry of Health. To determine the sample size, first, a pilot study was performed on 20 people (10 people in each group) and then the sample size will be determined using G POWER software and a 95% confidence and error rate of 0.05. Finally, 20 patients (40 in total) were included in the study in each group. After obtaining permission from the University Student Research Committee and the permission of the Ethics Committee (ethics code: IR.GMU.REC.1399.112), the researcher referred to the operating room of Allameh Bohlool Hospital, and then available manner selected patients. Inclusion criteria included no history of chronic diseases such as heart disease, hypertension and chronic respiratory disease. Other chronic diseases were monitored which were not statistically significant in the two groups.
Exclusion criteria included the presence of any unexpected event that affected the hemodynamic status of the patients, such as severe bleeding during surgery. The variables of anesthesia duration, kind of surgery, blood group, residential status, sex, age, weight and smoking were compared in the two groups, which were not statistically significant in the two groups.
Demographic information was obtained from the patient’s file and during the preoperative oral interview. Patients in both groups were monitored during the operation and were excluded from the study if they developed an acute condition and their hemodynamic status was disturbed. All patients underwent general anesthesia in this way: After connecting standard monitoring devices, midazolam 0.03 mg/kg and fentanyl 2 μg/kg were injected as a premedication. Induction of anesthesia was administered using intravenous Thiopental at a dose of 3 - 5 mg/kg and then Atracurium 0.5 mg/kg to induce muscle relaxation for intubation. After about 3 minutes of ventilation with 100% oxygen, endotracheal intubation was performed for male patients with 8 - 8.5 tubes and female patients with 7 - 7.5 tubes. At the end of the operation, the patients received 100% oxygen and the rest of the block was reversed using Atropine and Neostigmine. After suctioning the discharge, which was performed gently and uniformly in all patients, the endotracheal tube was removed. Patients’ hemodynamic variables were recorded minute by minute in a vital signs monitor including systolic blood pressure (SBP), diastolic blood pressure (DBP), pulse, and arterial oxygen saturation. Respiration was recorded in 5 minutes before the start of anesthesia and 5 minutes after the withdrawal from anesthesia. To analyze the information, data were first entered into SPSS software version 19. Descriptive statistics including frequency distribution tables, central indices, and dispersion were used to describe the research units. Shapiro-Wilk test was used to evaluate the normality of the distribution of quantitative variables. One-way analysis of variance was run to compare the mean of the quantitative variables with normal distribution such as blood pressure, pulse and SpO2 and Kruskal-Wallis test was used for quantitative variables with the abnormal distribution.
4. Results
Forty patients were included in the study. Two patients were excluded from the study due to femoral surgery and heavy bleeding. These two had a negative PCR test. The mean and standard deviation of the age of the research units was 47.55 ± 21.37 and their mean weight was 60.35 ± 13.49. The majority of research units (65%) were female, 80% were married, 75% were city dwellers and 55% were unemployed. 65% of patients did not report a history of chronic disease. 35% of the research units were positive for blood group A. The mean duration of anesthesia was 89.25 ± 46.85 minutes. There was no statistically significant difference between the two groups in terms of duration of anesthesia, kind of surgery, type of chronic disease, type of drug used, blood group, residential status, sex, age, weight and smoking (P > 0.05).
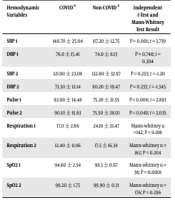
Shapiro-Wilk test was used to determine the normality of quantitative variables. The results showed that blood pressure and pulse had a normal distribution (P > 0.05) and SpO2 and respiration had an abnormal distribution (P < 0.05). Paired t-test showed that there was a significant difference between SBP before and during anesthesia in the COVID group (P = 0.0001); so, during anesthesia SBP was controlled by Propofol 100 - 200 μg/kg and remifentanil 0.5 - 0.5 μg/kg/min. There was also a significant difference between pulse before and during anesthesia (P = 0.001); so, during anesthesia the pulse was lower. DBP also decreased during anesthesia compared to before anesthesia; but was not significant (P = 0.715) (Table 1). Also, in the non-COVID group, there was no difference between SBP and DBP and pulse before and during anesthesia (P = 0.085, P = 0.683, P = 0.051, respectively). The results of the Wilcoxon test showed that there was a statistically significant difference in respiration and SpO2 in the COVID group before and during anesthesia (P = 0.0001; for each); so that during anesthesia SpO2 was higher and normalized and respiration was completely improved. Also, in the non-COVID group, there was a statistically significant difference in terms of respiration and SpO2 during anesthesia with pre-anesthesia (P = 0.0001; for each) (Table 1).
| Quantitative Variables | Mean ± SD | Test Results | Type of Test |
|---|---|---|---|
| COVID | |||
| SBP | t = 0.822; P-value = 0.0001 | T-paired test | |
| SBP 1 | 140.70 ± 25.04 | ||
| SBP 2 | 121.09 ± 23.08 | ||
| DBP | t = 0.087; P-value = 0.715 | T-paired test | |
| DBP 1 | 76.0 ± 15.41 | ||
| DBP 2 | 73.12 ± 0.14 | ||
| Pulse | t = 0.676; P-value = 0.001 | T-paired test | |
| Pulse 1 | 92.14 ± 0.48 | ||
| Pulse 2 | 90.15 ± 1.63 | ||
| Respiration | z = 2.929; P-value = 0.0001 | Wilcoxon test | |
| Respiration 1 | 17.0 ± 2.66 | ||
| Respiration 2 | 12.40 ± 0.96 | ||
| SpO2 | z = 3.745; P-value = 0.0001 | Wilcoxon test | |
| SpO2 1 | 94.60 ± 2.54 | ||
| SpO2 2 | 99.20 ± 1.75 | ||
| Non-COVID | |||
| SBP | t = 0.395; P-value = 0.085 | T-paired test | |
| SBP 1 | 117.20 ± 12.75 | ||
| SBP 2 | 121.90 ± 23.08 | ||
| DBP | t = 0.097; P-value = 0.683 | T-paired test | |
| DBP 1 | 74.70 ± 8.13 | ||
| DBP 2 | 80.20 ± 19.47 | ||
| Pulse | t = 0.845; P-value = 0.051 | T-paired test | |
| Pulse 1 | 75.20 ± 21.55 | ||
| Pulse 2 | 70.50 ± 28.01 | ||
| Respiration | z = 3.945; P-value = 0.0001 | Wilcoxon test | |
| Respiration 1 | 24.10 ± 21.47 | ||
| Respiration 2 | 17.50 ± 16.34 | ||
| SpO2 | z = 3.947; P-value = 0.0001 | Wilcoxon test | |
| SpO2 1 | 98.30 ± 0.67 | ||
| SpO2 2 | 99.90 ± 0.31 | ||
Abbreviations: SBP 1, systolic blood pressure before anesthesia; SBP 2, systolic blood pressure during anesthesia; DBP 1, diastolic blood pressure before anesthesia; DBP 2, diastolic blood pressure during anesthesia; Pulse 1, pulse before anesthesia; Pulse 2, pulse during anesthesia; Respiration 1, respiration before anesthesia; respiration 2, respiration during anesthesia; SpO2 1, SpO2 before anesthesia; SpO2 2, SpO2 during anesthesia.
An intergroup assay by independent t-test showed that before anesthesia, SBP was significantly different between the two groups (P = 0.0001); it was higher in the COVID-19 group than in the non-COVID-19 group. Before and during anesthesia, pulse was a statistically significant difference in the two groups (P = 0.006, P = 0.049; respectively); it was higher in the COVID-19 group than in the non-COVID-19 group. Also, SBP during anesthesia and DBP before and during anesthesia did not significantly change between the two groups (P = 0.233, P = 0.740, P = 0.233; respectively). In addition, the Mann-Whitney test showed no statistically significant difference between the two groups in terms of respiration and SpO2 during anesthesia (P = 0.204, P = 0.296; respectively), while respiration and SpO2 before anesthesia showed a significant difference between the two groups (P = 0.018, P = 0.0001; respectively); two groups: it was smaller in the COVID-19 group than in the non-COVID-19 group (Table 2).
| Hemodynamic Variables | COVID a | Non-COVID a | Independent t-Test and Mann-Whitney Test Result |
|---|---|---|---|
| SBP 1 | 140.70 ± 25.04 | 117.20 ± 12.75 | P = 0.001; t = 3.739 |
| DBP 1 | 76.0 ± 15.41 | 74.0 ± 8.13 | P = 0.740; t = 0.304 |
| SBP 2 | 121.90 ± 23.08 | 132.80 ± 32.97 | P = 0.233; t = -1.211 |
| DBP 2 | 73.30 ± 12.14 | 80.20 ± 19.47 | P = 0.233; t = -1.345 |
| Pulse 1 | 92.00 ± 14.48 | 75.20 ± 21.55 | P = 0.006; t = 2.893 |
| Pulse 2 | 90.10 ± 15.63 | 75.50 ± 28.01 | P = 0.049; t = 2.035 |
| Respiration 1 | 17.0 ± 2.66 | 24.10 ± 21.47 | Mann-whitney u =142; P = 0.018 |
| Respiration 2 | 12.40 ± 0.96 | 17.5 ± 16.34 | Mann-whitney u = 162; P = 0.204 |
| SpO2 1 | 94.60 ± 2.54 | 98.3 ± 0.67 | Mann-whitney u = 38; P = 0.0001 |
| SpO2 2 | 99.20 ± 1.75 | 99.90 ± 0.31 | Mann-whitney u = 176; P = 0.296 |
Abbreviations: SBP 1, systolic blood pressure before anesthesia; SBP 2, systolic blood pressure during anesthesia; DBP 1, diastolic blood pressure before anesthesia; DBP 2, diastolic blood pressure during anesthesia; Pulse 1, pulse before anesthesia; Pulse 2, pulse during anesthesia; Respiration 1, respiration before anesthesia; Respiration 2, respiration during anesthesia; SpO2 1, SpO2 before anesthesia; SpO2 2, SpO2 during anesthesia.
a Values are expressed as mean ± SD.
5. Discussion
The results of the present study, which aimed to compare the hemodynamic variables of the non-COVID-19 and COVID-19 patients during anesthesia showed that SBP before anesthesia and patient pulse before and during anesthesia were higher than in non-COVID-19 patients. Respiration and SpO2 before anesthesia were also lower in the COVID-19 than in the non-COVID-19 group. On the other hand, in COVID-19 patients, SBP, pulse, and respiration during anesthesia are significantly lower than before anesthesia, and also SpO2 during anesthesia is significantly higher than before anesthesia. Although the pulse before and during anesthesia showed a statistically significant difference between the two groups and was higher in the patient group, but the results of the intragroup comparison showed that in the COVID-19 and non- COVID-19 groups when receiving oxygen during surgery, the pulse was closer to a normal amount.
According to our best search in English and Persian sources, this is the first study that compares the hemodynamic status of the COVID-19 and non-COVID-19 patients before and during general anesthesia. Several studies have examined the hemodynamic status of patients in the intensive care unit or internal department, but in all of them, despite several different hemodynamic conditions, no hemodynamic instability was reported (1, 6, 14).
Some results of the present study showed high SBP before anesthesia which is in line with Caillon et al. who found that SBP increased in COVID-19 patients (15). This was due to more activity of the angiotensin-converting enzyme resulting from an increase in the virus or the effects of systemic inflammation. They emphasized that high SBP could be an accompanying factor or a marker of end-limb damage before it develops (15). Various studies have shown an association between the renin-angiotensin-aldosterone system and COVID-19. The COVID-19 virus uses the host-converting enzyme angiotensin II to enter lung cells. Angiotensin II levels are elevated due to the dysfunction of this enzyme. Angiotensin II is a vasopressor that causes high blood pressure (16). Stability of patients with COVID-19, even in deep sedation conditions of mechanically-ventilated patients, with a tendency to hypertension has been reported (17). Vicenzi et al. in 2020 showed that the COVID-19 elevated blood pressure and lowered lung function (18). They explained that activation of the renin-angiotensin-aldosterone system increases aldosterone. Aldosterone, an arterial vasoconstrictor, directly affects water and sodium retention as well as inflammation (19). Systemic vasoconstriction is physiological protection that allows perfusion of blood flow to the pulmonary regions. In this case, changes in the tone of the pulmonary arteries cause the redistribution of blood flow and ultimately expanded ventilation dead space (20). With the development of COVID-19, endothelial damage occurs, disrupting the pulmonary arteries and leading to ventilation-perfusion incompatibility, which is the primary cause of hypoxemia and also enhances thrombogenesis (21). In 2020, Guo et al. reported high SBP and hypoxemia with high pH in the arterial blood of the COVID-19 patients admitted to the intensive care unit (22). After the COVID-19 virus enters the cell and activates the renin-angiotensin-aldosterone system, angiotensin II and aldosterone increase, both of which affect renal hydrogen and bicarbonate and cause the acid-base disorder. They considered two classic and protective paths for this system. In the classical vasoconstriction pathway, cell proliferation, fibrosis, oxidative stress, and an increase in aldosterone occur, leading to metabolic alkalosis. However, the protective pathway leads to vasodilation, natriuresis, anti-inflammatory, and anti-proliferation. It has been proven that in COVID-19 the classical pathway is activated and the protective pathway is suppressed (22). Contrary to the results of the present study on systolic hypertension, Michard et al. in an online and international survey examined the hemodynamic status of 19 COVID-19 patients admitted to the intensive care unit (6). Echocardiographic results showed changes in heart function. Relative hypovolemia and hypotension were also reported, to the extent that moderate doses of norepinephrine were required. Most patients also needed vasopressor, which could not be due to the COVID-19 alone, as in most cases patients admitted received deep sedation, neuromuscular blockers, and positive pressure ventilation, which resulted in hypovolemia and hypotension (6).
Approximately 15 to 20% of patients with COVID-19 develop hypoxemia and require oxygen therapy and ventilation (8). In most patients who die from the disease, the disease progresses occultly and without shortness of breath, known as silent hypoxemia (23).
The effects of hypoxemia on cardiac output (24) and systemic vascular resistance (25) are well known. In hypoxemia to compensate and increase oxygen delivery, heart rate and blood volume increase with each beat, the arterial smooth muscle relaxes, and environmental resistance decreases (26). Of course, with the help of oxygen administration, arterial oxygen pressure and its saturation become normal, which is accompanied by improved cardiac output and systemic vascular resistance (14). In the present study, the pulse was higher than that of non-COVID-19 patients, probably due to hypoxemia and increased heart rate and blood volume per beat. Then, during anesthesia due to oxygen administration, the pulse is in the normal range. In addition, Sonzogni et al. found severe changes in hepatic blood flow with dilated portal vessels in an autopsy of the COVID-19 patients (27). This issue may explain the peripheral shunt, high cardiac index, and low systemic vascular resistance observed in the COVID-19 patients (27). In fact, it is believed pulmonary vasoconstriction reduces alveolar oxygen pressure in small units of the lungs, which reduces blood flow to the atelectasis areas by up to 50%, limiting intrapulmonary shunting and ultimately ventricular overload (28).
Another part of the results of this study showed a decrease in the number of breaths before anesthesia. Accordingly, Caravita et al. in 2020 examined the hemodynamic characteristics of patients with COVID-19 who required mechanical ventilation with a right heart catheterization (1). The results showed that in the COVID-19 patients’ cardiac output and heart rate and pulmonary artery pressure were higher and DBP and systemic vascular resistance were lower than the control group. Arterial and venous oxygen levels as well as the ratio of arterial oxygen pressure to respiratory oxygen fraction decrease in COVID-19 patients compared to controls which may explain part of the respiratory pathophysiology in the pandemic. Nevertheless, no description of invasive hemodynamics was reported in these patients. In the COVID-19 patients, lung compliance was mitigated by one-third of the normal range, and also lung compliance with perfusion diminished (1) which reflects impaired lung parenchyma, changes in surfactant due to viral infection, and severe respiratory failure (29). In 2020, Busana et al. examined systemic and pulmonary hemodynamic profiles in the early stages of COVID-19 in inpatients. The overall results showed hyperdynamic blood circulation (14). Also, a respiratory rate of fewer than 22 beats per minute and SBP of more than 100 mmHg, hypoxemia, mild hypocapnia, decreased mean arterial pressure and increased cardiac index and decreased systemic vascular resistance were reported. Despite that, right heart function, pulmonary artery pressure, and overall pulmonary resistance were within the normal range and were not affected by the pandemic. Their findings suggest that hypoxemia, viral sepsis, or peripheral shunting are possible mechanisms for vasodilation that are predominant in COVID-19 and may worsen gas exchange. Oxygenation disturbance can also be caused by severe ventilation-perfusion incompatibility (14). Pulmonary embolism and microthrombus are distinctive features of COVID-19 (30-32) that can lead to ventilation-perfusion incompatibility (33). However, normal pulmonary pressure and low pulmonary resistance have been reported in COVID-19 patients with high cardiac index, which does not mean the absence of embolism, but suggests that the lungs use an efficient capillary system in the early stages of the disease. At this time, dilation of the arteries takes precedence over obstruction of the arteries (14). On the other hand, COVID-19 has been shown to increase intrapulmonary neo angiogenesis (34), which can increase the vascular bed of the lungs and justify this finding. Also, defects in hypoxic vasoconstriction are a compensatory response to improve areas where the ventilation-to-perfusion ratio is reduced (35).
Consistent with the part of our findings on low SpO2, Alberdi-Iglesias et al. 2021 showed that SpO2 decreases in COVID-19 patients, which have a direct relationship with age, deterioration, and mortality (36). Mukhtar et al. in 2021 stated that low SpO2 is a serious risk factor in patients with COVID-19 (37). Also, Wilson-Baig et al. in 2021 found that SpO2 was lower than arterial blood oxygen in COVID-19 patients admitted to the intensive care unit (38). They suggested that high ferritin, d-dimer, or other proteins in patients with COVID-19 may adversely affect the signal-to-noise ratio, thereby reducing pulse oximetry accuracy. Second, arterial dilatation may cause venous beats, which in turn contribute to false SpO2 readings because venous oxyhemoglobin saturation is also measured in the pulsating vein. COVID-19 may also contribute to tissue hypoxia through microvascular complications. In addition, anaerobic respiration due to secondary infection by anaerobic bacteria in COVID-19 may inhibit mitochondrial cytochrome oxidase, resulting in hypoxia at the cellular level. Finally, the formation of a complex between the virus and hemoglobin may raise the absorption of red light relative to the absorption of infrared and thus lead to a decrease in SpO2 (38).
Regarding the comparison of hemodynamic variables before and during anesthesia in patients with COVID-19, it should be noted that SBP, pulse, and respiration during anesthesia were significantly lower than before anesthesia, and also SpO2 was significantly higher during anesthesia than before it. In the COVID-19 group, because blood pressure was high before anesthesia, with proper control and medication, blood pressure was lowered without any complications and returned to a normal range. In the non-COVID-19 group, because the blood pressure was within the normal range before anesthesia, there was no need to prescribe medication, so blood pressure was kept at the same level before and during anesthesia and there was no significant difference between the two groups. During anesthesia, due to oxygen administration in the COVID-19 patients, hypoxemia and high cardiac index disappeared and subsequently, the pulse was normalized. Also, due to ventilation and oxygenation during anesthesia, respiration and SpO2 levels were normalized in the COVID-19 patients, and patients did not show any acute clinical signs or/and complications, which was confirmed by Auld et al.’s study 2020 (10). They emphasized early intubation and standard lung ventilation strategies in the COVID-19 patients admitted to the intensive care unit. It was also believed that even severe respiratory disorders caused by COVID-19 improved with intubation and mechanical ventilation (10). One of the limitations of the present study is the lack of control over genetic differences and the nutrition of patients. The authors suggest that in future studies, hemodynamic variables in the COVID-19 patients be considered separately for different types of surgery as well as in the recovery unit.
5.1. Conclusions
According to the findings of the present study, in patients who do not have severe COVID-19 and acute clinical symptoms, despite several hemodynamic conditions, hemodynamic instability was not reported. Despite low respiration in in the COVID-19 patients, the respiration rate was in the normal range. In addition, although in the COVID-19 group the amount of blood pressure decreased during surgery, it was still within the normal range. Therefore, it can be said that in patients with COVID-19, surgery and anesthesia can be performed by observing standard principles in emergencies. However, more research is needed in this regard.