1. Background
Oral and dental diseases is considered one of the most common health problems in the world (1). Accordingly, poor oral hygiene is associated with various problems, such as periodontitis, tooth decay, tooth or gum pain and discomfort, infection, and even loss of teeth; (2) also increases the probability of chronic diseases such as diabetes, cardiovascular diseases, stroke, and respiratory disease (3).
Dental caries is still high in many developing countries, and almost half of the world’s population, or 3.5 billion people, suffers from tooth decay (4). Current evidence shows that risk factors for developing dental caries, including plaque formation, fermentable carbohydrates, cariogenic bacteria, the quantity and quality of saliva, host susceptibility, poor oral hygiene, and duration, play a role in the onset and progression of dental caries (5).
Growing evidence suggests that oral pathogens are identified as a key element in the formation of dental caries (6). Streptococcus mutans is one of the most effective cariogenic bacteria, which has a key role in dental caries by adhesion to oral and dental tissues; herein, carbohydrate fermentation results in lactic acid production and demineralization of enamel (7). Since Mechanical plaque removal is one of the most accepted methods of controlling plaque, there are many mechanical aids to remove or control dental plaque, including toothbrushes, dental floss, mouth rinses, and dentifrices (8). Brushing with toothpaste containing various chemical formulations by reducing the number and activity of dental plaque is considered the most common form of dental disease prevention worldwide (9).
Toothpaste is classified as a drug and, according to American Dental Association (ADA), pastes gels or powders are used with a toothbrush for cleaning dental plaque and improving of oral health (10). Commonly, a wide range of chemical agents, such as fluoride in toothpaste, is used for inhibitory effects by destroying the cell wall and disrupting enzymatic activity in plaque formation (11). However, it can be associated with adverse outcomes, including chemical resistance to the antibacterial agents also lead to local toxic effects such as irritation and desquamation of the oral mucosa (stomatitis, glossitis, gingivitis) or systemic side effects such as allergic toxic reactions (acute or chronic) and even dental staining (12).
Considering that the phytochemicals isolated from the plants have a large number of active biological components, they may be a good alternative to Barberry and Zizyphus jujuba, which are found in Birjand (a sunny city in eastern Iran, the capital of South Khorasan province). Berberine is the main compound of berberis, an isoquinoline alkaloid produced by Berberis vulgaris L. cultivated in Asia and Europe with different pharmacological effects such as anti-inflammatory, antimicrobial, antifungal, and antiviral effects in both in vitro and in vivo studies (13). Also, Z. jujuba Mill, which is cultivated mainly in Europe and most of Asia with many biologically active components, especially alkaloids and Saponin, which have antioxidant, anti-inflammatory, and antiviral properties and also improve immune function (14).
2. Objectives
Since chemical agents such as fluoride in toothpaste are associated with adverse outcomes, including chemical resistance to the antibacterial agents, the phytochemicals isolated from the plants as herbal toothpaste seem to be a good alternative. Thus, the present study aimed to evaluate the antibacterial activity, plaque, and gingival index determination of herbal in comparison with standard toothpaste against S. mutans and Lactobacillus spp.
3. Methods
3.1. Study Setting
The present study is a randomized control clinical trial conducted at Birjand Dental School, Birjand, Iran, on 60 participants aged 18 - 28 years who were simply randomly divided into two groups of herbal and standard toothpaste (n = 30 each group).
3.2. Sample Collection
According to the results obtained from the study of Esfahanizadeh et al. (15) and using the formula for comparing two averages and considering 90% power and 99% confidence S1 = 0.2, S2 = 0.23, d = 0.2 and taking into account the 10% drop in the number of samples about 30 people in each group was calculated.
Participants were admitted to the study if meeting the following criteria: Age 18 - 30 years and a minimum of 24 healthy teeth. Exclusion criteria included systemic disease, using drugs, periodontal disease, pregnancy, breastfeeding, smoking, and a history of allergies to toothpaste or herbal medicine.
3.3. Toothpaste Preparation
A non-Iranian toothpaste (crest cavity protection) and herbal toothpaste prepared from Berberine and Saponin (main compounds of Barberry and Z. jujuba) respectively in the department of Pharmacology, Mashhad University of medical sciences were used in this study. All toothpaste was prepared in the lab section of the department of Pharmacology and separately packaged in label-free tubes with the same color of herbal toothpaste for two groups as those participants were completely blind. All subjects brushed their teeth for 3 min 2 times a day with the Bass method.
3.4. Gingival Health Assessment
GI and PI index: To determination of the GI at the onset of the study and 3, 5, and 7 months after toothpaste usage, each of the four areas of the facial, mesial, distal, and lingual teeth is examined and received a score from zero to 3 as normal gum and without inflammation: Zero; mild inflammation: Slight color change and very little edema without bleeding during the probe: 1; moderate inflammation: Edema and inflammation with bleeding during the probe: 2; and severe inflammation: Severe and marked edema and inflammation, ulceration and tendency to bleed spontaneously: 3.
PI for each tooth was determined as the average score of 4 tooth levels (buccal/labial and distal and lingual surfaces of teeth) and was scored as without plaque: Zero, a very thin plaque clinging to the edge of the free gum and adjacent teeth: 1; medium accumulation of plaque on the edge of the gums visible to the naked eye: 2; and excessive plaque buildup between the gingival pockets or on the teeth and gingival margins: 3 (16). Then, oral cavity was examined for any allergic or adverse reaction on hard and soft tissues.
3.5. Sampling and Microbial Assay
Saliva sampling: Saliva samples were taken from the participants 4 times onset of the study, 3, 5, and 7 months after using toothpaste. The participants were educated not to eat or drink except water and not perform physical exercise for at least two h before the saliva collection. Participants were asked to swallow their saliva and at least 1 mL of their non-induced saliva collection in a sterile container for 5 minutes (17). The saliva sample was then transferred to the laboratory in less than 30 minutes for S. mutans and Lactobacillus spp. culture.
3.5.1. Bacteria Culture
The collected saliva was diluted ten-fold as serial dilution by sterile normal saline. Diluted saliva was cultured in duplicate on Mitis salivarius bacitracin (MSB) agar (Himedia, India) supplemented by potassium tellurite and MRS agar (Merck, Germany) for isolation of S. mutans and Lactobacillus spp., respectively. The plates were incubated in a candle jar with a microaerophilic atmosphere at 37° for 48 h. Colonies of S. mutans identified with morphologic characteristics (0.5 mm convex undulated colonies of light blue color). Colonies of Lactobacillus spp. were characterized by small grayish-white. Colony-forming units (CFUs) per mL of saliva were calculated based on the mean of the counted colonies and saliva dilution rates (18).
3.6. Statistical Analysis
Obtained data were analyzed using SPSS 19 software. The normality of the data was evaluated using Kolmogorov-Smirnov statistical test. Mann-Whitney test was used to compare the two groups with each other, and Friedman and Wilcoxon's tests were to analyze the changes during the onset of the study and three, five, and seven months after toothpaste usage. The criterion for the significant difference was P < 0.05.
4. Results
This study was conducted on 60 patients in two study groups. The mean age in the herbal toothpaste group was 23.5 ± 2.1 years, and in the standard toothpaste group was 23.3 ± 2.01 years (P = 0.56). In the herbal and standard toothpaste groups, 60% and 50% of subjects were male, respectively (P = 0.43). So no significant statistical difference was observed in terms of age and gender in the two studied groups.
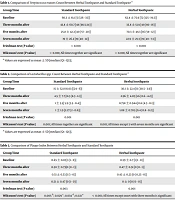
The levels of S. mutans and Lactobacillus spp. in saliva at the onset of the study and 3, 5, and 7 months after toothpaste usage are shown in Tables 1 and 2. Most participants had high levels of salivary S. mutans and Lactobacillus spp. at baseline. The results showed that the mean number of S. mutans and Lactobacillus spp. significantly decreased in both herbal and standard toothpaste groups compared to baseline after seven months of follow-up. There was no statistically significant difference between S. mutans levels in the three follow-up times between the herbal and standard toothpaste groups. In the case of Lactobacillus spp., the results show a significant decrease in the number of Lactobacillus spp. in the group using herbal toothpaste compared to the standard in 3 months (P = 0.02) and seven months (P = 0.002) of follow-up.
The mean of PI and GI at the baseline and 3, 5, and 7 months after herbal and standard toothpaste usage was shown in Tables 3 and 4. The results showed that the PI and GI significantly decreased in both herbal and standard toothpaste groups compared to baseline after seven months of follow-up. The PI significantly decreased in the herbal toothpaste group compared to the standard in the 5-month (P = 0.02) and 7-month (P < 0.001) follow-ups. Also, the GI significantly decreased in the herbal toothpaste group compared to the standard in the 5-month and 7-month follow-ups (P = 0.03).
| Group/Time | Standard Toothpaste | Herbal Toothpaste | Mann-Whitney (P Value) |
|---|---|---|---|
| Baseline | 81.1 ± 60.3 [50 (35 - 126)] | 75.9 ± 62.4 [72 (19.5 - 125)] | 0.56 |
| Three months after | 60.7 ± 43.8 [48 (24-98.5)] | 51.9 ± 35.8 [49 (16 - 80)] | 0.46 |
| Five months after | 42.3 ± 25.8 [40 (20 - 57)] | 48.3 ± 70.3 [36 (12 - 59)] | 0.56 |
| Seven months after | 26.2 ± 10 [19 (12 - 40)] | 25.5 ± 22.9 [19 (7.5 - 40)] | 0.54 |
| Friedman test (P value) | < 0.001 | < 0.001 | |
| Wilcoxon’s test (P value) | < 0.001; All times together are significant | < 0.001; All times together are significant |
Comparison of Streptococcus mutans Count Between Herbal Toothpaste and Standard Toothpaste a
| Group/Time | Standard Toothpaste | Herbal Toothpaste | Mann-Whitney (P Value) |
|---|---|---|---|
| Baseline | 52.8 ± 18.03 [15 [(6 - 24)] | 22.4 ± 30.1 [11 (3.9 - 30.5)] | 0.77 |
| Three months after | 7.7 ± 11.3 [4.5 (1.1 - 8.3)] | 2.96 ± 4.03 [1.6 (0.6 - 4.2)] | 0.02* |
| Five months after | 3.5 ± 5.9 [1 (0.4 - 3.5)] | 0.944 ± 0.758 [0.6 (0.1 - 4.3)] | 0.26 |
| Seven months after | 1.9 ± 2.27 [1 (0.6 - 2)] | 0.705 ± 1.08 [0.4 (0.2 - 0.8)] | 0.002* |
| Friedman test (P value) | 0.001 | 0.001 | |
| Wilcoxon’s test (P value) | 0.001; All times together are significant | 0.001; All times except 5 with seven months are significant |
Comparison of Lactobacillus spp. Count Between Herbal Toothpaste and Standard Toothpaste a
| Group/Time | Standard Toothpaste | Herbal Toothpaste | Mann-Whitney (P Value) |
|---|---|---|---|
| Baseline | 0.83 ± 0.65 [1 (0 - 1] | 0.7 ± 0.59 [1 (0 - 1)] | 0.43 |
| Three months after | 0.73 ± 0.58 [1 (0 - 1)] | 0.47 ± 0.51 [0 (0 - 1)] | 0.08 |
| Five months after | 0.53 ± 0.51 [1 (0 - 1)] | 0.23 ± 0.43 [0 (0 - 0.25)] | 0.02 b |
| Seven months after | 0.47 ± 0.51 [0 (0 - 1)] | 0 ± 0 [0 (0 - 0)] | < 0.001 a |
| Friedman test (P value) | 0.003 | 0.001 | |
| Wilcoxon’s test (P value) | 0.005 b; 0.029 c; 0.034 d; 0.021 e | < 0.001; All times except onset with three months is significant |
Comparison of Plaque Index Between Herbal Toothpaste and Standard Toothpaste
| Group/Time | Standard Toothpaste | Herbal Toothpaste | Mann-Whitney (P Value) |
|---|---|---|---|
| Baseline | 1.07 ± 0.64 [1 (1 - 1.25)] | 1.07 ± 0.69 [1 (1 - 2)] | 0.99 |
| Three months after | 1.03 ± 0.49 [1 (1 - 1)] | 0.87 ± 0.51 [1 (1 - 1)] | 0.19 |
| Five months after | 0.87 ± 0.35 [1 (1 - 1)] | 0.63 ± 0.49 [1 (0 - 1)] | 0.03 b |
| Seven months after | 0.8 ± 0.41 [1 (1 – 1)] | 0.53 ± 0.51 [1 (0 - 1)] | 0.03 b |
| Friedman test (P value) | 0.013 | < 0.001 | |
| Wilcoxon’s test (P value) | 0.033 b; 0.008 c | 0.003 d; 0.002 b; 0.008 c |
Comparison of Gingival Index Between Herbal Toothpaste and Standard Toothpaste
5. Discussion
Since plaque is considered the main etiological factor for several oral diseases, optimal plaque control, including mechanical control, is important for preventing gingivitis, periodontitis, and dental caries (19). Hence, various chemical substances have been developed to control periodontal diseases that may lead to harmful local and systemic side effects when used for a long time (11, 12). Therefore, research has recently been done on alternative products to synthetic chemicals.
The results of our study showed that the mean of PI and GI was significantly reduced in herbal and standard toothpaste groups after seven months of follow-up examination as compared to baseline. Our study is in line with a randomized clinical trial study by Taghavi et al., in which PI and GI scores had significant differences between baseline and follow-up examinations (20). Also, the comparison between the two groups in our study showed that the PI and GI were significantly reduced at five months and seven months after using herbal toothpaste compared to standard toothpaste. Our study is in line with a double-blind clinical trial study by Makarem and Asodeh (21) that showed berberine gel significantly reduced microbial plaque and gingivitis in school-aged children compared with placebo.
Several studies have shown the effect of herbal toothpaste on plaque control compared to standard types. Similar results were observed by de Oliveira et al. that the Aloe vera plant product was effective in reducing plaque but showed less effectiveness of the plant product on gingivitis and gingival bleeding compared to standard types (22). Also, the study by Amoian et al. on 40 participants over four weeks in 2 groups of control and Calendula flower extract toothpaste that was found significant reductions in PI, GI, and bleeding on probing (BoP) in Calendula toothpaste than control toothpaste in weeks 2,3 and 4 (23). The reducing effects of PI and GI scores can be attributed to the anti-inflammatory effects of berberine and Saponin (22, 23). Several mechanisms have been mentioned for the antimicrobial effects of berberine, such as inhibition of cell division, disrupting of protein synthesis, inhibiting of enzyme activity, and destroying the surface structures of bacteria (24-26). Two mechanisms exert the effect of saponins on the biofilm system: First, surfactant properties by surface tension reduction in an aqueous solution can form micelles, and this, in critical point concentration, can structurally change biological macromolecules. Second, perforation and rupture of the cell membrane of bacteria by binding to surface sterols in eukaryotic cell membranes (27). Considering that the chemicals used in toothpaste, mouthwashes, etc., when used regularly, may cause undesirable side effects such as changes in mouth taste, tooth discoloration, and ulceration of the oral mucosa (22) however, there was no adverse reaction with herbal toothpaste in our study during follow-up.
Our study showed that the mean number of S. mutans and Lactobacillus spp. significantly decreased in both groups of using herbal and standard toothpaste compared to baseline after seven months of follow-up, but there was no statistically significant difference between S. mutans levels in the three follow-up times between the herbal and standard toothpaste groups. Our result is in line with Patil et al.'s (28) study on 100 students of 4 - 6-year-old children using Himalaya herbal dental cream or cheerio gel containing fluoride (n = 50), despite the 90% reduction of saliva S. mutans after a 5-month follow-up period in two groups, no statistically significant difference observed between the groups (26). Our study showed that the herbal toothpaste was more effective than the standard in reducing Lactobacillus spp., especially after seven months of use. Many studies have been conducted on reducing S. mutans and Lactobacillus spp. using commercial and herbal toothpaste. Our result is in line with the Biria et al.’s study (29) that using herbal toothpaste containing bamboo salt and non-herbal conventional toothpaste leads to a significant reduction of S. mutans and Lactobacillus spp., after a 4-week follow-up; however, unlike our study, it is not statistically different between the two groups in the case of Lactobacillus spp. It seems that the duration of using herbal toothpaste is an effective factor in reducing the amount of Lactobacillus spp. Mehta et al. study (30) on 55 children aged 8 - 14 years showed herbal (Freshol) mouthwash and chlorhexidine can significantly reduce the amount of S. mutans saliva, PI, and GI score during periods of 10 days. Our study is in line with Ferrazzano et al. (31) on 66 healthy individuals of 12 - 18 years in two groups of green tea extract, or placebo mouth rinse exhibited a statistically significant reduction in colony counts of Lactobacilli saliva sample relative to the control group.
5.1. Conclusions
Our study showed considering the new herbal toothpaste for seven months in vivo that relatively well-controlled gingivitis and dental plaque also, there were no adverse effects it can be used effectively. Of course, more studies are needed to evaluate its effectiveness.
5.2. Limitations
First, in this study, only two of the important cariogenic bacteria in dental biofilm formation were examined so that it can study other cariogenic bacteria and at the molecular level. Second, the effect of new toothpaste in this study was not examined on special groups, including elderly subjects also pregnant and breastfeeding women.