1. Background
An elevated level of bilirubin in the blood is the most common and benign problem among newborns, and it is the most important factor in the evaluation of neonatal jaundice in newborns (1, 2). Depending on the cause of jaundice, it can be present from the birth, or emerge at any time during infancy. Jaundice emerges in 60% of full-term neonates and 80% of preterm infants in the first week of life. Jaundice usually starts in the face and, as levels of bilirubin increase, it extends to the abdomen and legs. Generally, 6 to 7% of full-term babies’ bilirubin levels are greater than 12., and fewer than 3% have bilirubin levels higher than 15 (1). If the onset and duration of jaundice is very different with the increase in bilirubin in the physiologic jaundice, the jaundice is considered as pathologic. Those with progressive jaundice risk developing hemolysis or septicemia transcutaneous and show high values of bilirubin, so their bilirubin serum levels should be measured (1). As mentioned, jaundice is a common problem in infants. If severe indirect hyperbilirubinemia if left untreated, it is neurotoxic. Direct hyperbilirubinemia is often a sign of serious liver disease or systemic disease. The greatest risk associated with hyperbilirubinemia is creating neurological dysfunction caused by high bilirubin (1). Side effects of acute bilirubin hyperbilirubinemia are encephalopathy bilirubin and kernicterus (3). To prevent these complications, it is suggested that all infants be screened for hyperbilirubinemia (3). The determination of plasma bilirubin levels is a common cause for taking blood from infants, but blood draws cause pain in newborn babies; this created stress may leave long-lasting results. In addition, there is a risk of infection and scarring associated with blood draws. Heel pricks causes sudden increases in hypertension, which that may lead to bleeding vessels in preterm infants. In addition, the amount of blood lost during a blood draw should be reduced to the lowest level as possible, so decreasing the number of blood draws in infants is very important (4). According to the American pediatric association’s 2004 guidelines, all infants should be evaluated for hyperbilirubinemia. Both methods (total serum bilirubin, or TSB and transcutaneus billirubin, or TCB) are accepted for assessment (4). TCB can measure serum bilirubin levels with a non-invasive method of measuring the amount of light passing through the skin without the need for a blood sample (3). TCB is used for screening for hyperbilirubinemia and makes it possible to reduce the frequency of blood sampling. BiliChek contains 137 wavelengths between 380 - 700 nm and analyzes them through intermediate wavelength (4). Although the existing guidelines allow for use of TCB devices for the evaluation of jaundice in term and near-term neonates, the accuracy of TCB devices for the estimation of serum bilirubin in preterm infants remains unclear (5). On the other hand, there are controversies between the various studies about the value and use of BiliChek in premature infants. Ebbesen et al. (2012) found that BiliChek is suitable for screening in both NICU and healthy newborn infants with jaundice, with regarding the need for phototherapy (6), and Willems et al. (2004) showed that the BiliChek application in the NICU environment has the potential to reduce the number of blood samples taken by 40% (7). However, several studies have provided results for correlation coefficients between TCB and TSB, and the pooled estimates, according to the site of measurement, were 0.35 - 0.65 with 95% confidence interval. These studies argue that other devices, such as JM-103, exhibited better precision than the BiliChek and the application of BiliChek needs more study (8-11).
2. Objectives
We intended to examine the relationship between levels of TCB (BiliChek method) and TSB in Iranian neonates and study the role of different factors in this regard, such as infants’ gender, gestational age, gestational age, birth weight, maternal age, type of delivery, birth rank and kind of jaundice.
3. Patients and Methods
In this study which was performed at Amiralmomenin hospital in Semnan, Iran, we considered a similar study by Badiee et al. (2012), which examined 64 healthy infants who had gestational age of 37 weeks and jaundice as the sole cause of hospitalization. Infants hospitalized due to other diseases, such as sepsis, were excluded from the study (12). Since all babies are subject to a blood sample upon hospital admission of to determine their serum total bilirubin, this study, using BiliChek (Spect Rx, Norcross, GA, USA), we measured their transcutaneous bilirubin in three areas: the forehead, sternum and thigh. The maximum interval between blood sampling and BiliChek was 30 minutes (8-11). The BiliChek apparatus was a fibrotic instrument assessing multiple wavelengths by spectral reflectance. It was calibrated before initiating the study. Two investigators who did not have prior knowledge of the capillary blood bilirubin concentration measured the TCB. Then, patients' information, such as age, type of jaundice and bilirubin values, were recorded in a checklist. All data were analyzed with SPSS 20 (SPSS Inc., Chicago, USA), and P < 0.05 were considered to indicate statistical significance. For comparing the means of TSB and TCB, we applied the Paired Student’s t-test. We also used Pearson’s correlation coefficients and the ROC curve in MedCalk software for the data analysis.
4. Results
4.1. Laboratory
The laboratory bilirubin mean ± SD is 14.4 ± 4.0 mg per deciliter, respectively (Table 1).
| Type and location | Standard Deviation | The Standard Error | Mean | Maximum | Minimum |
|---|---|---|---|---|---|
| Forehead | 3.5 | 0.44 | 13.8 | 20.5 | 7.0 |
| Chest | 3.5 | 0.44 | 12.8 | 19.6 | 6.1 |
| Thigh | 2.6 | 0.33 | 9.7 | 14.2 | 5.0 |
| Laboratory | 4.0 | 0.50 | 14.0 | 23.8 | 6.2 |
aFrequency = 64.
4.2. Skin
The amount of bilirubin mean ± SD in the forehead, chest and thigh are 13.8 ± 3.5 mg per deciliter, and 12.8 ± 3.5 mg per deciliter and (9.7± 2.6 milligrams per deciliter, where BiliChek values in all three areas are lower than laboratory values (Table 1).
4.3. Determining the Cut-off Point in the Diagnosis of Hyperbilirobinemia
4.3.1. The cut-off Point in the Diagnosis of Forehead Bilirubin Hyperbilirubinemia
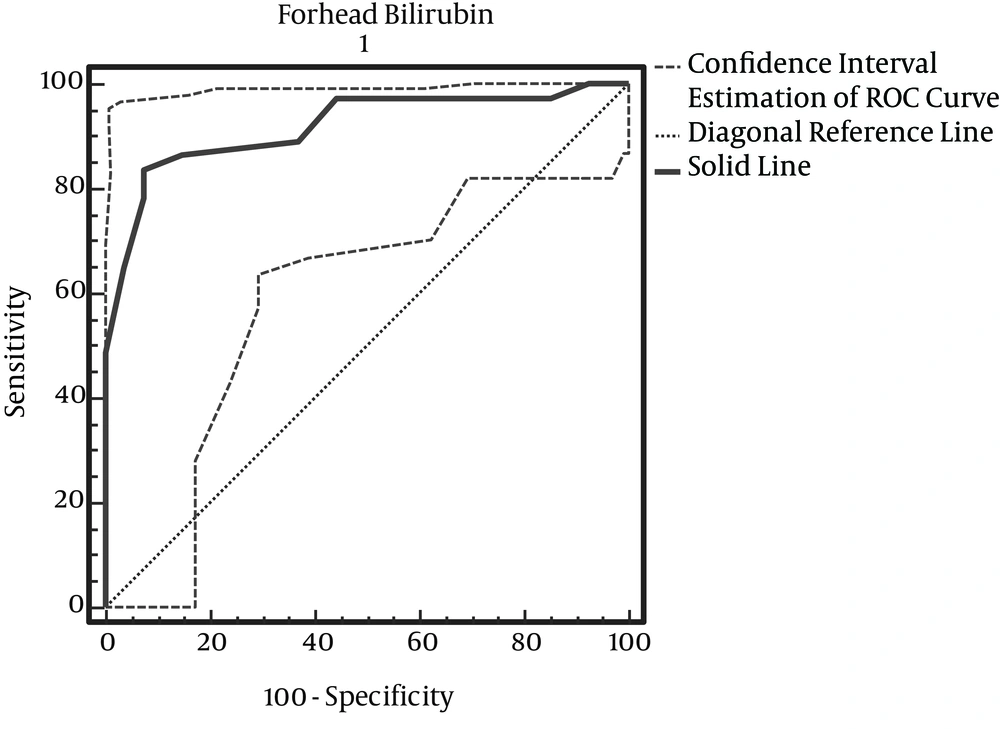
According to Table 2, with maximum accuracy, the cut-off point of forehead bilirubin is 13.3 (Figure 1).
| Criterion | Sensitivity | 95% CI | Specificity | 95% CI | +LR | -LR | +PV | -PV |
|---|---|---|---|---|---|---|---|---|
| ≥ 7 | 100.00 | 90.4 - 100.0 | 0.00 | 0.0 - 12.9 | 1.00 | NA | 57.8 | NA |
| > 7 | 100.00 | 90.4 - 100.0 | 7.41 | 1.1 - 24.3 | 1.08 | 0.00 | 59.7 | 100.0 |
| > 7.9 | 97.30 | 85.8 - 99.5 | 14.81 | 4.3 - 33.7 | 1.14 | 0.18 | 61.0 | 80.0 |
| > 8.8 | 97.30 | 85.8 - 99.5 | 25.93 | 11.2 - 46.3 | 1.31 | 0.10 | 64.3 | 87.5 |
| > 9.7 | 97.30 | 85.8 - 99.5 | 37.04 | 19.4 - 57.6 | 1.55 | 0.07 | 67.9 | 90.9 |
| > 10.6 | 97.30 | 85.8 - 99.5 | 55.56 | 35.3 - 74.5 | 2.19 | 0.05 | 75.0 | 93.8 |
| > 11.5 | 89.19 | 74.6 - 96.9 | 62.96 | 42.4 - 80.6 | 2.41 | 0.17 | 76.7 | 81.0 |
| > 12.4 | 86.49 | 71.2 - 95.4 | 85.19 | 66.3 - 95.7 | 5.84 | 0.16 | 88.9 | 82.1 |
| > 13.3 | 83.78 | 68.0 - 93.8 | 92.59 | 75.7 - 98.9 | 11.31 | 0.18 | 93.9 | 80.6 |
| > 14.2 | 78.38 | 61.8 - 90.1 | 92.59 | 75.7 - 98.9 | 10.58 | 0.23 | 93.5 | 75.8 |
| > 15.1 | 64.86 | 47.5 - 79.8 | 96.30 | 81.0 - 99.4 | 17.51 | 0.36 | 96.0 | 66.7 |
| > 16 | 48.65 | 31.9 - 65.6 | 100.00 | 87.1 - 100.0 | NA | 0.51 | 100.0 | 58.7 |
| > 16.9 | 35.14 | 20.2 - 52.5 | 100.00 | 87.1 - 100.0 | NA | 0.65 | 100.0 | 52.9 |
| > 17.8 | 16.22 | 6.2 - 32.0 | 100.00 | 87.1 - 100.0 | NA | 0.84 | 100.0 | 46.6 |
| > 18.7 | 8.11 | 1.8 - 21.9 | 100.00 | 87.1 - 100.0 | NA | 0.92 | 100.0 | 44.3 |
| > 19.6 | 2.70 | 0.5 - 14.2 | 100.00 | 87.1 - 100.0 | NA | 0.97 | 100.0 | 42.9 |
| > 20.5 | 0.00 | 0.0 - 9.6 | 100.00 | 87.1 - 100.0 | NA | 1.00 | 42.2 |
4.3.2. Sternum Bilirubin Cut-off Point in the Diagnosis of Hyperbilirubinemia
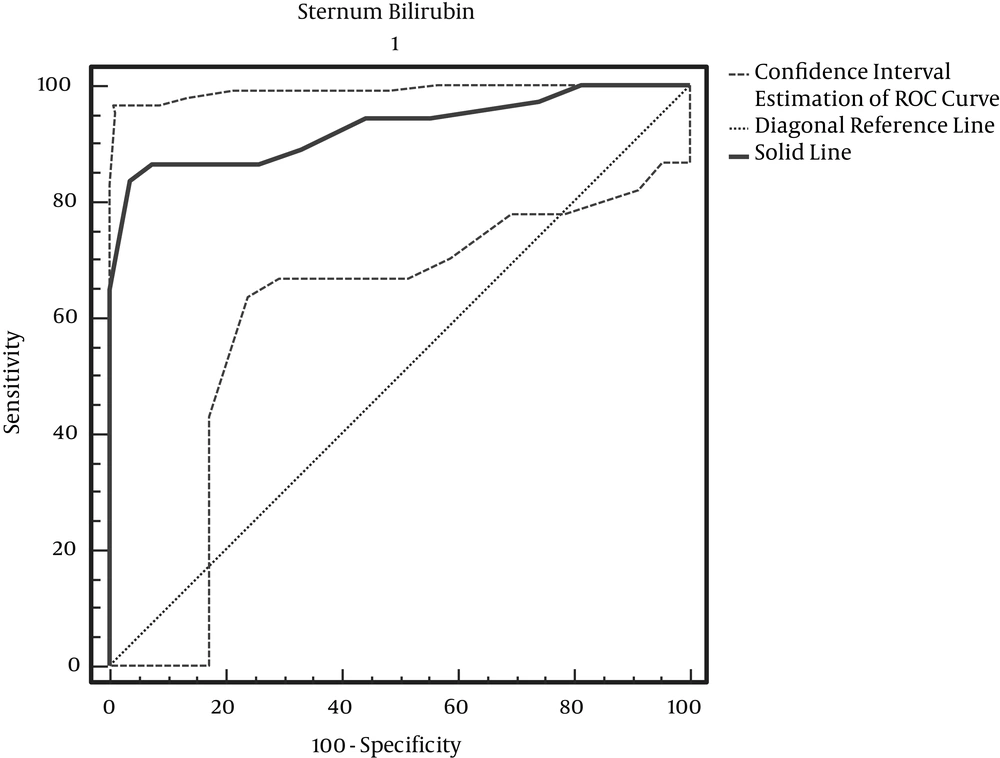
According to Table 3, with maximum accuracy, the Sternum Bilirubin cut-off point is 13.3 (Figure 2).
| Criterion | Sensitivity | 95% CI | Specificity | 95% CI | +LR | -LR |
|---|---|---|---|---|---|---|
| ≥ 6.1 | 100.00 | 90.4 - 100.0 | 0.00 | 0.0 - 12.9 | 1.00 | NA |
| > 6.1 | 100.00 | 90.4 - 100.0 | 11.11 | 2.5 - 29.2 | 1.12 | 0.00 |
| > 7 | 100.00 | 90.4 - 100.0 | 18.52 | 6.4 - 38.1 | 1.23 | 0.00 |
| > 7.9 | 97.30 | 85.8 - 99.5 | 25.93 | 11.2 - 46.3 | 1.31 | 0.10 |
| > 8.8 | 94.59 | 81.8 - 99.2 | 44.44 | 25.5 - 64.7 | 1.70 | 0.12 |
| > 9.7 | 94.59 | 81.8 - 99.2 | 55.56 | 35.3 - 74.5 | 2.13 | 0.10 |
| > 10.6 | 89.19 | 74.6 - 96.9 | 66.67 | 46.0 - 83.4 | 2.68 | 0.16 |
| > 11.5 | 86.49 | 71.2 - 95.4 | 74.07 | 53.7 - 88.8 | 3.34 | 0.18 |
| > 12.4 | 86.49 | 71.2 - 95.4 | 92.59 | 75.7 - 98.9 | 11.68 | 0.15 |
| > 13.3 | 83.78 | 68.0 - 93.8 | 96.30 | 81.0 - 99.4 | 22.62 | 0.17 |
| > 14.2 | 64.86 | 47.5 - 79.8 | 100.00 | 87.1 - 100.0 | NA | 0.35 |
| > 15.1 | 43.24 | 27.1 - 60.5 | 100.00 | 87.1 - 100.0 | NA | 0.57 |
| > 16 | 29.73 | 15.9 - 47.0 | 100.00 | 87.1 - 100.0 | NA | 0.70 |
| > 16.9 | 16.22 | 6.2 - 32.0 | 100.00 | 87.1 - 100.0 | NA | 0.84 |
| > 17.8 | 5.41 | 0.8 - 18.2 | 100.00 | 87.1 - 100.0 | NA | 0.95 |
| > 18.7 | 2.70 | 0.5 - 14.2 | 100.00 | 87.1 - 100.0 | NA | 0.97 |
| > 19.6 | 0.00 | 0.0 - 9.6 | 100.00 | 87.1 - 100.0 | NA | 1.00 |
4.3.3. Femor Bilirubin Cut-off Point in the Diagnosis of Hyperbilirubinemia
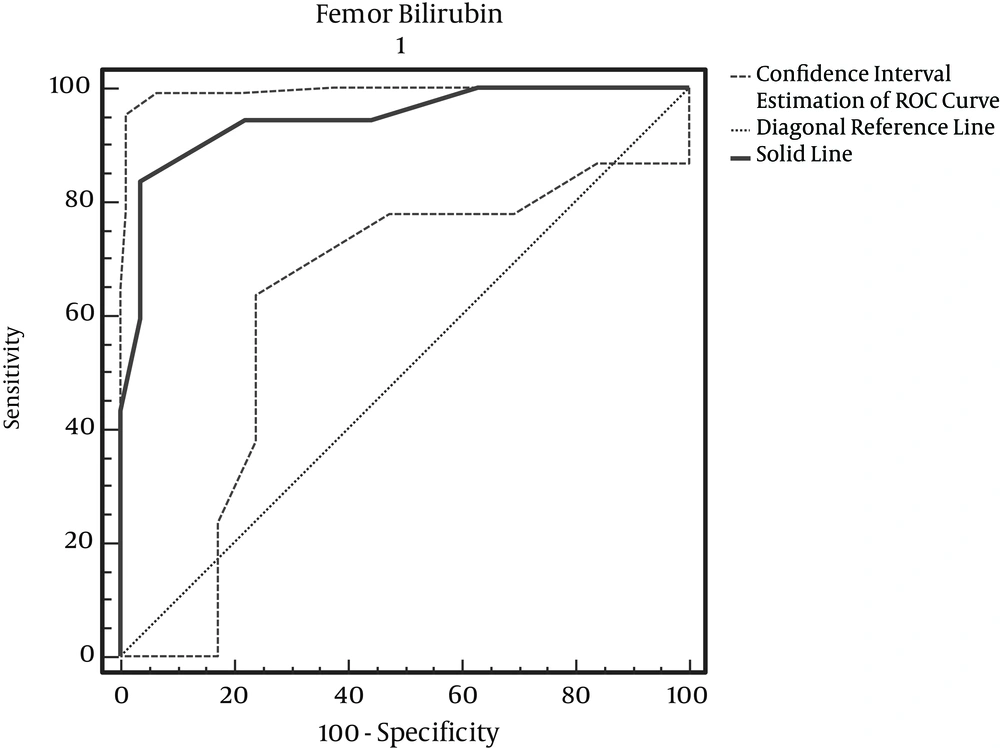
According to Table 4, with maximum accuracy, the femur bilirubin cut-off point is 9.7 mg/dl (Figure 3).
| Criterion | Sensitivity | 95% CI | Specificity | 95% CI | +LR | -LR | +PV | -PV |
|---|---|---|---|---|---|---|---|---|
| ≥ 5.2 | 100.00 | 90.4 - 100.0 | 0.00 | 0.0 - 12.9 | 1.00 | NA | 57.8 | NA |
| >5.2 | 100.00 | 90.4 - 100.0 | 22.22 | 8.7 - 42.3 | 1.29 | 0.00 | 63.8 | 100.0 |
| > 6.1 | 100.00 | 90.4 - 100.0 | 37.04 | 19.4 - 57.6 | 1.59 | 0.00 | 68.5 | 100.0 |
| > 7 | 94.59 | 81.8 - 99.2 | 55.56 | 35.3 - 74.5 | 2.13 | 0.10 | 74.5 | 88.2 |
| > 7.9 | 94.59 | 81.8 - 99.2 | 62.96 | 42.4 - 80.6 | 2.55 | 0.09 | 77.8 | 89.5 |
| > 8.8 | 94.59 | 81.8 - 99.2 | 77.78 | 57.7 - 91.3 | 4.26 | 0.07 | 85.4 | 91.3 |
| > 9.7 | 83.78 | 68.0 - 93.8 | 96.30 | 81.0 - 99.4 | 22.62 | 0.17 | 96.9 | 81.2 |
| > 10.6 | 59.46 | 42.1 - 75.2 | 96.30 | 81.0 - 99.4 | 16.05 | 0.42 | 95.7 | 63.4 |
| > 11.5 | 43.24 | 27.1 - 60.5 | 100.00 | 87.1 - 100.0 | NA | 0.57 | 100.0 | 56.3 |
| > 12.4 | 21.62 | 9.9 - 38.2 | 100.00 | 87.1 - 100.0 | NA | 0.78 | 100.0 | 48.2 |
| > 13.3 | 8.11 | 1.8 - 21.9 | 100.00 | 87.1 - 100.0 | NA | 0.92 | 100.0 | 44.3 |
| > 14.2 | 0.00 | 0.0 - 9.6 | 100.00 | 87.1 - 100.0 | NA | 1.00 | NA | 42.2 |
4.4. Comparison of Three Methods for Diagnosis of Hyperbilirobinemia
According to Table 5, the three methods of chest, thigh, and forehead evaluation do not have significant differences in diagnoses.
| Area | AUC | SE | 95% CI |
|---|---|---|---|
| Bilirubin-thigh | 0.945 | 0.028 | 0.858 to 0.986 |
| Bilirubin-forehead | 0.918 | 0.035 | 0.822 to 0.972 |
| Bilirubin-chest | 0.927 | 0.033 | 0.834 to 0.977 |
4.5. Pair Comparisons of Both Skin Methods
The results show the differences between forehead and thigh bilirubin levels are 0.028 ± 0.027 with P = 0.337, and the difference between chest and thigh bilirubin is 0.025 ± 0.018 with P = 0.480, and the difference between the forehead and chest bilirubin is 0.025 ± 0.009, with P = 0.720.
5. Discussion
For the determination of hyperbilirubinemia in healthy infants, we still require invasive blood sampling, and this study was performed to determine if TCB could be a reliable screening test. Our results revealed a considerable positive correlation between TCB measurements and TSB. In previous studies, the reported range of the correlation coefficient between TCB and TSB in newborns was between 0.39 and 0.92. In our study, the correlation coefficients and the pooled estimates, according to the site of measurement, were as follows: forehead: 0.895 (95% confidence interval [CI]): (0.82 – 0.97); thigh: 0.925 (95% CI: 0.86 – 0.99); and chest: 0.915 (95% CI: 0.83 – 0.98). Also, the correlation coefficients for area of the forehead, in studies by Willems (2004), Badiee (2012), and in a study by Szabo et al. (2004) for areas of the forehead chest was more than 0.75; this was similar to the results of our study (7, 12, 13). In determining the cut-off point, the results obtained from the ROC curve showed that the cut off point for using BiliChek on the forehead is 13.3 milligrams per deciliter, with sensitivity and specificity levels of 83.87% and 92.59%, respectively. For the chest, it is 13.3 milligrams per deciliter, with sensitivity and specificity levels of 83.87% and 96.30%, respectively. For the thigh, it is 9.7 mg per deciliter, with sensitivity and specificity levels of 83.87%, and 96.30%, respectively. Among the above three areas, there is no significant difference in which skin method is used for predicting hyperbilirubinemia. In pair comparisons of both skin methods, the results show the difference between bilirubin levels measured at the forehead and thigh as 0.028 ± 0.027, with P = 0.337; the difference between the chest and thigh bilirubin readings was 0.025 ± 0.018, with P = 0.480; and the difference between forehead and chest bilirubin results was 0.025 ± 0.009, with P = 0.720. According to comparisons of the two methods, it can be said that the average amount of bilirubin detected in both methods is similar and close to each other. In a study by Szabo et al. (2004), the correlation coefficients for the two methods of forehead and chest were 0.67 and 0.77, respectively, meaning that there is no significant difference in the two approaches (13). Our results also showed that variables such as type of delivery, birth rank, blood type, birth weight and maternal age are independent of hyperbilirubinemia levels, and there is no a significant relationship between them and the condition. There is only a significant relationship between bilirubin levels and birth age (P = 0.001). Badiee et al. (2012), in their study, demonstrated that the relationship between TCB and TSB is not influenced significantly by gestational age (12). Conversely, in the study by Knupfer et al. (14), the TCB – TSB correlation decreased with lowering gestational age. On the other hand, Willems and colleagues showed that the BiliChek method is a reliable screening device in very preterm infants (gestational age < 30 weeks). Similarly, De Luca et al. (15) found no effect of gestational age, gender or PH on TCB readings.
5.1. Conclusion
In our study, we determined that BiliChek can be used in the diagnosis of hyperbilirubinemia, but this use is limited and applies only to values higher than the cutoff points of 13.3 mg/dL on the forehead and chest and 9.7 mg/dL for thigh.