1. Background
De Quervain tenosynovitis was first described in 1985 by Fritz de Quervain (1). The extensor retinaculum holds the tendons of the extensor muscles in place and covers the first dorsal compartment, a fibro-osseous tunnel. Chronic overuse of the wrist may lead to swelling and thickening of the ligaments of the extensor retinaculum. As a result, entrapment of tendons, which pass through the first dorsal compartment of the wrist (under the fibers of the extensor retinaculum and over the radial styloid process), causes pain. Abductor pollicis longus and the extensor pollicis brevis tendons are affected by this condition (2-4). Pain and swelling on the thumb side of the wrist, impaired function of the thumb, and gripping problems are the most common symptoms of the de Quervain disease, worsening with an increase in the motion of the thumb (5, 6). Its prevalence, at the highest rate, is estimated 0.5% in males and 1.3% in females aged 40 - 60 years (7, 8).
Diagnosis is based on clinical manifestations and examinations, such as the Finklestein test, revealing the possibility of restricted tendon movement through a mixed maneuver, including ulnar deviation and first metacarpophalangeal joint flexion (3, 4). Other diagnostic tests, such as radiography, might be used to differentiate it from other causes of wrist pain, such as osteoarthritis and fractures (9).
Different therapies are suggested to treat de Quervain tenosynovitis; nonsteroidal anti-inflammatory drugs, injection of corticosteroids, immobilization with a splint, iontophoresis, therapeutic pulsed ultrasound, activity modification, and surgery are the main treatments of choice, which their outcomes are discussed in various studies (6, 10, 11). Along with these well-known therapies for musculoskeletal disorders, new treatment methods are suggested and evaluated every day, one of which is extracorporeal shockwave therapy. Primary studies on the efficacy of this modality in fracture healing utilized animal models (12-14).
Although the mechanism of action of extracorporeal shockwave therapy is not yet fully understood (15), satisfying outcomes made it a treatment of choice for various orthopedic disorders, such as plantar fasciitis, bone fractures, osteonecrosis, and tendinopathies (16-19). Although a few studies suggested that shockwave therapy is not as effective as non-invasive techniques, it is a satisfying, safe, and effective procedure with low complications compared to its advantages (15, 20-23). Studies evaluating the effect of shockwave therapy on different types of tenosynovitis are limited and, to the best of authors’ knowledge, only one study discussed its effects on de Quervain tenosynovitis thus far (24).
2. Objectives
Therefore, the present study aimed at assessing the efficacy of extracorporeal shockwave therapy in the treatment of de Quervain tenosynovitis.
3. Methods
3.1. Study Design
The current controlled, randomized, clinical trial was conducted on 26 patients (n = 13 in each group) with de Quervain tenosynovitis referred to physical medicine and rehabilitation centers affiliated to Isfahan University of Medical Sciences from 2015 to 2018.
3.2. Inclusion and Exclusion Criteria
The inclusion criteria were: receiving a diagnosis of de Quervain tenosynovitis, age over 18 years, and no history of undergoing physiotherapy modalities or corticosterone injection therapies for any reasons during the past month. All the patients completed the written informed consent form for participation in the study.
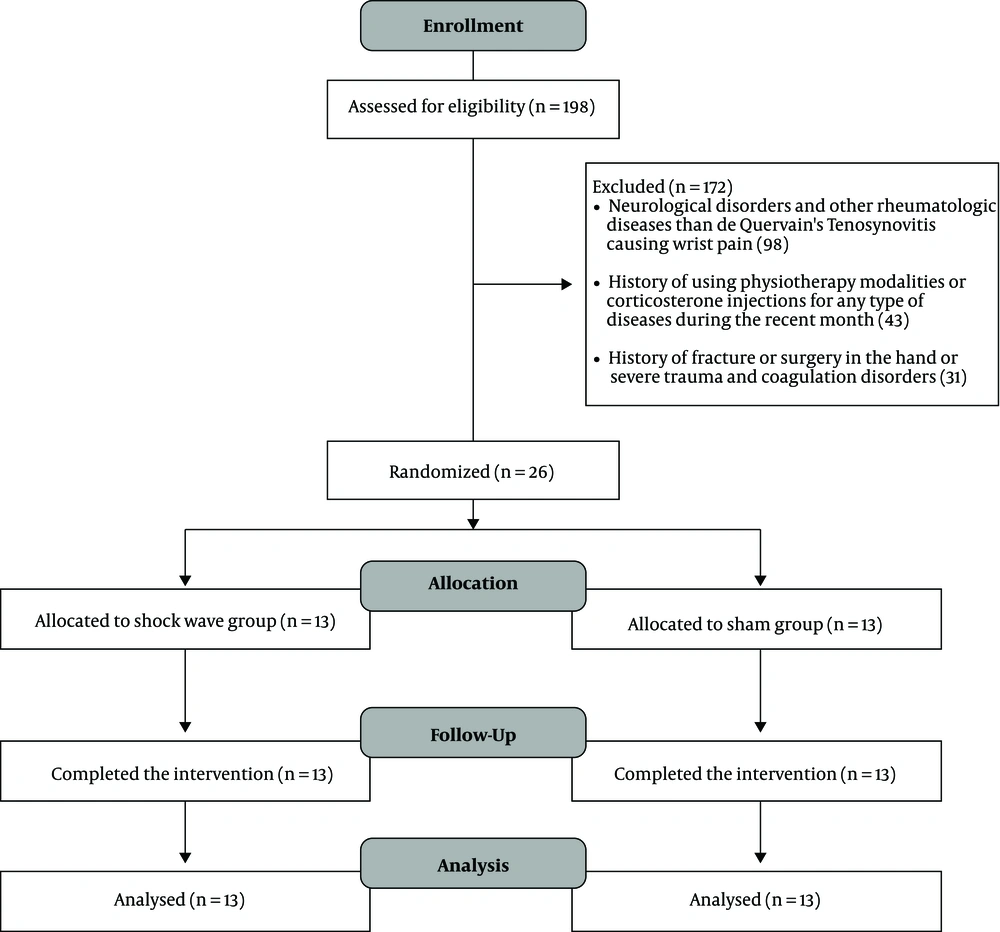
De Quervain tenosynovitis was diagnosed based on the standard criteria, including pain and tenderness in the lateral wrist and a positive result for the Finkelstein test. However, subjects were excluded from the study in case of other neurological disorders and rheumatologic diseases causing wrist pain, pregnancy, history of fracture or surgery on the hand, severe trauma, and coagulation disorders. Also, patients unwilling to continue the study during treatment, the ones with severe or acute complications after the intervention, and the ones who needed to alter the treatment were excluded from the study. At the study onset, demographic information of patients, including age, gender, and duration of disease, was recorded. Afterward, the patients were randomly divided into two parallel groups as extracorporeal shock wave therapy and sham by Random Allocation Software through the blocking method. The study had a single-blinded design, and patients were blind to allocations (Figure 1).
3.3. Clinical Assessments
In the shock wave therapy group, each patient first sat on a chair, and the hand with the disorder was placed on a surface. After rubbing the gel, a conductive medium to guide the waves, the probe of the device (Duolith® SD1, STORZ, Switzerland) was placed on the spot to transmit the waves to the site. There were three therapeutic sessions with a week interval for each patient. The treatment protocol utilized in the intervention group was 1000 impulses with 2 bar pressure at a frequency of 15 Hz on the radial head and focused on the tissue surrounding the maximal pain point. The treatment regimens in the sham group were similar to those of the intervention group with a turned-off device without induction of shock waves (sham shock wave). In both the intervention and sham groups, conservative treatments included administration of an immobilized splint of the thumb and an effective dose of an anti-inflammatory agent, such as celecoxib (200 mg daily), for two weeks.
Before and after the treatment, the disabilities of the arm, shoulder, and hand (DASH) questionnaire, the visual analogue scale (VAS), and the hand-grip strength test were used to evaluate the patients in both the intervention and sham groups. DASH scale includes 30 items on the symptoms and function of the upper extremity involved with orthopedic and neurological disorders, scored based on a five-point Likert scale, ranging from 1 (no stiffness or no symptoms) to 5 (an inability to perform activities or the most severe symptoms). The total score ranges from 30 to 150 (25).
The VAS, scored from 0 to 10, was used to evaluate the intensity of pain in the wrist, and the hand-grip strength test was performed using a hand-grip dynamometer in all patients. The procedures were explained and demonstrated to the patients. The patient was seated comfortably on a chair with a forearm resting on the table. Then they were asked to hold the dynamometer and pull the handle, and accordingly, the highest voluntary contraction was recorded (26). The patients were followed up for two months and visited on arrival and immediately after the completion of treatment (after three weeks), as well as three and six weeks after the treatment cessation, and the patient outcomes were evaluated in these visits using the assessment tools.
The research proposal of the present study was confirmed (ID no.: 395805), and its protocol was approved by the Ethics Committee of Isfahan University of Medical Sciences (ethical code: IR.mui.rec.1395.3.805).
3.4. Statistical Analysis
The sample size was calculated based on a confidence interval of 90%, a test power of 75%, a standard deviation of 0.9, and mean differences of 0.3. The IBM SPSS software for Windows (SPSS Inc., Chicago, IL, USA, version 20) was used to analyze the data. The data were expressed as mean ± standard deviation (SD) or frequency (percentage). The Shapiro-Wilk test was used to assess the normality of data distribution. The results showed that the residuals of hand-grip strength (in all four times) and VAS (before treatment) did not have a normal distribution (P < 0.05). However, the residuals of DASH (in all four times) and VAS (after the completion of treatment, as well as three and six weeks after the cessation of treatment) had a normal distribution according to the results of the Shapiro-Wilk test (P > 0.05). The independent samples t-test was also used for intragroup comparisons, and according to the results, their residuals had a normal distribution. The Mann-Whitney test was also performed for data with non-normal distribution residuals. Moreover, the repeated-measures ANOVA was used to compare pre- and post-VAS and DASH measures and the Friedman test to compare hand-grip strength data. Also, the chi-Squared test was employed for intergroup comparison of qualitative data. Changes in the variables were compared by the repeated measures ANOVA. A P-value of < 0.05 was considered as the level of significance.
4. Results
In the current study, 26 patients were divided into the intervention (six males and seven females with a mean age of 44.61 ± 11.36 years) and sham (four males and nine females with a mean age of 48.23 ± 14.45 years) groups. There were no significant differences between the groups in terms of age, gender, involved side, and history of the disease (P > 0.05). All the patients completed the study (Table 1). Changes in VAS scores were significant at different intervals. The VAS score in the intervention group significantly reduced after the intervention (P < 0.05), but the changes were insignificant in the sham group (P > 0.05). The means of VAS score were significantly lower in the intervention group after treatment as well as three and six weeks after treatment cessation than those of the sham group (P < 0.05). However, there was no significant difference between the groups in terms of VAS score before treatment (P > 0.05). DASH score significantly decreased in both groups six weeks after treatment cessation, and the means of DASH score were significantly lower in the intervention group than sham after treatment completion as well as three and six weeks after the cessation of treatment (P < 0.05). However, the difference was insignificant before the treatment onset (P > 0.05). The hand-grip strength in both the intervention and sham groups increased significantly six weeks after the treatment cessation (P < 0.05); however, the difference was insignificant at all intervals (P > 0.05) (Tables 2 and 3).
| Variable | Before Treatment | After Treatment | 3 Week After the Treatment Cessation | 6 Week After the Treatment Cessation | P Times | P Times Group | P Group |
|---|---|---|---|---|---|---|---|
| VAS | < 0.001 | < 0.001 | |||||
| Intervention | 7.83 ± 1.46 | 5.41 ± 1.78 | 4.91 ± 1.37 | 4.25 ± 1.13 | < 0.001 | ||
| Sham | 8.75 ± 0.96 | 8.50 ± 1.01 | 8.16 ± 1.02 | 7.75 ± 1.14 | 0.24 | ||
| Mean difference | -0.92 | -3.09 | -3.25 | -3.50 | |||
| P-value | 0.11 | < 0.001 | < 0.001 | < 0.001 | |||
| DASH | < 0.001 | 0.01 | |||||
| Intervention | 75.66 ± 15.15 | 65.75 ± 9.91 | 60.16 ± 10.54 | 56.58 ± 9.56 | < 0.001 | ||
| Sham | 81.83 ± 10.81 | 76.08 ± 10.71 | 73.33 ± 010.79 | 74.91 ± 13.03 | < 0.001 | ||
| Mean difference | -6.17 | -10.33 | -13.17 | -18.33 | |||
| P-value | 0.64 | 0.04 | 0.02 | 0.002 | |||
| Hand-grip strength | < 0.001 | 0.84 | |||||
| Intervention | 13.23 ± 6.58 | 14.29 ± 6.61 | 15.75 ± 7.010 | 16.41 ± 7.05 | 0.01 | ||
| Sham | 13.75 ± 7.36 | 14.08 ± 7.42 | 14.58 ± 7.82 | 15.01 ± 8.15 | 0.004 | ||
| Mean difference | -0.52 | 0.21 | 1.17 | 1.40 | |||
| P-value | 0.65 | 0.70 | 0.65 | 0.40 |
Assessment Tools in the Groups at Different Intervalsa
| Variable | Before Treatment-After the Treatment Completion | Before Treatment-3 Week After the Treatment Cessation | Before Treatment- 6 Week After the Treatment Cessation |
|---|---|---|---|
| VAS | |||
| Intervention | 2.30 ± 1.60 | 2.84 ± 1.40 | 3.58 ± 1.31 |
| Sham | 0.30 ± 0.75 | 0.61 ± 0.76 | 1.00 ± 0.85 |
| DASH | |||
| Intervention | 10.61 ± 9.11 | 16.30 ± 8.28 | 19.08 ± 8.71 |
| Sham | 6.07 ± 3.86 | 9.00 ± 5.44 | 6.91 ± 11.22 |
| Hand-grip strength | |||
| Intervention | -1.13 ± 1.20 | -2.55 ± 1.34 | -3.18 ± 1.32 |
| Sham | -0.30 ± 0.48 | -0.76 ± 0.72 | -1.25 ± 1.05 |
Mean Differences of VAS, DASH, and Grip Strength at Different Intervalsa
5. Discussion
The present study aimed at evaluating the effects of shockwave therapy on de Quervain tenosynovitis, and according to the obtained results, VAS score significantly decreased in the intervention group after the treatment completion, as well as three and six weeks after the treatment cessation, whereas no improvement was observed in the sham group at different intervals. These findings revealed that pain may persist with conservative treatment, and extracorporeal shockwave therapy could reduce it in patients with de Quervain tenosynovitis. Although some models are proposed for the mechanism of pain relief induced by shockwave therapy, the information about this subject is not very detailed (27-30). Extracorporeal shockwave therapy is applied to many musculoskeletal disorders since its benefits are proved based on the results of studies (31). Large areas and deep tissue can be targeted by the shockwave, and it does not depend on the reports of any imaging-based guidance system to determine the target area, which makes its utilization easier (31-34). Since it does not stimulate pain in the target area, and its effects might be blocked by local anesthetics, it is applied without local anesthetics (31, 35, 36). It is believed that chronic pain caused by many conditions, such as de Quervin disease, is due to synaptic threshold modification creating memory reflex, and shockwave therapy can remove these memories (27). Therefore, pain relief may happen as the advantage of extracorporeal shockwave therapy, as demonstrated in the present study. DASH scores significantly reduced after the completion of treatment in both groups, but the differences were more significant in the intervention group. The obtained results demonstrated that conservative treatment could effectively improve the function of the upper limb in de Quervain tenosynovitis over time, but adding extracorporeal shockwave treatment significantly improved the outcomes since the scores significantly increased compared to the sham group just after the treatment completion and in follow-ups. In the present study, the results of the hand-grip strength test revealed a significant improvement after the completion of treatment with shockwave therapy. Likewise, conservative treatment was effective, and there were no significant differences between the groups, although shockwave therapy resulted in better outcomes. These results mean that although extracorporeal shockwave therapy improved hand-grip strength, almost the same results were obtained by conservative therapy, and deciding on the treatment of choice should rely on patient satisfaction with treatment and the cost-benefit ratio. Elerain (24) conducted a similar study to compare the outcomes of radial shockwave therapy and physiotherapy on 32 patients with de Quervain tenosynovitis assigned to two groups. One group received an ultrasound at a frequency of 1 Hz and the intensity of 1.2 W/cm2, and the other group underwent radial shockwave therapy at a frequency of 5 Hz and pressure of 1/5 MPa (15 bar). Findings of his study indicated that general pain decreased and pain during thumb and wrist movement reduced in both the groups. Also, duration and periodicity of pain decreased in both groups. These findings were not significantly different between the groups; however, the author suggested that shockwave therapy might lead to faster pain relief and complementary splinting, a key element in the efficacy of this type of treatment. Some of the differences between the study by Elerain (24) and the present one were the frequency, impulses, and intensity used for the shockwave group. Another difference was that three therapeutic sessions were used in the present study with a week interval for follow-up in both groups, whereas 10 sessions were held in his research for the physiotherapy group (two sessions per week) and three sessions for the shockwave group (without intervals). The control groups were also different in two studies; the present research immobilized the thumb with a splint and administered celecoxib whereas, in the study by Elerian (24), subjects underwent ultrasound therapy and active exercises for hand muscles. These differences might lead to different results, although it is agreed that shockwave therapy is efficient, especially in a shorter time. Similar results are reported in the studies using shockwave therapy to treat tenosynovitis and tendinopathies (37-39). The scarcity of complications, safety, effective pain relief, and rapid outcomes are the most notable benefits of this method, which were in line with the findings of the present study.
5.1. Limitations
The main limitation of the study was the small sample size due to the low prevalence of the disease between males and females. It is recommended to perform studies with larger sample sizes to compare different treatment methods for de Quervain tenosynovitis and evaluate multidimensional aspects of this disorder in terms of pain, upper limb function, and follow-up outcomes. Second, the effects of arm splint and anti-inflammatory drugs were not compared directly and separately from the shockwave therapy as the conservative treatment applied to both groups. Third, general pain was evaluated through VAS, and other detailed aspects of pain, such as duration and pain caused by different movements of the wrist, were not investigated. Fourth, the outcomes of other treatments, such as corticosteroid injection, physiotherapy, and surgical interventions, were not compared with those of shockwave therapy in the present study, although controlled clinical trials compared their outcomes with those of sham groups (10, 14, 40-42).
5.2. Conclusions
Extracorporeal shockwave therapy is a safe and easy method to reduce pain, enhance upper extremity functions, and strengthen hand-grip in patients with de Quervain tenosynovitis if accompanied by conservative therapies, such as thumb splint, and the outcomes may be achieved in a shorter time than other treatments used alone. Pain relief is more significantly achieved in this technique, as it may not be effectively observed in other methods alone. However, due to the small sample size, conclusive results cannot be obtained.