1. Background
Delirium is commonly observed in the elderly in acute and non-acute care settings and is associated with poor outcomes (1-3). Prevalence estimates typically range from 9 - 32% of older hospitalized inpatients (4). According to studies, around 70% of patients in intensive care units (ICU) are affected by delirium (5) and the hospital mortality rate is 25 - 33% (6). The one-year mortality rate of 35 - 40% is observed for hospitalized seniors with delirium (7), which is similar to the mortality linked to sepsis or acute myocardial infarction (8). The delirium rate in Iran is 21.8%, while its rate in hospitalized cases in ICU and general ward is 24.7% and 17.5%, respectively (9).
Investigating the outcomes after delirium in the elderly and providing interventions to improve patient outcomes are of particular interest (10, 11). Although it is an acute disease, it is linked to many long-term complications as well as an increase in healthcare burden and costs (12). Delirium is linked with persistent impairments in brain function, including cognitive decline and increased risk of dementia and death (13, 14). Although numerous studies have documented that persistent cognitive impairment affects 30 - 80% of survivors (15-20), it is necessary to determine the risk factors and predictors of this pernicious complication of critical illness (21).
Despite a high rate of delirium in cases admitted to hospitals in Iran (9), little has been investigated about the future cognitive dysfunction after delirium and related risk factors. Identifying manageable risk factors regarding the cognitive deterioration in delirium is needed for developing preventive approaches to reduce morbidity (22). Such a decrease in the rate of patients with cognitive dysfunction decreases the possibility of premature nursing home placement as well as the cost of caring for the elderly and increases one’s quality of life (QoL) (23).
2. Objectives
This study aimed to follow a cohort of patients with delirium at the time of hospital admission over one year for mortality. We reported an association between a delirium episode during hospitalization and the prevalence of cognitive dysfunction among cases who survived after the initial hospital admission.
3. Methods
The three-month mortality and related risk factors, as well as clinical characteristics of patients were described in our previous study (24). The present study followed these patients for one year. Briefly, 100 delirious participants (79.9 ± 15.1 years, 24% female) were selected from the general hospital from January to October 2018. Twelve patients died in hospital and cumulative three-month mortality was 40%. Finally, 60 patients were followed for one year. Inclusion criteria were patients participating in the previous study (three-month cohort) and signing a consent letter. Exclusion criteria were sedation or unconsciousness of patients. The mini-mental state examination (MMSE) was performed in all individuals. We followed up the patients’ cognitive dysfunction and death, that were the primary outcomes. Follow-up phone calls were conducted from 23 October 2018 to 21 November 2019 to record the death dates. We interviewed living patients to evaluate cognition function for 12 months after enrollment. Four patients missed the follow-up and were deleted from the study. This research was conducted based on the Helsinki Declaration guidelines and approved by the local Ethics Committee of Mazandaran University of Medical Sciences, Iran (ethics code: IR.MAZUMS.IMAMHOSPITAL.REC.1399.049). Informed consent was obtained from all patients or their relatives.
Cognitive impairment was measured using the MMSE as a 20-item scale to assess cognitive impairment more generally. MMSE cannot discriminate between delirium and dementia. It is scored from 0 - 30 and evaluates seven cognitive domains of (1) orientation to time (0 - 5); (2) orientation to place (0 - 5); (3) three-word registration (0 - 3); (4) three-word delayed recall (0 - 3); (5) working memory (0 - 5); (6) language: The comprehension of a three-step command, naming, repeating, and writing (0 - 8); and (7) visuoconstruction: Copy of intersecting pentagons (0 – 1) (25). Scores 25 - 30 indicate normal cognition; 21 - 24: Mild dementia; 10 - 20: Moderate dementia; and ≤ 9: Severe dementia.
The analyses were conducted with SPSS software version 22. We analyzed the data using t-test, chi-square, and Fisher's exact test. Also, the number of survival days according to delirium diagnosis was assessed by Kaplan-Meier curves with the log-rank test. Cox semi-parametric regression was applied for evaluating the impact of underlying variables on the occurrence of outcomes. Using the log-rank test, effective factors were identified and significant variables along with variables with P < 0.3 were entered into the model. Then, using the backward elimination, we determined the remaining variables in the semi-parametric Cox model. P < 0.05 was considered significant.
4. Results
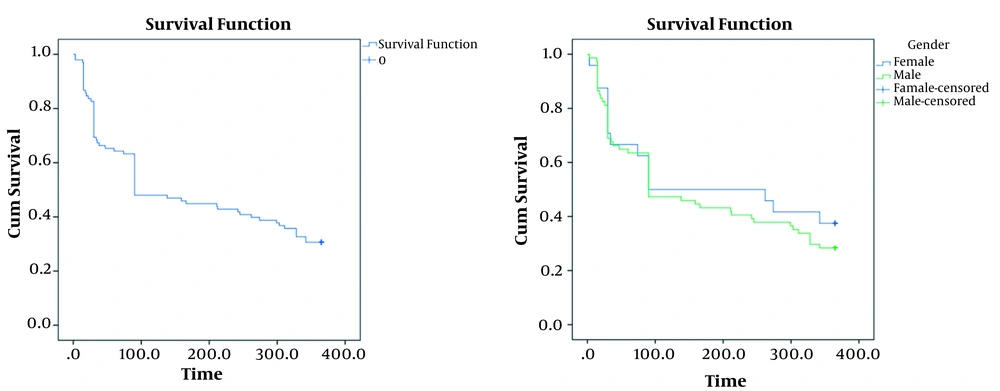
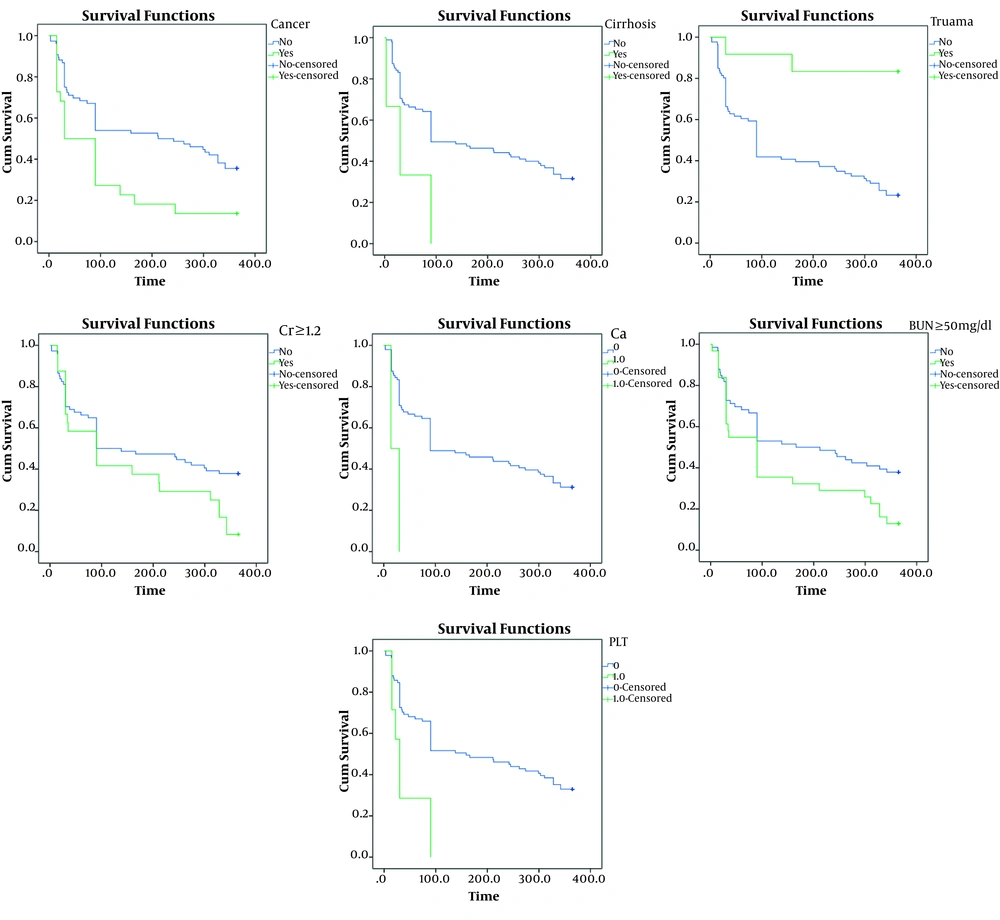
During the 12-month follow-up, 30 of the 100 individuals remained alive. Survival rate is illustrated in Figure 1. The possibility of one-year mortality using the Kaplan-Meyer method was 31%. The mean survival time of patients was 180.8 ± 15.14 days with a confidence interval of 151.16 - 210.53 and also the median survival of patients was 90.3 ± 25 days with a confidence interval of 40.45 - 139.5 (Figure 1A). The survival rate of females was longer compared with males; however, this difference was not significant (P = 0.48, Figure 1B). Kaplan-Meier survival curves regarding effective factors associated with mortality rates are presented in Figure 2. There was a significant association in the one-year mortality rate and underlying factors, such as cancer (P = 0.008), cirrhosis (P = 0.04), trauma (P = 0.001), BUN ≥ 50 mg/dL (P = 0.02), Cr ≥ 1.2 mg/dL (P = 0.04), hypocalcemia (P = 0.005), and thrombocytopenia (P = 0.002). Table 1 indicates the semi-parametric Cox regression model results for the survival rate. According to this model, the hazard ratio in patients with hypocalcemia was 5.62 times higher than other patients. Moreover, patients with thrombocytopenia and trauma had a hazard ratio of 2.84 and 0.14, respectively.
| Variables | HR | SE | Chi-Square | Significant Level | 95% CI | |
|---|---|---|---|---|---|---|
| Lower Limit | Upper Limit | |||||
| Ca | ||||||
| No | - | - | - | - | - | - |
| Yes | 5.62 | 4.14 | 2.34 | 0.02* | 1.32 | 2.38 |
| PLT | ||||||
| No | - | - | - | - | - | - |
| Yes | 2.84 | 1.17 | 2.54 | 0.01* | 1.27 | 6.37 |
| Trauma | ||||||
| No | - | - | - | - | - | - |
| Yes | 0.14 | 0.1 | -2.67 | 0.008* | 0.03 | 0.59 |
Abbreviations: Ca, calcium; CI, confidence interval; HR, hazard ratio; PLT, platelets; SE, standard error.
a Significant (P < 0.05).
According to the MMSE scores, 14 (46.7%) participants had normal cognition, 2 (6.7%) had mild dementia, 10 (33.3%) had moderate dementia, and 1 (3.3%) had severe dementia. Table 2 shows the mean (± SD) scores of MMSE domains. The total MMSE score in our patients was 23.55 (± 5.34), with a score of 30 obtained by 4 (13.3%) individuals. Of those scoring less than 30, the recall, working memory, and visuoconstruction domains were the commonest to contain an error (100%) followed by the language (92.3%), two-orientation (30.8%), and registration (7.7%) domains. Table 3 indicates the results of univariate analysis for MMSE score. Individuals who developed cognitive impairment were older (P < 0.001). The overall MMSE scores and sub-scores showed no difference between male and female patients (P = 0.58). There was a significant association between cognitive impairment rate and underlying factors, such as pulmonary infections (P = 0.02), trauma (P = 0.02), and renal failure (P = 0.04).
| Domain | Score |
|---|---|
| Orientation | 9.55 ± 1.28 |
| Registration | 2.93 ± 0.38 |
| Recall | 2.92 ± 1.46 |
| Working memory | 1.85 ± 0.81 |
| Visuoconstruction | 0.22 ± 0.42 |
| Language | 6.11 ± 2.06 |
| Total | 23.55 ± 5.34 |
a Values are expressed as mean ± SD.
| Variables | MMSE < 24 | MMSE ≥ 25 | P-Value |
|---|---|---|---|
| Age | 81.6 ± 9.7 | 52.7 ± 17.7 | < 0.001* |
| Sex | 0.58 | ||
| Female | 5 (55.6) | 4 (44.4) | |
| Male | 10 (55.6) | 8 (44.4) | |
| Underlying Disorders | |||
| Pulmonary infections | |||
| Yes | 6 (85.7) | 1 (14.3) | 0.02* |
| No | 7 (35) | 13 (65) | |
| Trauma | |||
| Yes | 1 (12.5) | 7 (87.5) | 0.02* |
| No | 12 (63.2) | 7 (36.8) | |
| Renal failure | |||
| Yes | 0 | 4 (100) | 0.04* |
| No | 13 (56.5) | 10 (43.5) | |
Abbreviation: MMSE, mini mental state exam.
a Significant (P < 0.05).
5. Discussion
In the present prospective research, the association between delirium and the one-year survival and cognitive dysfunction was assessed. The major findings are as follows: (1) the possibility of one-year survival was 31%; (2) the one-year mortality rate showed a significant association with underlying factors, such as cancer, cirrhosis, trauma, hypocalcemia, thrombocytopenia, and renal disease; (3) the probability of cognitive dysfunction was 53.3%; (4) the frequency of cognitive impairment showed a significant association with underlying factors, like pulmonary infections, trauma, and renal failure. Our findings are crucial for older hospitalized cases.
The incidence of cognitive dysfunction due to delirium was 53.3%, which is consistent with other studies (26, 27). However, the delirium effect on survival after discharge is controversial. Our results are consistent with the findings of studies that reported the mortality risk for delirious cases following one-year hospitalization (7, 28). In contrast, Wolters et al. reported no significant association between delirium and one-year survival (29). According to Leslie et al., the risk of mortality in delirium cases (at least 70 years old) in the hospital elevated by 62% after 12 months of discharge (30). However, in our results, there was no significant association between age and one-year survival after delirium. One explanation for this inconsistency can be the heterogeneity of patients referring to the general hospital. Therefore, younger patients (17 participants aged below 55) with serious clinical conditions and delirium were included in our study. In line with our results, Mitchell et al. showed that delirium in hospital had an association with all-cause one-year mortality following hip fracture in older cases with no dementia (31). Also, similar to previous studies, the other underlying factors can affect mortality rate including cancer (32), hepatic (33), renal (34), and hematologic diseases (35, 36).
Our findings are in line with the existing literature, which showed that substantial cognitive under performance is a possible consequence of delirium (13, 37, 38). In line with previous studies, our findings showed the association between cognitive impairment rate and underlying factors such as trauma (39), pulmonary (40, 41), and renal diseases (42). This relationship has several possible explanations and mechanisms; that delirium can be associated with brain damage which may be irreversible. There is good evidence supporting the relationship between delirium and neuroinflammation and neuronal apoptosis, cerebral atrophy, and decreased white-matter integrity (43-45). Another possibility is that the other changes in delirium can cause or exacerbate brain damage through malnourishment (46), dehydration (47), trauma (48), and psychological stress (49, 50). Our results support those reported by McCusker et al. indicating a relationship between delirium and lower scores in the MMSE 12 months after hospitalization (51). Our findings also strongly support an association between delirium and dementia (52, 53). The long-term cognitive deficit has recently been shown after delirium in older adults, that is possibly distinct from classical dementia signatures (54).
Our study had some limitations. First, we were unable to identify a relation between underlying factors and specific domains of MMSE due to the small sample size. Future researchers are suggested to conduct studies with larger sample sizes. Second, we did not follow the patients in a shorter period of time to evaluate patients who died about cognitive impairment symptoms after hospital discharge, in more checkpoints. Our findings add to other studies because of assessing the relationship between delirium and cognitive impairment in non-surgical cases outside the critical care units, with normal cognitive status in the baseline that were admitted in the general hospital. Our results are crucial for clinical practice to identify the underlying factors linked to death and cognitive dysfunction in hospitalized delirious elderly people in an Iranian population. We provided appropriate data for a preventive strategy and clinical management regarding cases who experienced delirium in the general hospital, allowing a reduction in post-discharge cognitive impairments and mortality.
5.1. Conclusions
In conclusion, delirium should be regarded as an important and serious problem because of the possibility of mortality risk. Assessment and screening for delirium are necessary for all older hospitalized inpatients. A brief cognitive assessment is effective to identify delirium, enhance proper management, and reduce its associated complications. Family members should be trained and involved in care, particularly for monitoring of risk factors upon discharge.