1. Background
Cesarean section (CS) is the most common abdominal surgery in women around the world and can be associated with morbidity and mortality (1). Recently, the growing rate of CS has increased its complications, including CS scars as an associated potential morbidity (2) and the development of a thin residual myometrium or niches at prior CS sites (3, 4). The presence of a niche and thin residual myometrium thickness (RMT) has been associated with obstetric complications (4), such as scar rupture or dehiscence, adherent placenta (4), ectopic pregnancy at the cesarean section scar, abnormal uterine bleeding during non-pregnancy time, dysmenorrhea (5), postmenstrual spotting (5, 6), pelvic pain, infertility, and other gynecologic complications (7, 8). Accordingly, some have suggested that the laparoscopic repair of CS scar can reduce further complications (9, 10); however, more studies are required in this regard.
For a better understanding of this complication, studies have investigated the risk factors of cesarean scar defect (CSD) and the role of maternal factors, such as the mother’s obesity, gestational diabetes mellitus, and the number of C/S deliveries, as well as advanced labor during emergency C/S (11, 12). Meanwhile, from a pathophysiologic perspective, C/S wound healing is a complex biochemical cascade that involves several peptides and growth factors, such as platelet–derived growth factor (PDGF), transforming growth factor beta (TGF–β) isoforms, tumor necrosis factor alpha (TNF–α), vascular endothelial growth factor (VEGF), so any factor that can hinder one or more of the four stages of surgical site repair, including hemostasis, inflammation, proliferation, and remodeling, can also predispose women to CSD (13).
Platelet–rich plasma (PRP), an activated part of plasma rich in platelets and growth factors, has been found effective in the wound healing of surgical scars and improving scar repair quality (14). Accordingly, we hypothesized that PRP could also help wound healing in CSD. Therefore, and because there is insufficient evidence about the effectiveness of PRP in improving the thickness and completeness of the uterine scar, the present study was designed and performed to address this issue.
2. Methods
2.1. Study Design
A randomized double-blinded, placebo-controlled clinical trial was performed at the Hazrat-e Rasool Hospital of Tehran (affiliated with Iran University of Medical Sciences) from November 2019 to November 2020. The purpose of the study was to assess the effects of PRP on C/S scar repair after surgery. In accordance with Hertzog’s recommendations (15), the sample size was calculated as at least 15 women in each group, and 10 additional women were added to each group as compensation for a possible loss of samples during follow up, so the number of subjects per group was considered n = 15.
All singleton pregnant women who were candidates for their first CS were initially assessed for inclusion criteria, including: (1) undergoing elective CS (breech, orthopedic problems, retinal detachment, macrosomia, low-lying placenta, preeclampsia, perineal repair due to a severe accident) with cervical dilatation less than 5 cm and effacement less than 30%, (2) term pregnancy, (3) the rupture of membranes earlier than 24 hours, (4) lack of chorioamnionitis criteria, including fever and tenderness of the uterus, (5) no use of any anti-inflammatory or anticoagulant drugs for 3 - 4 days prior to the surgery, (6) and no prior surgeries on the uterus. Women with uterine anomalies, any malignancy, those with myoma at the incision site, placental previa or acreta, thrombocytopenia (platelet count < 150 × 103 per μL), women with a family history of platelet disorders, and finally individuals who were not interested to participate in this research were excluded from the study.
This study was designed as a randomized double-blinded, placebo-controlled trial. The investigators (one radiologist, an assistant who was not a member of the surgical team) were blinded to group allocation. Women (n = 30) were randomly assigned to one of the two groups (intervention and control) using a computer-generated random table and a block size of 2. Coded envelopes using sequential numbers were prepared by a statistician who was not involved in the research project, and the surgeon allocated the women to one of the two groups based on the envelopes.
Before enrollment into the study, the researcher explained the study’s protocol and objectives to the participants and requested them to sign a written informed consent form. All the women underwent CS by an experienced team of gynecological surgeons, and all of them received 2 gr preoperative intravenous cephalothin. All surgeries were performed by the same team of surgeons. Pfannenstiel incision was made in the lower segment of the uterus. After the birth of the fetus and the spontaneous removal of the placenta, uterine incision was closed by the conventional technique, including two-layer closure of the uterus using continuous running sutures for both layers and the 0 chromic suture of the endometrial layer, which was similarly performed in both groups. After closing the uterine incision, the participants in the intervention group were injected with PRP (both the upper and lower sides of the hysterotomy between decidua and myometrium). For preparing PRP, an 8.5 mL venous blood sample was collected into tubes with 1.5 mL anticoagulant (a total volume of 10 mL). The samples were centrifuged for 10 minutes at 1600 rpm. The supernatant (the plasma layer) was removed; the lower part was transferred to another tube (without anticoagulant) and centrifuged at 3500 rpm for six minutes. The result of these two steps of centrifugation was about 2 - 3 mL PRP, seen as a white halo at the bottom of the tube. In the control group, the incision site was treated by routine care without PRP injection. Finally, the fascia, subcutaneous tissue, and skin were repaired following the same manner in the two groups.
2.2. Outcomes, Measurements, and Follow-up
General parameters including age, gravidity, parity, gestational age, weight, height, body mass index (BMI), birth weights of neonates, and indications for CS were asked from the women or extracted from their medical records.
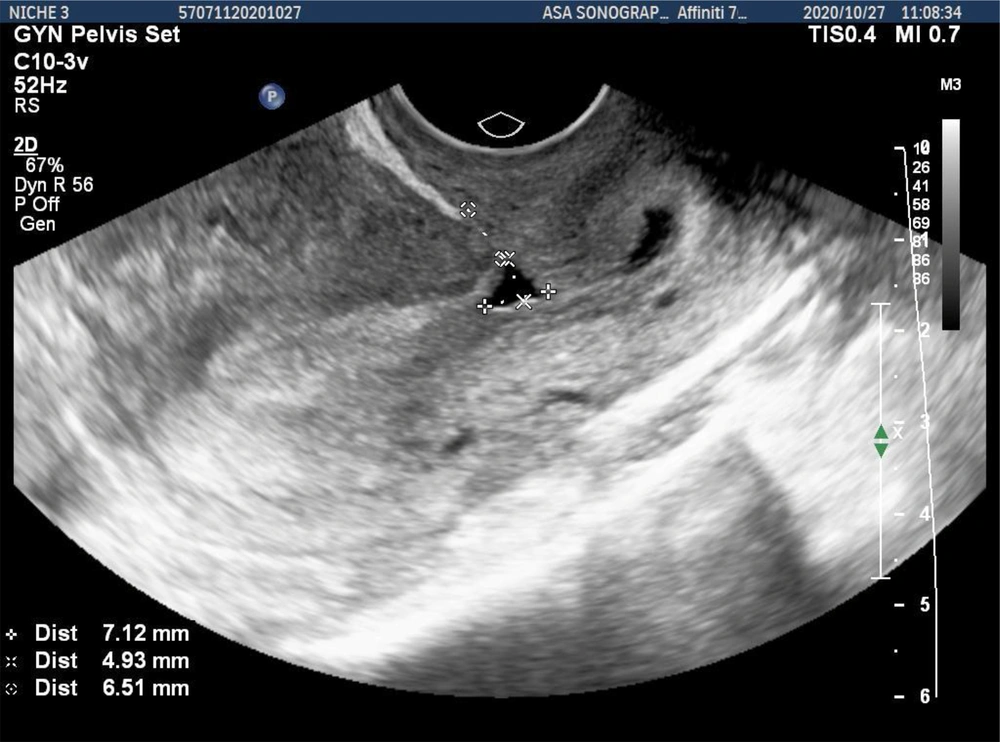
The primary outcome of the study was scar thickness, including the frequency of myometrium defects at the site of the uterine scar (niche), niche height, residual myometrium thickness (RMT), and adjacent myometrium thickness (AMT). A niche was defined as the hypoechoic part at the site of CS scar with a depth of ≥ 2 mm, which was visible on transvaginal sonography (TVS) (16) (Figure 1).
Blood loss, fever, and infections were regarded as secondary outcomes. For estimating blood loss, hemoglobin (HB) levels were measured using 2 mL blood samples before and 24 hours after surgery. Blood samples were collected into K2 & K3‐EDTA containing vacuum tubes and sent to the laboratory of the hospital. Fever was defined as an oral body temperature of > 37.5°C. The body temperature was measured at 6-, 12-, and 24-hour post-surgery. Re-admission of the women due to infection was also recorded.
All the women were followed for 12 weeks, and TVS was performed in the radiology department using Mindray DC7 V10-4 at the frequency of 7 - 9 MHz at the end of the follow-up period. All ultrasound examinations were done by the same radiologist to avoid bias, and the radiologist was not informed of group allocations.
2.3. Statistical Analysis
The data was computerized and analyzed using the statistical package of social sciences (SPSS) version 22.0 software (SPSS Inc., Chicago, IL., USA). Descriptive statistics (mean ± standard deviation) and frequency (percent)) were used to present the data. The χ2 test and Fisher’s exact test were used to assess associations between qualitative and categorical data. The one–sample Kolmogorov–Smirnov test was used to determine the normal distribution of the data, and Levene’s test was used to investigate the equality of variances. Variables with normal distribution were compared between the two groups using the independent samples student t-test. For comparing the means of two non-normally distributed variables (i.e., the number of pregnancies and deliveries), the Mann–Whitney U test was used. P < 0.05 was considered statistically significant.
3. Results
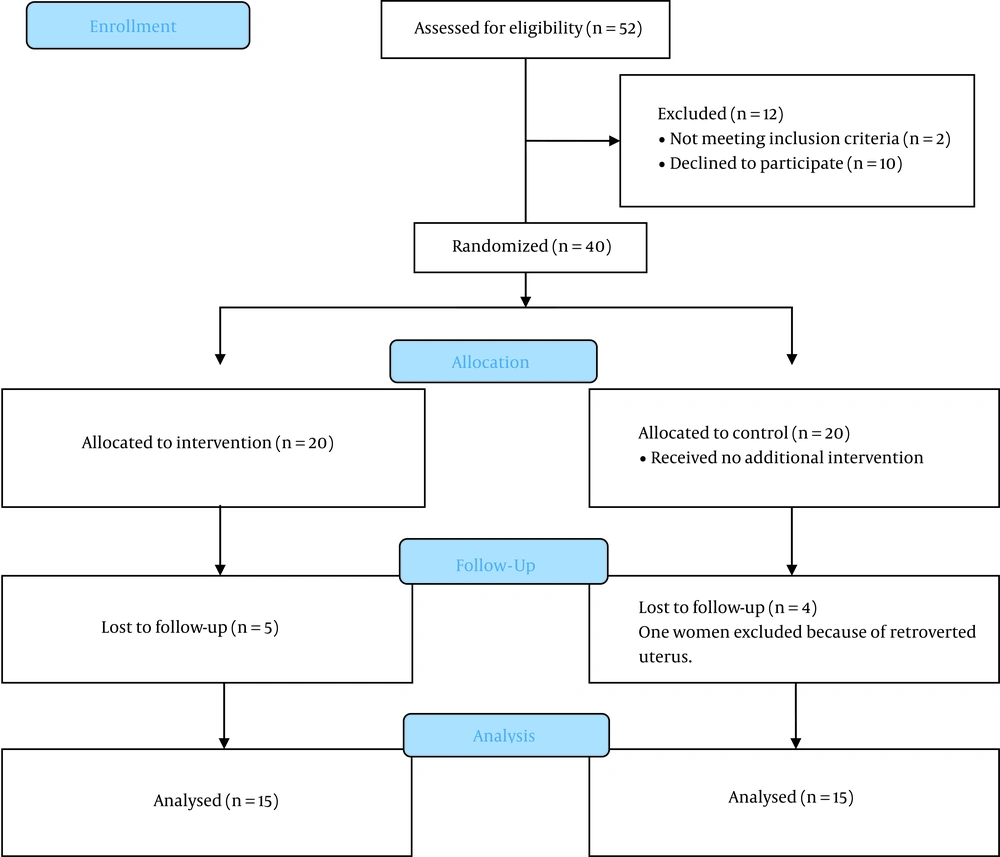
Totally, 52 women were assessed for eligibility, and 40 of whom were included in the study (20 women in each group). Of all eligible cases, five cases were excluded from each group due to lack of cooperation and having a severe retroverted uterus. Ultimately, the analysis was performed with 15 cases in each of the intervention and control groups (Figure 2).
The mean age of the study population was 27.79 ± 6.8 years, and 79 % of them were primigravidas. At the baseline, the two groups revealed no significant differences in terms of age, weight, height, gravidity, parity, BMI, and neonates’ birth weights (Table 1). In general, the most common indication for CS was fetal distress (33.3%), followed by severe preeclampsia (30%) and breech presentation (16.7%). The general parameters of the two intervention and control groups have been presented in Table 1.
| PRP Group (n = 15) | Control Group (n = 15) | P Value | |
|---|---|---|---|
| Maternal age, y | 0.63 a | ||
| Min - Max | 21 - 39 | 19 - 37 | |
| Mean ± SD | 27.2 ± 5.3 | 28.39 ± 5.01 | |
| Gravidity | 0.48 b | ||
| Min - Max | 1 - 3 | 1 - 2 | |
| Mean ± SD | 1.46 ± 1.06 | 1.70 ± 1.14 | |
| Parity | 0.79 b | ||
| Min - Max | 0 - 2 | 0 - 1 | |
| Mean ± SD | 0.4 ± 0.2 | 0.46 ± 0.67 | |
| Gestational age, w | 0.54 a | ||
| Min - Max | 35 - 41 | 29 - 41 | |
| Mean ± SD | 38.6 ± 0.38 | 37.46 ± 2.91 | |
| Weight, kg | 0.47 a | ||
| Min - Max | 52 - 110 | 58 - 95 | |
| Mean ± SD | 75.8 ± 14.45 | 78.51 ± 11.9 | |
| Height, cm | 0.8 a | ||
| Min - Max | 150 - 173 | 145 - 189 | |
| Mean ± SD | 161 ± 6.1 | 162.82 ± 7.56 | |
| BMI, kg/m2 | 0.5 a | ||
| Min - Max | 19.13 - 40.90 | 18.76 - 35.76 | |
| Mean ± SD | 29.78 ± 4.8 | 29.1 ± 3.65 | |
| Birth weight of neonates, g | 0.41 a | ||
| Min - Max | 2430 - 4460 | 2450 - 4390 | |
| Mean ± SD | 3319 ± 269 | 3186 ± 405 | |
| Indications for primary C/S; No. (%) | 0.9 b | ||
| Breech | 3 (20) | 2 (13.3) | |
| Orthopedic problems | 3 (20) | 1 (6.7) | |
| Preeclampsia | 3 (20) | 6 (40) | |
| Retinal detachment | 0 | 1 (6.7) | |
| Macrosomia | 3 (20) | 4 (26.7) | |
| Low-lying placenta | 1 (6.7) | 1 (6.7) | |
| Perineal repair due to trauma | 1 (6.7) | 0 |
Abbreviations: y, year; w, week; SD, standard deviation; BMI, body mass index; kg, kilogram; cm, centimeter; g, gram; C/S, cesarean section.
astudent’s t test.
b Mann-Whitney U test.
The measurement of myometrium scar parameters in both groups was performed at 12 weeks after the surgery, and a totally, nine of 30 (30%) women were observed with uterine niches. Out of the total cases with niches, 13.3 % (2 of 15) and 46.7% (7 of 15) were in the PRP-treated and control groups, respectively, showing a statistically significant difference (P = 0.002).
Comparing the two groups, AMT showed no statistically significant difference. The mean uterine niche height in the intervention group was significantly lower compared to the control group (P = 0.048). According to the results presented in Table 2, the means of RMT in the intervention and control groups were 7.62 ± 0.95 and 5.6 ± 3.5 mm, respectively. This difference was statistically significant (P < 0.001). Myometrium scar parameters in the intervention and control groups have been presented in Table 2.
| Variables | PRP Group (n = 15) | Control Group (n = 15) | P Value a |
|---|---|---|---|
| No. of women with uterine incision defect; No. (%) | 2 (13.3) | 7 (46.7) | 0.002 |
| Niche height b | 0.048 | ||
| Min - Max | 1.8 - 2.8 | 4.6 - 6.2 | |
| Mean ± SD | 2.1 ± 0.5 | 5.5 ± 0.8 | |
| RMT | < 0.001 | ||
| Min - Max | 5.6 - 9.3 | 1.1 - 10.88 | |
| Mean ± SD | 7.62 ± 0.95 | 5.6 ± 3.5 | |
| AMT | 0.84 | ||
| Min - Max | 6.6 - 9.4 | 7.1 - 9.3 | |
| Mean ± SD | 8.3 ± 0.78 | 8.5 ± 0.84 |
Abbreviations: SD, standard deviation; RMT, residual myometrium thickness; AMT, adjust myometrium thickness.
a Mann–Whitney U test.
bMeasured in the sagittal plane.
Mean preoperative hemoglobin levels in the intervention and control groups were 12.01 ± 1.3 and 11.98 ± 1.4 g/dL, respectively. The highest postoperative HB drop seen in the intervention and control groups was not statistically significant (2.5 vs. 2.9 g/dL, retrospectively). According to the results of the Mann-Whitney U test, there was no significant difference in the HB level between the intervention and control groups. The results of the secondary outcomes (blood loss and fever) have been presented in Table 3. No readmission due to infection was recorded in the study groups.
| Variables | PRP (n = 15) | Control (n = 15) | P Value |
|---|---|---|---|
| Hb level before surgery (g/L) | 12.01 ± 1.3 | 11.98 ± 1.4 | 0.06 b |
| Hb level 24 hours after surgery (g/L) | 10.9 ± 1.4 | 11.2 ± 1.6 | 0.87 b |
| Difference in Hb level (after vs. before) (g/dL) | 1.11 ± 1.6 | 0.78 ± 08 | 0.14 b |
| Body temperature after surgery (°C) c | |||
| 6 hours | 37.13 ± 0.7 | 37.02 ± 0.15 | 0.5 b |
| 12 hours | 36.43 ± 0.56 | 36.18 ± 0.91 | 0.32 b |
| 24 hours | 36.67 ± 0.76 | 36.98 ± 0.67 | 0.993 b |
| Fever d | |||
| 6 hours | 1 (6.7) | 0 | 0.2 c |
| 12 hours | 1 | 0 | 1.00 c |
| 24 hours | 0 | 0 | 1.00 c |
Abbreviation: Hb, hemoglobin.
a Continuous variables have been presented as mean ± SD, and categorical variables as No. (%).
b Mann-Whitney U test.
c Fisher's exact test.
d Temperature > 37.5°C.
4. Discussion
The world has been faced with an increasing CS rate and its subsequent scar complications. The complications related to uterine scars have encouraged researchers to investigate the behavior of CS scars and their associated potential morbidity (2). Recently, some studies have specifically addressed the development of niches or thin residual myometrium at the site of CS (3, 4). The technique of uterine closure (single- versus double-layer closure) and suture materials (Vicryl and chromic sutures) are among the crucial determinants of myometrial scar healing and complications, especially in subsequent pregnancies (3, 17). Over the years, PRP has been considered as a therapy for chronic and acute wounds and is derived from the blood of patients. The product consists of growth factors, cytokines, chemokines, and a fibrin scaffold and stimulates wound healing (18). Despite a few publications on wound healing after CS, the effects of PRP on the integrity and thickness of the uterine scar have not been investigated (19). In the present study, we evaluated the efficacy and safety of PRP on the integrity and thickness of the uterine scar. Our results showed the favorable effects of PRP injection during C/S on the integrity and thickness of scares. Statistically, a lower number of niches with smaller heights and greater residual myometrium thicknesses were reported in the intervention group compared to the control group. In the present study, the creation of a niche in the PRP-treated group was almost one-fourth of the control group, and this difference was statistically significant.
These results are consistent with the results of previous studies, suggesting PRP injection as an effective therapeutic method for improving wound healing in several gynecological surgeries (20, 21) and surgical incisions due to its ability to promote angiogenesis and initiate inflammatory reactions (14, 22). Nonetheless, as far as the authors are concerned, no study has evaluated the efficacy of PRP on CS scars to be compared with our research. Therefore, this issue calls for performing ethically well-designed randomized controlled trials (RCTs) in future.
Tehranian et al. in a RCT investigated the efficacy of subcutaneous application of PRP on 67 high-risk women and 71 control subjects (without PRP application). Being high-risk for wound healing was defined as BMI > 25 kg/m2, prior C/S, twin pregnancy, use of corticosteroids, and having diabetes mellitus and anemia (19). Examination of the CS incision site on the days 1st and 5th and eight weeks after C/S showed reductions in the Vancouver scar scale (VSS), visual analogue scale (VAS) of pain, and the redness, edema, ecchymosis, discharge, and approximation (REEDA) score in the intervention group compared to the control (19), supporting our findings considering the effectiveness of PRP in improving wound healing in CSD. In the study of Wanas et al., comparing the effectiveness of ozonated PRP and calcium chloride-activated PRP vs. the control group, it was shown that both types of PRPs improved wound healing in CSD, according to the REEDA, VAS, and VSS scores on days 1, 7, and 21 postoperative; meanwhile, ozonated PRP showed better results (23), confirming the conclusion of the present study. Also, a study by Kamel showed better REEDA and VSS scores at 1, 5, and 30 days after C/S in the diabetic women receiving subcutaneous PRP (24). Although the results of the above-mentioned studies confirmed the general conclusion of the present study, there are several differences between our study and theirs. First, we evaluated the results of CSD using ultrasound as a reliable and accurate diagnostic method for the evaluation of niches and CSD (25). However, previous studies (19, 23, 24) have used visual assessment methods. Furthermore, study populations differed, as we included the women undergoing C/S for the first time to omit the effect of prior CSD on the outcomes; on the other hand, these studies (19, 23, 24) have included high-risk women. These differences are due to various objectives of the studies. While we aimed to investigate the effectiveness of PRP on a general population of women undergoing C/S in order to assess the applicability of this method, as an effective preventive measure, in routine care, the above-mentioned studies aimed at investigating the applicability of this method in high-risk women only.
Furthermore, we compared the participants of the two groups in terms of demographic and clinical characteristics, as well as the duration of surgery to investigate whether or not these factors that have previously been mentioned as risk factors of niche development (11, 12) had different distributions between the study groups. Nevertheless, our results confirmed that the two groups were comparable in this regard. Moreover, we registered no adverse effects for PRP injection in the study population.
To the best of our knowledge, the present study was the first to evaluate the effects of PRP injection on the integrity and thickness of the uterine scar caused by CS. The randomized double-blinded design of this study was one of its strengths; randomization was done using a computer-generated table of random numbers. The researcher analyzing the primary and secondary outcomes was blinded to the course of the intervention to reduce the risk for bias. Although the present study yielded important data, its limitations are worthy of mentioning, including the small sample size and short duration of follow-up. Therefore, long-term well-designed studies are recommended to investigate the efficacy of PRP on the integrity and thickness of the uterine scar.
4.1. Conclusions
In conclusion, the results of this pilot study supported the efficiency of PRP, as a promising and safe agent, in the prevention of CSD. Therefore, PRP injection can be used as a potential preventive measure and be included in routine wound care after C/S. Future studies are needed to confirm PRP efficacy in different settings.