1. Context
Giardiasis is a global disease caused by a flagellate protozoan called Giardia. The only pathogenic species of this parasite in humans is Giardia intestinalis (also known as G. duodenalis and G. lamblia). Although this disease is often asymptomatic, the most common symptom is diarrhea. The causative agent of the disease is resistant cysts, which are excreted from the host intestine and transmitted by oral-fecal, water, and food (1, 2). Thus, identifying the parasite's reservoir is very important to prevent it.
Studies have shown different hosts and as a result different reservoir for various assemblages of G. intestinalis. Based on its protein and DNA polymorphism, eight major genetic groups or assemblages (A, B, C, D, E, F, G, and H) have been identified for this parasite (3-5). Numerous studies have been also performed to determine the sub-assemblages of this parasite around the world (6). Most of human isolates are in assemblages A and B (3, 4, 6). Because these two assemblages could also infect other mammals, the zoonotic transmission potential of G. intestinalis is epidemiologically important (3). In some parts of Iran, studies have been conducted to identify the sub-assemblages of G. intestinalis in humans. So, it is valuable to summarize these studies to determine the predominant sub-assemblages (subtypes) and epidemiological transmission pattern of this parasite in Iran. On the other hand, such studies could identify information gaps regarding the sub-assemblage of this parasite, thereby opening new avenues for further research. Hence, the present study aimed to provide an overview on the determination of genotypes of G. intestinalis and summarize the studies on this area in Iran.
2. Evidence Acquisition
2.1. Search Process
All English or Persian articles and dissertations performed on Giardia genotype in Iran (from 1980 to 2020) indexed in PubMed, Google scholar, Science Direct, Scopus, Medline, Medlib, Scientific Information Database (SID), IranMedex, IranDoc, and Magiran were collected and reviewed. The keywords were a combination of Giardia, intestinal protozoa, assemblage, genotype, G. intestinalis, G. duodenalis, G. lamblia, Giardia genotype, Iran, Islamic Republic of Iran, genotype of Giardia in Iran. Figure 1 shows the search process.
2.2. Selection criteria
The following inclusion criteria were considered for this study. Full papers and dissertations related to determining Giardia genotypes based on different genes of this parasite, genetic characters’ and human Giardia genotype in Iran published from 1980 to 2020 were included in this work. In contrast, case studies, experimental studies, and animal studies, as well as duplicate articles were excluded.
2.3. Data Extraction
Selected papers were carefully reviewed by two researchers and information, such as first author, year of publication, type of study, location of study, language, subjects, sample size, diagnosis test, assemblage or genotype detection technique, and genotype were extracted and recorded.
2.4. Statistical Analysis
In this study, meta-analysis was used to estimate the proportion of dominant genotypes of Giardia parasite in Iran (7). Confidence intervals were also calculated using the exact binomial distribution. Further, publication bias was investigated using the Egger test (8). Heterogeneity among the studies was considered by benefiting from the Cochran’s Q test with a significant level lower than 0.1 and I2 statistic greater than 60% (9). Due to the observed heterogeneity, we used random effects model to estimate the prevalence of predominant genotype of Giardia parasite in Iran.
The meta-analysis of proportions was further performed by using “Metaprop” commands on STATA software (Ver.14). The significance level was considered as 0.05 (7). Freeman-Tukey double arcsine transformation was used in the random effects model when the proportion in some studies was very large and close to 100% (7). Additionally, a Forest plot was drawn based on the random effects model for the estimated proportion of assemblages with confidence interval of 95%.
3. Results
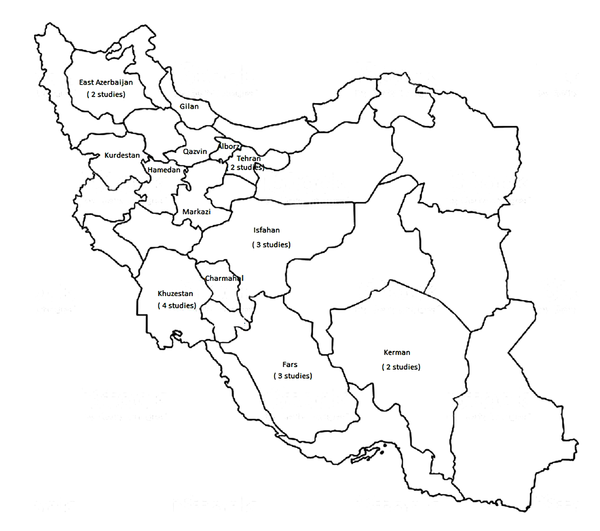
After searching the databases, 173 articles were found related to the Giardia exploration in Iran. After excluding the unrelated and duplicated studies, a total number of 23 associated articles to the sub-assemblages of human G. intestinalis in Iran were included. Figure 1 shows the process of designing and searching for articles, and Figure 2 illustrates the geographical areas of the included articles (Figures 1 & 2).
From 23 studies, one was cohort study and the rest were cross-sectional. These studies had been conducted in 13 provinces of Iran. Four studies in Khuzestan, three studies in Fars, three studies in Isfahan, two studies in East Azerbaijan, two studies in Tehran, two studies in Kerman, and one study in other provinces (7 provinces) (Figure 2). Totally, 1059 human specimens were examined in all the 23 included studies. The PCR-RFLP technique had been used for detecting of Giardia assemblages in 11 studies, while in others, the sequencing technique had been used (Table 1).
| Line | First Author | Type of Study | Place of Study (City) | Characteristic of the Sample Under Study | Number of Samples Molecularly Tested | Diagnosis Method | Determination Method Assemblage | Assemblage | Description and Conclusion | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | Mix | Neg | |||||||||
| 1 | Babaei et al. (10) | Cross sectional | Tehran | Stool samples were reported to be positive microscopically | 38 | PCR (Based on the gdh gene) | RFLP | 33 (87) | 3 (7.8) | 2 (5.2) | - | Epidemiologic transmission pattern was anthroponotic |
| 2 | Fallah et al. (11) | Cross sectional | Tabriz | Laboratory samples of Tabriz Children's Hospital laboratory and reference laboratory samples that were positive microscopic method | 34 | PCR (Based on the tim gene) | Seq | 17 (50) | 13 (38) | 1 (3) | 3 | |
| 3 | Fallah et al. (12) | Cross sectional | Tabriz | Tabriz Children's Hospital and reference laboratory samples | 34 | PCR (Based on the gdh gene) | RFLP | 6 (17.6) | 12 (35.3) | - | 16 | Epidemiologic transmission pattern was anthroponotic |
| 4 | Etemadi et al. (13) | Cross sectional | Kerman | Kerman Medical Centers | 30 | PCR (Based on the gdh gene) | RFLP | 23 (76.6) | 7 (23.4) | - | - | |
| 5 | Pestehchian et al. (14) | Cross sectional | Isfahan | Isfahan Health Centers | 67 | PCR (Based on the gdh gene) | RFLP | 40 (59.7) | 25 (37.3) | 2 (3) | - | Epidemiologic transmission pattern was anthroponotic and zoonotic. |
| 6 | Sarkari et al. (15) | Cross sectional | Fars province | Fars Province Laboratories | 205 | PCR (Based on the gdh gene) | RFLP | 128 (62.4) | 36 (17.6) | 8 (3.9) | 33 | Epidemiologic transmission pattern was anthroponotic |
| 7 | Manouchehri Naeini et al. (16) | Cross sectional | ShahreKord | Shahrekord Medical Diagnosis Laboratories | 31 | PCR (Based on the tpi gene) | Seq | 11 (35.5) | 16 (51.6) | 4 (12.9) | - | Association was seen between diarrhea and genotype A. And association was seen between asymptomatic infection and genotype B |
| 8 | Rafiei et al. (17) | Cross sectional | Ahwaz | Ahwaz Health Centers | 100 | PCR (Based on the gdh gene) | RFLP | 18 (18) | 28 (28) | 54 (54) | - | There is a association between clinical symptoms and genotype A |
| 9 | Roointan et al. (18) | Sectional (September 2011 to July 2012) | Ahwaz | Children referred to health center clinics | 50 | PCR (Based on the gdh gene) | RFLP | 5 (10) | 8 (16) | 37 (74) | - | |
| 10 | Etemadi et al. (19) | Cross sectional | Kerman | Three medical diagnostic laboratories | 30 | PCR (Based on the gdh gene) | RFLP | 23 (76.6) | 7 (23.4) | - | - | |
| 11 | Pestechian et al. (20) | Cross sectional | Isfahan province | Medical diagnostic laboratories | 67 | PCR (Based on the tpi gene) | RFLP | 40 (59.7) | 25 (37.3) | 2 (3) | - | |
| 12 | Rayani et al. (21) | Cross sectional | Shiraz | Health centers and hospitals in Shiraz | 50 | PCR (Based on the SSU-rDNA) | Seq | 32 (80) | 8 (20) | - | 10 | Epidemiologic transmission pattern was anthroponotic |
| 13 | Rayani et al. (22) | Cross sectional | Shiraz | Health centers and hospitals in Shiraz | 40 | PCR (Based on the gdh gene) | Seq | 32 (80) | 8 (20) | - | - | Epidemiologic transmission pattern was anthroponotic |
| 14 | Bahrami et al. (23) | Cross sectional | Kurdistan province | 14 Medical Diagnosis Laboratories | 23 | Nested-PCR (Based on the tpi gene) | Seq | 12 (52) | 11 (48) | - | - | Epidemiologic transmission pattern was zoonotic |
| 15 | Nasiri Goorabi et al. (24) | Cross sectional | Baharestan city | Health centers of Baharestan city in Tehran province | 25 | PCR (Based on the β-giardin gene) | RFLP | 16 (64) | 9 (36) | - | - | Epidemiologic transmission pattern was anthroponotic and anthropozoonotic |
| 16 | Hooshyar et al. (25) | Cross-sectional | Kashan | Medical Diagnosis Laboratories | 47 | PCR (Based on the gdh gene) | RFLP | 24 (51.1) | 9 (19.1) | 11 (23.4) | 3 | Epidemiologic transmission pattern anthropozoonotic |
| 17 | Effati et al. (26) | Cross-sectional | Alborz Province | Shahid Bahonar Children's Hospital of Karaj And Medical diagnostic laboratories Fardis, Nazar abad and Eshtehard | 15 | Nested-PCR (Based on the tpi gene) | Seq | 2 (13.3) | 5 (33.3) | - | 8 (53.4) | Epidemiologic transmission pattern anthropozoonotic and zoonotic |
| 18 | Kashinahanji et al. (27) | Cross sectional | Hamedan | Clients of health centers | 23 | Nested-PCR (Based on the tpi gene) | Seq | 18 (78.2) | 5 (21.7) | - | - | Epidemiologic transmission pattern zoonotic |
| 19 | Shahnazi et al. (28) | Cross sectional) | Qazvin | People who come in contact with food | 20 | PCR (Based on the gdh gene) | Seq | 11 (55) | 6 (30) | - | 3 | |
| 20 | Mirrezaie et al. (29) | Cross sectional | Andimeshk | Imam Ali Hospital and Health Center in Andimeshk | 40 | PCR (Based on the gene gdh and β-giardin) | Seq | 23 (57.5) | 17 (42.5) | - | - | Only the number of samples that were sequencing was listed, but 84 samples were tested for PCR. |
| 21 | Mahmoudi et al. (30) | Cross-sectional a | Rasht | Visitors to 3 educational hospitals of Gilan University of Medical Sciences | 41 | PCR (Based on the gdh gene) | Seq | 38 (92.6) | 3 (7.4) | - | - | 60 samples were PCR, but 41 samples were sent for sequencing. |
| 22 | Rafiei et al. (31) | Cohort study | Shushtar | Referrals to health centers | 24 | PCR (Based on genes SSU-rRNA ) | Seq | 12 (50) | 12 (50) | - | - | Based on SSU-rRNA gene 90 samples was PCR test, which confirmed 82 samples, but PCR based on gdh, Tpi, and bg genes was performed on 24 samples |
| 23 | Abdi et al. (32) | Cross sectional | Arak | Giardia-infected samples from Arak University of Medical Sciences Medical Diagnosis Laboratory it was gathered | 25 | PCR (Based on the betagiardin gene) | Seq | 20 (80) | - | - | - | Assemblage of 5 samples was unknown |
Extraction of Information About the Assemblage of Giardia intestinalis from the Selected Articles a
The results of statistical analysis to estimate the prevalence of A, B, as well as the mixed assemblages of G. intestinalis in the Iranian population are as follows.
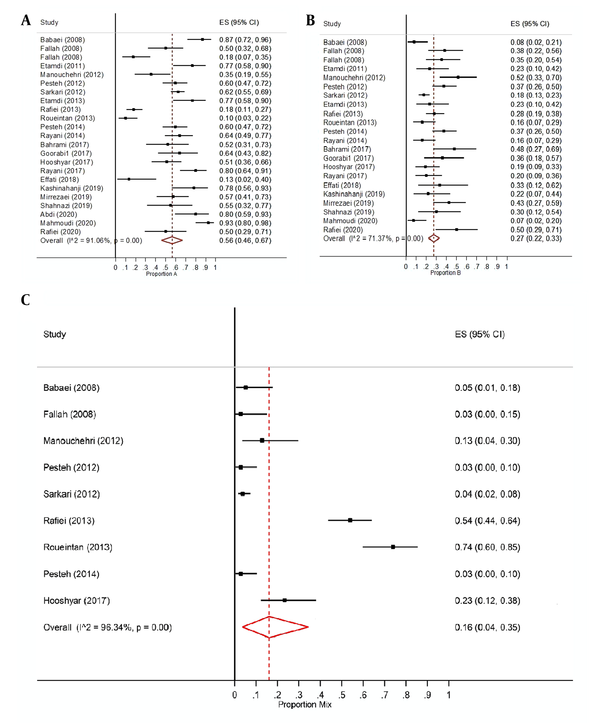
Results of the Egger test revealed that in 23 studies, the proportion of assemblage A lacked publication bias (P = 0.72), the proportion of assemblage B, had publication bias (P = 0.086), while the proportion of both assemblage (mix) was deficient in publication bias (P = 0.71). On the other hand, results of the Cochran's Q test and I2 statistic showed a heterogeneity among studies for assemblages A (I2 = 91.06%, Q = 246.02, P-value<0.001), B (I2 = 71.37%, Q = 73.35, P-value < 0.001), and mixture of both assemblages (I2 = 96.34%, Q = 218.55 I2, P-value < 0.001). So, we applied the random effects model in this meta-analysis. Figure 3A & B) displays the forest plot for the estimated proportions of assemblage A and B with confidence interval of 95% in western half of Iran. As shown in this figure, the overall proportion of assemblage A in this region of country is 0.56 with a confidence interval of 95% (0.46, 0.67) (P < 0.001), whilst the overall proportion of assemblage B has been estimated at 0.27 in the same region with a confidence interval of 95% (0.22, 0.33) (P < 0.001). Moreover, Figure 3C presents the forest plot for the mixed proportion of both A and B assemblages. This figure shows that the overall mixed proportion of both cases is 0.16 in mentioned area with a confidence interval of 95% (0.04, 0.35) (P < 0.001).
As noticed in Table 2, out of 23 selected studies, sub-assemblages of G. intestinalis have been presented in 16 studies.
| Line | First Author | Place of Study | Sample Size | Assemblage (Genotype or Sub Assemblage) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| A | B | Mix | Neg | |||||||
| AI | AII | BIII | BIV | B Novel | ||||||
| 1 | Babaei et al. (10) | Tehran | 38 | 33 (87) | - | 3 (7.8) | - | - | 2 (5.2) | - |
| 2 | Fallah et al. (11) | Tabriz | 34 | - | 6 (33.3) | 8 (44.4) | 4 (22.2) | - | - | 16 |
| 3 | Etemadi et al. (13) | Kerman | 30 | 5 (16.6) | 18 (60) | 7 (23.4) | - | - | - | - |
| 4 | Pestehchian et al. (14) | Esfahan | 67 | - | 40 (59.7) | 23 (34.32) | 2 (2.98) | - | 2 (2.98) | - |
| 5 | Sarkari et al. (15) | Fars province | 205 | - | 128 (74.41) | 30 (17.44) | 6 (3.49) | - | 8 (4.66) | 33 |
| 6 | Rafiei et al. (17) | Ahwaz | 100 | - | 18 (18) | 28 (28) | - | - | 54 (54) | - |
| 7 | Roointan et al. (18) | Ahwaz | 50 | - | 5 (10) | 8 (16) | - | - | 37 (74) | - |
| 8 | Etemadi et al. (19) | Kerman | 30 | 5 (16.6) | 18 (60) | 7 (23.4) | - | - | - | - |
| 9 | Rayani et al. (21) | Shiraz | 50 | - | 32 (80) | 4 (10) | 4 (10) | - | - | 10 |
| 10 | Rayani et al. (22) | Shiraz | 40 | - | 32 (80) | 4 (10) | 4 (10) | - | - | - |
| 11 | Bahrami et al. (23) | Kurdistan | 23 | 4 (17.4) | 8 (34.8) | 5 (21.7) | 4 (17.4) | 2 (8.7) | - | - |
| 12 | Hooshyar et al. (25) | Kashan | 47 | - | 24 (51.1) | 7 (14.9) | 2 (4.2) | - | 11 (23.4) | 3 |
| 13 | Effati et al. (26) | Alborz | 4 | - | 1 (25) | 2 (50) | 1 (25) | - | - | - |
| 14 | Shahnazi et al. (28) | Qazvin | 20 | 11 (55) | 6 (30) | - | - | - | 3 | |
| 15 | Mahmoudi et al. (30) | Rasht | 41 | - | 38 (92.6) | - | 3 (7.4) | - | - | - |
| 16 | Rafiei et al. (31) | Shushtar | 24 | - | 12 (50) | 12 (50) | - | - | ||
Extraction of Information About Sub-assemblages of Giardia intestinalis from the Reviewed Articles a
Results of statistical analysis are presented below to estimate the proportion of sub-assemblages A, B, and mixed of Giardia intestinalis in the population of the western half of Iran.
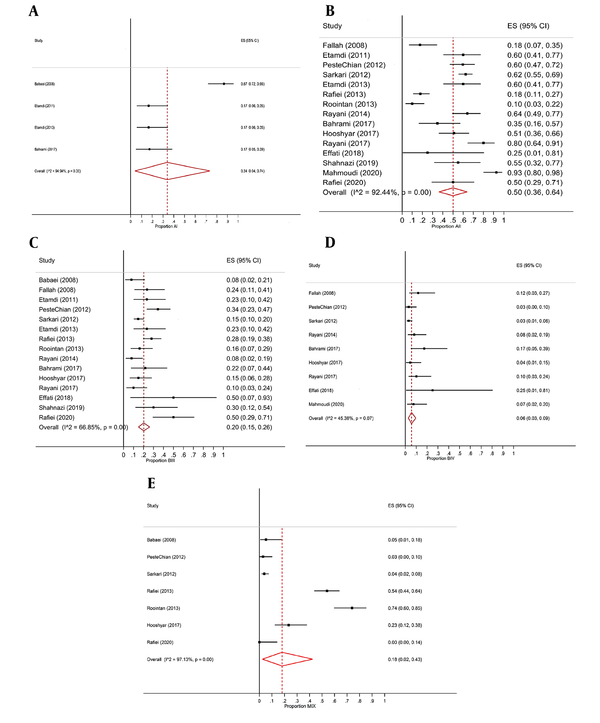
Furthermore, results of the Egger test indicated that the proportion of Giardia intestinalis sub-assemblages A (AI, AII) in the mentioned area lacked publication bias (P > 0.05). On the other hand, the Cochran's Q test and I2statistic showed a heterogeneity among the studies for AI sub-assemblages (I2 = 97.13%, Q = 209.02, P-value <0.001), and AII sub-assemblages (I2 = 92.44%, Q = 185.11, P-value <0.001). So, we applied the random effects model in this meta-analysis. As seen in Figure 4A-D, the overall proportion of AI (Figure 4A) was estimated 0.34 with a confidence interval of 95% (0.04, 0.74) (P < 0.05). In addition, the overall proportion of AII (Figure 4B) was estimated to be 0.5 with a confidence interval of 95% (0.36, 0.64) (P < 0.001).
Similarly, results of the Egger test demonstrated that the proportion of BIII (Figure 4C) sub-assemblages had no publication bias (P > 0.05), while the proportion of BIV sub-assemblages did (P < 0.05). The Cochran's Q test and I2 statistic for BIII sub-assemblages (I2 = 66.85%, Q = 42.23, P-value < 0.001), and BIV sub-assemblages (I2 = 45.38%, Q = 14.65, P-value <0.1) showed heterogeneity among studies. Therefore, the random effects model was used in the proportion meta-analysis of G. intestinalis sub-assemblages in the western half of Iran. Additionally, based on Figure 4D), the overall proportions of BIII and BIV were estimated to be 0.2 [with a confidence interval of 95% (0.15, 0.26) (P < 0.001)] and 0.06 [with a confidence interval of 95% (0.03, 0.09) (P < 0.001)], respectively.
On the other hand, results of the Egger test indicated that the mixed proportion of different sub-assemblages of G. intestinalis in mentioned region lacked publication bias (P = 0.675). In addition, outcomes of the Cochran's Q test and I2 statistic verified the heterogeneity among the studies (I2 = 97.13%, Q = 209.02, P-value < 0.001). Therefore, the random effects model was used in the proportion meta-analysis for various sub-assemblages of G. intestinalis. Eventually, based on Figure 4E, we can notice that the overall proportion for various G. intestinalis sub-assemblages is 0.18 with a confidence interval of 95% (0.02, 0.43) (P < 0.001).
4. Discussion
In this systematic study, the prevalence of assemblages and sub-assemblages of human G. intestinalis in different geographical regions of Iran was analyzed. The result of this study showed that most of studies on assemblages and sub-assemblages of G. intestinalis have been performed in the western half of Iran. There was little of the same information in the eastern half of this country. The main results of this study are as follows.
The overall prevalence of assemblages A, B, and the mixed assemblage (A and B) in the mentioned area were 0.56, 0.27, and 0.16, respectively. Also, prevalence of AI, AII, BIII and BIV sub-assemblages of were 0.34, 0.5, 0.2 and 0.06, respectively. In addition, the total prevalence of all sub-assemblages (AI, AII and BIII, BIV) was estimated to be 0.18.
Giardiasis is one of the most important health problems worldwide because the parasite that causes this disease, G. intestinalis, is a gastrointestinal protozoan common among human, domestic, and wild animals (3, 5). Hence, the epidemiological study of Giardiasis is important to identify the host spectrum of different species of this parasite as well as its assemblages, sub-assemblages, strains and genotypes. On the other hand, such studies, if performed using molecular methods, will help us improve our understanding about the zoonotic transmission potential of animal’s Giardia species and to determine how many cases of human Giardiasis have animal source (3). Although being sporadic, molecular taxonomic studies can also clarify the relationship between the parasitic and its host genotypes, pathogenesis, and clinical symptoms (33).
Also, different species of Giardia parasite have different hosts. Currently, six species of this parasite have been recognized by researchers. Of these six species, only G. intestinalis (also known as G. lamblia and G. duodenalis) could infect humans and many other mammals. Studies have shown that these species of Giardia have the widest range of hosting and the greatest health importance (34). The results of epidemiological and genotypic studies have also confirmed the possibility of zoonotic transmission of G. intestinalis (33).
Study on allozymes has further shown that all human isolates of G. intestinalis belong to two genetic assemblages (A and B) each of which contains at least four genetic clusters or sub-assemblages (I to IV). On the other hand, both of these assemblages are zoonotic whose examination can help us track the source of infection (35). Further, in Brazil, the findings of two separate studies showed that in communities, where people care for pets, humans and animals (especially cats) could become infected with Giardia assemblage A. In such communities, there seems to be the possibility of cross-transmission of this parasite between animals and humans (36, 37). Another study in Poland verified assemblage A in Giardia infected cats (38). Research works in Mexico, Spain, and Jamaica approved the same assemblage of G. intestinalis (A) in dogs that had close connection with human (39-41). Furthermore, a study conducted to determine the sub-assemblage of G. duodenalis in livestock of Urmia, Iran showed that the predominant assemblage among the livestock of this city is type E, which is not zoonotic (42). Similar results were obtained in other studies conducted on Arabian horses, and ruminants in Ahvaz and Yazd province, Iran, respectively (43, 44). As well as studies performed in Turkey on horse and cattle (45, 46).
As the results of this systematic study showed, prevalence of assemblage A of G. intestinalis with estimated of 0.56 is the dominant assemblage in the human society of western half of Iran. By comparing Giardia sub-assemblages in animals and humans, it can be concluded that in mentioned region of Iran zoonotic transmission of this parasite from livestock and pets is very scarce. In contrast, a general comparison between the prevalence of Giardia in this region of Iran, Brazil, Mexico, Spain, and Jamaica suggested that the source of contamination in these countries may be stray domestic animals, including cats and dogs (36, 39-41). Currently, the large number of stray cats and dogs are the most important urban problems in Iran. These animals can be involved in the transmission and spread of a number of zoonotic parasitic infections including Giardia. Works on the prevalence and genotype determination of Giardia in Iranian domestic animals is so little that we had to report results of the only study in this field in Iran, which showed that the prevalence of G. duodenalis in cats in Ahvaz was 2% and 3.33% by microscopic method and immunochromatography assay, respectively (47).
Moreover, studies in other countries showed that assemblage A of G. intestinalis had the most possibility of zoonotic transmission since it is the most common species of this parasite in animals and it also lacks a specific host. It should be also noted that humans are usually infected with genotype AII, while animals are often infected with type AI (34). Results of three studies conducted in Iran to determine the genotypes of the G. duodenalis in animals also showed that although the predominant assemblage of the parasite in these animals is of category E, a number of these animals were infected with assemblage A and genotype AI (42-44). Thus, it would be likely that the source of zoonotic transmission of Giardia is not livestock (cattle) and horses and there might be other animals responsible for this transmission in Iran.
The results of determining assemblage of Giardia in two neighboring countries of Iran showed that the dominant assemblage in Saudi Arabia and Turkey were A and B, respectively (4).
In addition, the association between the genotypes of the G. intestinalis and clinical symptoms was investigated in a human experimental study. The results showed that the clinical signs of Giardiasis appear only in people infected with the GS / M isolate of the parasite. This insolate belongs to Assemblage B of Giardia (3, 33). Further, results of this study led to a theory stating that pathogenesis of parasite changes with variation of parasite strain. Results of animal experimental studies also reinforced this theory (3, 33). Moreover, a study on the infected Egyptian school children confirmed the symptoms of Giardiasis appeared in patients with Assemblage B of Giardia (48). A similar study in Cuba also confirmed the same assemble (B) (48, 49).
In Iran, several studies have been performed to investigate the relationship between Giardia genotype and the presence or absence of clinical signs. For example, Rafiei et al. showed that the predominant genotype of Giardia in Ahvaz is AII and BIII, but no significant difference was observed between the presence/absence of symptoms and the incidence of these genotypes (17). In another study by Etemadi et al., patients with assemblage A suffered from milder symptoms (mild diarrhea) than those with assemblage B (13).
On the other hand, according to the findings of the current study, it seems that in most studies that have been performed to determine the genotype of Giardia parasite in Iran, in fact, sub-assemblages the parasite were identified and reported.
To achieve more accurate results in Iran and have the possibility for better comparisons with other countries’ studies, it is necessary to conduct more comprehensive studies in different parts of Iran regarding the prevalence and genetic characteristics (assemblage, sub-assemblage and genotype) of G. intestinalis in humans, livestock, and pets.
4.1. Conclusions
Taking into account all the above-discussed studies, it is obvious that the epidemiological transmission pattern of G. intestinalis in Iran is zoonotic transmission. However, it seems important to determine the genetic characteristics of the parasite in order to discover the source of contamination in human societies. Therefore, to reduce the risk of this disease transmission from animal to human (zoonotic), revision of treatment, prevention and control guidelines of this disease in cats and other pets is a priority. On the other hand, the results of the current study shed new light on the prospects and opportunities for future research in the field of epidemiology and genetics of this parasite. Hence, we recommend researchers to use the results of this systematic study to identify areas of Iran that need research in the field of genetic and epidemiological studies of Giardia.