1. Background
Pain is an unpleasant, complex, multidimensional, unique experience associated with potential or actual tissue damage (1). Usually, postoperative pain lasts for 2 - 3 hours or more, and the severity of pain depends on site of surgery, the patients’ body response to injury, and individual threshold for pain (2). Acute postoperative pain is a complex physiological response to the edema and trauma of surgery. The consequence of prolong uncontrolled pain can lead to some complications such as respiratory complications, risk of deep vein thrombosis (DVT), prolonged intestinal dysfunction, and prolonged hospitalization (3). Compared to well-studied management of acute postoperative pain, studies on chronic pain management with high incidence after some surgeries (10% to 65%) are limited (4).
The prevalence of chronic postoperative pain varies between 5 - 85 % and depends on the type of surgery (5). Most importantly, it involves the neuropathic component seen even in the initial stages after surgery (6).
Breast cancer is known as the most common malignancy in women and strongly affects women's health and quality of life. Common breast cancer treatment is surgery, which is performed as a partial mastectomy with or without excision of the axillary lymph node (simple), or a radical mastectomy with axillary lymph node dissection (7). Although advances in the treatment of breast cancer have increased the life expectancy of these patients, unfortunately, most of these treatments are associated with many side effects, which significantly diminishe the quality of life (8). Post-mastectomy pain syndrome (PMPS) is one of the significant complications of breast cancer treatment with a prevalence of about 20% - 68%, that is a neuropathic pain and seen in and around the surgical site and lasts more than three months after surgery (9).
Many drugs are used to treat chronic postsurgical neuropathic pain, including antidepressants, anticonvulsants, N-methyl-D-aspartate receptor antagonists, stabilizer of cell membranes (lidocaine), and Alpha-2 agonists such as dexmedetomidine (6, 10).
Dexmedetomidine is an α2 agonist, facilitated analgesia and anesthesia in humans and reduced the severity of postoperative pain and nausea (11). The use of preoperative α2 receptor agonists also improves hemodynamic stability due to its numerous beneficial effects, including analgesic effects, inhibition of sympathetic outputs, anti-anxiety properties, and reduction of norepinephrine doses. Moreover, it has myocardial protection due to its positive effects on myocardial oxygen supply and demand (12). Dexmedetomidine alone or in combination with other analgesics is used to control pain after surgery (13). This is supported by different studies like Ge et al., who concluded the use of dexmedetomidine as a suitable drug for the treatment of pain after surgery (14). Furthermore, the result of a study which was done by Jain et al. on 84 patients with breast cancer showed that infusion of dexmedetomidine at a dose of 2 μg/kg before breast surgery significantly reduced pain scores, need for analgesia during the first 72 hours, as well as the severity of chronic pain three months after surgery (15). The other study by Sitilci et al. on 69 patients with breast cancer concluded a similar decrease in pain score after mastectomy following infusion of this drug at a dose of 0.5 μg/kg/h (13).
However, the majority of studies evaluated the sedative and analgesic effects of preoperative or postoperative dexmedetomidine on acute postoperative.
2. Objectives
The aim of this study was to evaluate the effect of different doses of dexmedetomidine intraoperatively infused on acute and chronic pain checked concomitantly after simple mastectomy.
3. Methods
This is a randomized, double-blind clinical trial, which was done in Shahid Faghihi Hospital of Shiraz University of Medical Sciences. The patients who were scheduled for simple mastectomy from October 2018 to May 2019 were enrolled in the study after obtaining approval from the Ethics Committee. The Code of Ethics Committee and IRCT code registered for this study was IR.SUMS.MED.REC1398.297 and IRCT 20141009019470N90, respectively.
The sample size was calculated based on pain scores from the previous study, the effect of the perioperative infusion of dexmedetomidine on chronic pain after breast surgery (15). The sample size estimated 55 cases for each group, using moderate effect size = 0.5, power 80%, at a significance level of 5% with dropout rate of 10%. Moreover, 110 patients were randomly assigned to two groups using block randomization in blocks of size 10 (list blocks was extracted from www.sealedenvelope.com). In this regard, the study population consisted of 110 patients with the age of 18 to 65 years and (American Society of Anesthesiologist) ASA physical states I or II who signed written informed consent and underwent elective simple mastectomy. Patients with body mass index (BMI) more than 30 kg/m2, any hepatic, cardiac, and renal dysfunction (Cr > 1.5), long term diabetes (> 10 years), smoking, alcohol abuse, arthritis, and chronic use of analgesics, known psychiatric disorders, and pregnant women were excluded from the study.
The demographic information was collected with the modified questionnaire. It was used to collect patients’ information such as age, weight, BMI, history of cardiovascular disease, diabetes, hypertension, and hypothyroidism. Visual Analogue Scale (VAS) was used to collect information of acute pain in recovery room and first 24 hours postoperatively, and the standardized questionnaire of BPI (Brief Pain Inventory) was chosen for measuring chronic postoperative pain three months after surgery via a telephone call.
VAS (Visual Analogue Scale) for pain is a self-reported linear-visual pain measurement tool, which is included a 10 cm ruler with the word “painless” written on the left end and the” most severe pain” on the right end. It is divided from zero to ten scale where (0 - 1) present no pain and (10) is defined as Unbearable pain. BPI consists of two main parts, measuring the severity of pain and the extent of pain interference with daily affairs. The part that measures the severity of pain (sensory dimension) consists of four questions, and the part that measures the extent of pain interference with the general functions of individuals (reactive dimension) consists of seven questions (16).
In operation room, the type of anesthesia and the pain measurement tool (VAS) were described to the patients. All patients underwent standard monitoring, which included pulse oximetry, electrocardiogram (ECG), non-invasive blood pressure, and End-Tidal carbon dioxide (ETco2) levels. Eligible patients in both groups were received midazolam (0.05 mg/kg body weight), fentanyl (1 - 3 μg/kg body weight), morphine (0.1 mg/kg body weight) as premedication, and thiopental (3 - 5 mg/kg body weight), and Atracurium (0.5 mg/kg body weight) for induction of anesthesia. Intubation was performed with an endotracheal tube of appropriate size. Anesthesia was maintained with isoflurane (1.5% - 2.5%), and a mixture of 50% - 50% oxygen and nitrous oxide and ETco2 was maintained between 35 - 40 mmHg using controlled mechanical ventilation.
During the operation, if the increase in mean arterial pressure and pulse rate was more than 20% - 30% of the initial rate, a single dose of fentanyl was used at a dose of 1 mg/kg body weight and recorded in the data collection form. To prepare dexmedetomidine for infusion, the volume of vial of dexmedetomidine (two cc), equal to 200 micrograms, was increased to 40 cc by adding normal saline (equivalent to five micrograms per cc). For the placebo group, we drew and prepared 40 cc of normal saline in a 60-cc syringe, and based on protocol: 0.08 cc/kg/h of the solutions were given to each group.
At the end of surgery, the patients of both groups received the full dose of reverse. After returning spontaneous respiration with adequate tidal volume (at least 5 cc/kg) and the normal response to muscle power examination, the patients were extubated and transferred to recovery room. During the 2-hour stay of patients in recovery room (every half-hour) and in the first 24 hours after surgery (every four hours), the presence and severity of acute pain were measured by VAS criteria.
The responsible nurses checked the patients and infused analgesic medication in coordination with an anesthesiologist and based on the result of VAS of pain as shown in Table 1.
| Patient Pain Score (VAS) | Analgesic |
|---|---|
| 1 < VAS < 3 | 1 gram of Apotel |
| 3 < VAS < 7 | 1 mg of morphine was given and repeated every 15 minutes until the VAS dropped below three. |
| 7 < VAS < 10 | Two mg of morphine was given and repeated every 15 minutes until the VAS dropped below 7. |
Importantly, the patients and nurse anesthetist who used study drugs or collected the data were blinded to study groups during and after the procedure.
3.1. Statistical Analysis
In this study, continuous variables were reported as mean with standard deviation, and the comparisons were done between groups by independent sample t-test and paired sample t-test. Categorical variables were presented as numbers and percentages, and the comparisons between groups were performed by chi-square and Fisher exact test. The data were analyzed using the statistical package for social science (SPSS Inc., Chicago, version 23) and P-value < 0.05 was considered statistically significant.
3.2. Ethical Considerations
This study was performed after obtaining ethics approval from Ethics Committee of Shiraz University of Medical Sciences and receiving clinical trial code number from Iran Clinical Trial Registration Center and informed consent of patients. Observance of Islamic decency, especially respect for human rights, was one of the requirements of the researcher. A letter of introduction was taken from the vice chancellor for Research of Shiraz University of Medical Sciences to conduct the research, and it was given to the director of Shahid Faghihi Medical Center. The purpose of the research was explained to the relevant authorities, and their cooperation was obtained.
4. Results
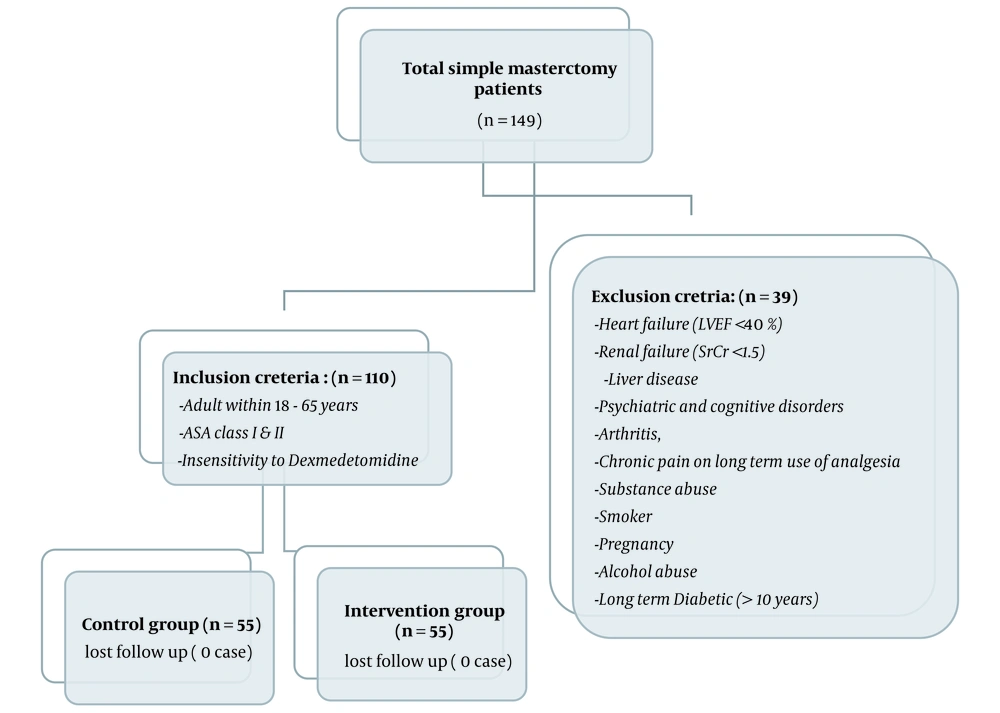
One hundred forty-nine patients with simple mastectomy during this period were screened. Thirty-nine patients who did not meet inclusion criteria or due to changes of surgical procedure for any reason were excluded (Figure 1). One hundred and ten eligible patients who were signed the informed consent form were randomly divided into intervention (n = 55) and control (n = 55) groups using permutation block randomization method. The results of demographic questionnaire of patients are reported in Table 2. There was no significant difference between the two groups in terms of age, weight, BMI, and history of diabetes, hypertension, cardiovascular disease, and hypothyroidism (P > 0.05).
| Variables | Total | Control Group (N = 55) | Variable Intervention (N = 55) | P-Value |
|---|---|---|---|---|
| BMI | 26.30 ± 1.27 | 26.21 ± 1.37 | 26.39 ± 1.16 | 0.376 |
| Age | 48.15 ± 1.55 | 48.3 ± 1.5 | 48 ± 1.6 | 0.112 |
| Weight | 74.35 ± 2.2 | 73.7 ± 2.3 | 75 ± 2.1 | 0.216 |
| History of diabetes | 21 (19) | 12 (57) | 9 (43) | 0.467 |
| History of hypertension | 29 (53) | 17 (59) | 12 (41) | 0.279 |
| History of cardiovascular disease | 5 (4.5) | 2 (40) | 3 (60) | 0.647 |
| History of hypothyroidism | 8 (7.3) | 5 (62.5) | 3 (37.5) | 0.463 |
a Values are expressed as mean ± SD or No. (%).
The results of VAS pain score of patients are reported in Table 3. Based on the finding of the study, there was a significant difference between the intervention and control groups in terms of VAS pain score (P-value < 0.001). Moreover, there was a significant difference in terms of time to first request of analgesia (Apotel, Morphine) in the first 24 hours after surgery between the two groups (P-value = 0.036). There was also a significant difference between the study groups in terms of the required analgesic (P-value < 0.001). Furthermore, the results of the study showed that in both groups, the majority of patients did not receive Apotel analgesia, and in this regard, there was no significant difference between the two groups (P-value = 0.503). Also, the results showed a significant difference between the two groups in terms of receiving one and two mg morphine (P-value < 0.001).
| Variables | Control Group | Variable Intervention | P-Value |
|---|---|---|---|
| Average VAS in recovery | 4.44 ± 2.25 | 0.96 ± 0.14 | < 0.001 |
| Frequency of requests for any type of analgesic | < 0.001 | ||
| 0 | 16 (100) | 0 | |
| 1 | 39 (100) | 0 | |
| 2 | 0 | 29 (100) | |
| 3 | 0 | 26 (100) | |
| Frequency of receiving of one mg of morphine | < 0.001 | ||
| 0 | 49 (98) | 1 (2) | |
| 1 | 6 (20) | 24 (80) | |
| 2 | 0 | 29 (100) | |
| 3 | 0 | 1 (100) | |
| Frequency of receiving of two mg of morphine | 55 (68) | 26 (32) | < 0.001 |
| 0 | 28 (100) | ||
| 0 | 1 (100) |
a Values are expressed as No. (%) or mean ± SD.
The Brief Pain Inventory (BPI) was used to evaluate the effect of dexmedetomidine on chronic postoperative pain. The analysis of this questionnaire on pain, three months postoperative, after simple mastectomy showed that there were no significant differences in terms of chronic pain between the two groups (P-value = 0.871, Table 4).
| Chronic Pain | Intervention Group | Control Group | P-Value |
|---|---|---|---|
| Patients with pain | 3 (5.5) | 52 (94.5) | 0.871 |
| Patients without pain | 7 (12.7) | 48 (87.3) |
a Values are expressed as No. (%).
5. Discussion
This was a double-blind, randomized clinical study that was conducted to evaluate the effect of intraoperative infusion of dexmedetomidine on acute and chronic pain management after simple mastectomy. This study was done on 110 patients (55 people in each group). Based on the result of this study, the mean VAS pain score in the intervention group is significantly lower than the control group. Moreover, the number of requests and the amount of analgesia (Apotel and Morphine) in the first 24 hours after surgery in the intervention group was significantly less than patients in the control group. Also, the time of the first request for analgesia in the patients of the intervention group was significantly longer than control group.
Dexmedetomidine is a cost-effective agent with favorable safety profile, especially compared to common respiratory depression side effects of narcotic and other analgesic agents. The effectiveness of this agent on management of acute pain and limiting amount of pain killer intake after operation was evaluated in different operations. Similar to the study by Ge et al., infusion of dexmedetomidine during abdominal hysterectomy on patients reduced postoperative morphine intake (14). In lumbar surgery, the result of studies consistently showed improvement in acute pain management and quality of life in patients who received dexmedetomidine compared to the placebo or other alternative managements (17, 18). Even in cardiovascular operation, the usage of this medication was studied, and its significant effects were proven on reduction of need for analgesia for up to 24 hours postoperative (19).
In case of simple mastectomy, different studies were conducted on the effect of dexmedetomidine on postoperative complications of patients. For instance, in one study that included 56 patients who underwent radical mastectomy and general anesthesia, dexmedetomidine had a significant effect on improving sleep disorders and reducing acute pain of patients (20). One of the important outcomes of use of dexmedetomidine, which was studied previously, is the extended time of need for the first dose of analgesic post operatively (21, 22). We found a similar outcome in our intervention group; the time to first complain of pain in the surgical ward was significantly longer in the dexmedetomidine group than in the control group. Also, morphine dose was significantly lower in the dexmedetomidine group than in the control group. Furthermore, the other finding of this study was that the mean pain score in patients in the intervention group was significantly lower than patients in the control group. Moreover, the number of requests for analgesia in the first 24 hours after simple mastectomy was lower in the intervention group than in the control group, and the maximum number of required analgesic for the first 24 hours increased up to three times in the control group. These finding is the same as previous finding of some other studies like Alipour et al. where the mean pain intensity at 1, 3, 6, 12 and 24 hours postoperatively in the intervention group was significantly lower than the control group. Also, the mean total dose of tramadol in the intervention group was significantly lower than in the control group (23). After three months of surgery, the study showed that although the incidence of chronic pain was different in the intervention and control groups, there was no statistically significant difference between the two groups.
Although studies are consistent on the effectiveness and safety of dexmedetomidine in reduction of acute postoperative pain, there are controversial outcomes from limited low grade of studies about its effectiveness in chronic pain. For instance, some studies were noted that dexmedetomidine had no effect on chronic pain (22). While the results of research by Jain et al. on 84 patients with breast cancer showed that dexmedetomidine infusion before surgery significantly reduced the acute postoperative pain and decreased the need for analgesia during the first 72 hours, and also minimized the severity of chronic pain three months after surgery in patients undergoing breast cancer surgery (15). The results of this study are consistent with our study in terms of the effectiveness of dexmedetomidine on pain relief and analgesia, but about the long-term effect of dexmedetomidine on chronic pain in the study of Jain et al., different outcomes were achieved. This discrepancy may be due to the difference in the sample size, the dose of study drug, the pain measuring instrument, and importantly recent advances in surgical technique.
5.1. Limitations
One of the most important limitations of our study was that we did not use PCA pump for pain control during the postoperative period because in the tertiary center where this study was done there was no acute pain service for this purpose. Also, in this study, only one dose of dexmedetomidine was used; therefore, further studies are needed to determine the appropriate dose of dexmedetomidine for postoperative pain control. Moreover, the cost of dexmedetomidine can also be a problem. However, intraoperative dexmedetomidine may reduce the total amount of analgesics used to control postoperative pain, leading to cost-effective pain management. Furthermore, to improve dexmedetomidine usage in real practice, relevant administrators of hospitals can provide comprehensive information about this treatment for medical students and medical staff.
5.2. Conclusions
According to the finding of this study, dexmedetomidine has made a significant difference in the severity of pain and analgesic requirement of patients undergoing simple mastectomy; therefore, dexmedetomidine may be used as an adjunct to pain management in these patients. This medication reduces the need for analgesics and most importantly makes longer time for the request of first dose of postoperative painkillers. However, the results of this study did not show a significant difference between dexmedetomidine and placebo in the occurrences of chronic pain. Further studies with different doses and larger sample sizes are required to achieve more accurate results.