1. Background
Cancer is a major cause of mortality and one of the main obstacles to increased life expectancy in different countries (1). Based on a report by the World Health Organization (WHO) in 2019, cancer is the first or second major cause of mortality among people under 70 years in 112 countries. It is also known as the third or fourth cause of mortality in more than 23 countries (2). According to the GLOBOCAN 2020, with 9.5 million new cancer patients and 5.8 million deaths in 2020, cancer is considered a serious health problem in Asia (1).
Cancer patients are commonly treated with numerous treatment plans, such as radiotherapy, surgery, or chemotherapy, and receive multiple services from different specialists during treatment (3). The complexity of chemotherapy regimens, need for individualized dose adjustment based on the body surface area (BSA) or kidney function, use of cytotoxic drugs with a narrow therapeutic index, and services that provided by multiple staff, including nurses, clinical pharmacists, and medical oncologists make the chemotherapy process highly vulnerable to errors. Chemotherapy errors are considered the second most common cause of fatal medication errors (3-6). These errors occur in all cancer treatment plans and may happen in all phases of the chemotherapy process, including prescription, preparation, and administration. They include all types of medication errors, such as wrong patient treatment, administration of wrong regimen, drug, or drug dose, wrong route of administration, errors related to schedules or infusion, and compounding errors (6-8).
While the chemotherapy administration phase is complex and requires many different safety checkpoints to prevent errors, according to the literature, the highest rate of medication errors is related to the prescription phase, with almost 33% of all errors categorized as fatal or life-threatening for cancer patients (9). The chemotherapy preparation phase is usually considered a critical step; however, the risks and errors remain uncertain, because there are no studies specifically designed to detect these errors (8). Other issues of concern that may compromise the safety and quality of the chemotherapy process include long patient waiting time for chemotherapy due to uncertainties in the chemotherapy workflow and appointment schedules; poor communication and coordination between different care providers (e.g., oncologists, nurses, and pharmacists); and inconsistency in the paper-based documentation of prescriptions and administration information, making it difficult to find previous prescriptions and keep track of the chemotherapy cycle (3, 4, 10-12).
Improvement of the safety and quality of the chemotherapy process is the main goal of oncology centers in all hospitals (13, 14). Several strategies are employed to decrease chemotherapy errors and risks to cancer patients, such as the use of pre-printed standard forms, development of policies and procedures for the secure handling of high-risk medications, continuous training of healthcare providers, and use of a computerized physician order entry (CPOE) system (6, 15, 16). Studies show that implementation of a CPOE system as a tool for electronic medication ordering, especially chemotherapy agents, can prevent or significantly reduce medication errors in any stage of the medication use process (a 44 - 88% reduction in prescription phase errors) (5, 16-19).
Many hospital wards have considered CPOE as a beneficial and powerful intervention for the standardization of practice and improvement of patient outcomes (20-24). The CPOE system has been successful in enhancing secure electronic prescriptions by validating the input, eliminating possible errors, creating alerts, checking drug-drug interactions, performing automatic calculations, and directing orders based on standardized protocols (22, 25-27). Although the use of health information systems is of growing interest in many healthcare processes (28), their implementation in oncology wards, especially for the computerized management of chemotherapy, has been very slow (29, 30).
2. Objectives
In this study, we aimed to design and implement an information system that involves a CPOE system, a patient management system, a prescription verification system, and a nurse administration system. We also described how this system was established to improve the care of cancer patients who were candidates for chemotherapy at our hospital.
3. Methods
3.1. Setting
This study was conducted at the Department of Hematology and Oncology of Taleghani Teaching Hospital in north of Tehran, Iran. Taleghani Teaching Hospital is one of the largest referral cancer centers in Iran. In 2019, the oncology department of this hospital prescribed more than 20,849 chemotherapy orders, equivalent to 1.892 chemotherapy sessions per month in the inpatient and outpatient wards (24 hours a day, seven days a week).
3.2. Team
A multidisciplinary team involved in the development of the GWS system consisted of informaticians, and the chemotherapy team consisted of two oncologists, three oncology fellowships, and one chemotherapy nurse for the chemotherapy process assessment.
3.3. Chemotherapy Workflow
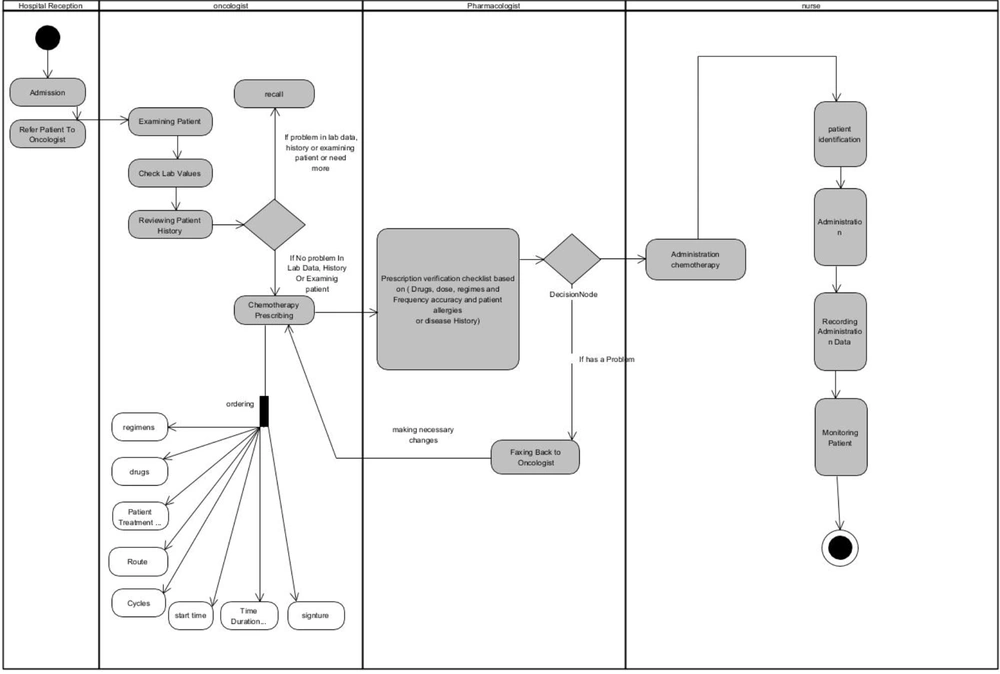
Eleven months before the system implementation, we made a clinical team composed of two oncologists, three oncology fellowships, and one chemotherapy nurse. Our goal was to analyze the current chemotherapy workflow and treatment protocols that led to a chemotherapy process flowchart. The flowchart was divided into four major parts, including the medical reception, chemotherapy prescription, prescription verification, and administration phase. Each step of the chemotherapy process included a subsection. Figure 1 presents the chemotherapy process workflow.
Generally, the chemotherapy process is initiated from the hospital reception, where the patient is admitted to the inpatient or outpatient ward. Appointment scheduling, documentation of patient information, and updating the patient status in the patient management system are carried out at this stage, and the patient is then referred to an oncologist. The oncologist logs onto the system, opens the patient’s profile, and calls the patient’s name from the waiting room to examine him/her, take his/her vital signs, check the laboratory results, and review his/her history from the patient management system. If a patient requires further testing or there is a problem in the laboratory results, physical examination, or history-taking, the chemotherapy prescription will be done in the next appointment. The oncologist can check the patient’s status or progress notes, as well as the history of treatment, by clicking the patient management module. The chemotherapy regimen can be automatically adjusted based on the patient’s demographic, clinical, and laboratory data, documented in the information system by clicking the chemotherapy prescribing module.
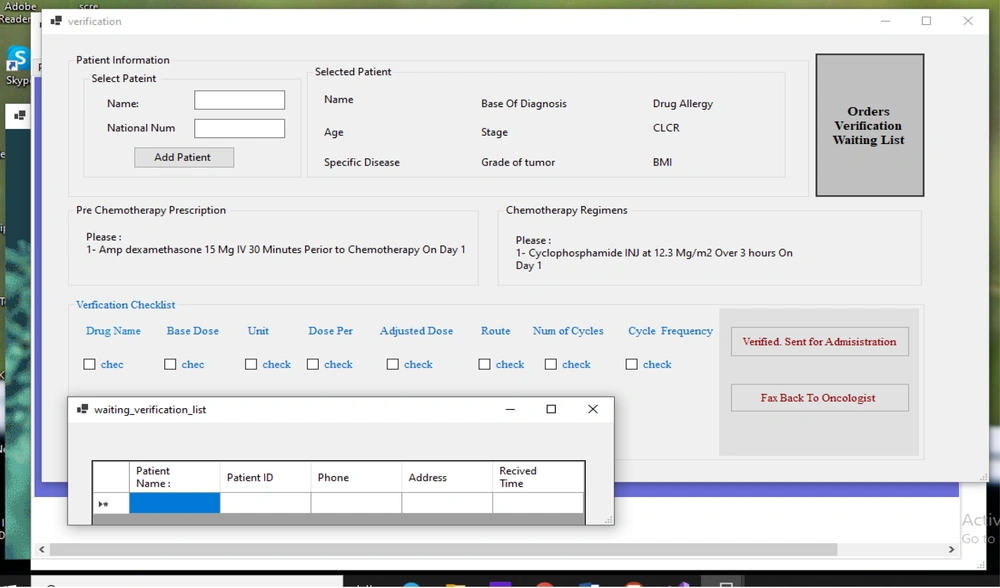
Before the administration phase in the nursing unit, each prescription should be reviewed by a clinical pharmacist according to standardized protocols. For this purpose, the prescription is sent from the chemotherapy prescribing interface to the verification system screen by the oncologist, and then, the clinical pharmacist checks and validates the prescription details, such as the following items: (1) information related to the patient, such as age, weight, BSA, level of serum creatinine, specific diseases, and history of allergies; (2) data related to the patient’s prescriptions, agreement between the cancer stage and the selected regimen, drugs and chemotherapy premedication, and selection of the prescribed administration route, cycle, and schedule; (3) data related to the drug, including the drug dose or cumulative dose (electronic signature and validation).
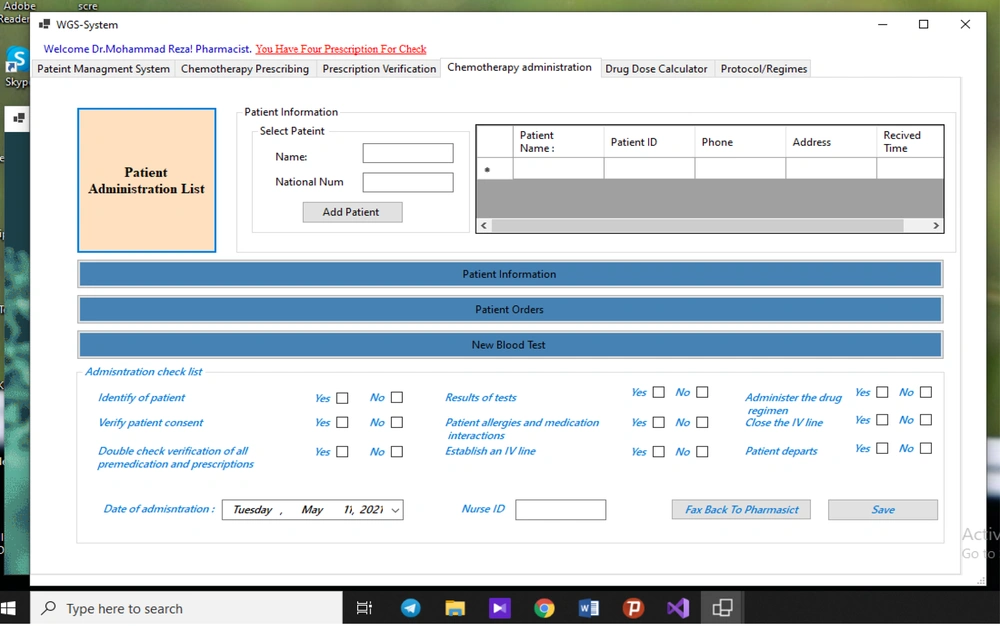
The oncology pharmacists receive prescriptions automatically. In the event of a problem with prescription, it is sent back to the oncologist; otherwise, the prescription is sent to the nurse portal for administration. The nurse logs onto the system and checks the prescriptions. When a patient arrives for an administration, the nurse checks his/her identity and condition for chemotherapy, prepares the prescription, verifies the drug, administers the chemotherapy drug, monitors patient’s status, and documents the drug administration data in the administration system.
3.4. Protocol Development
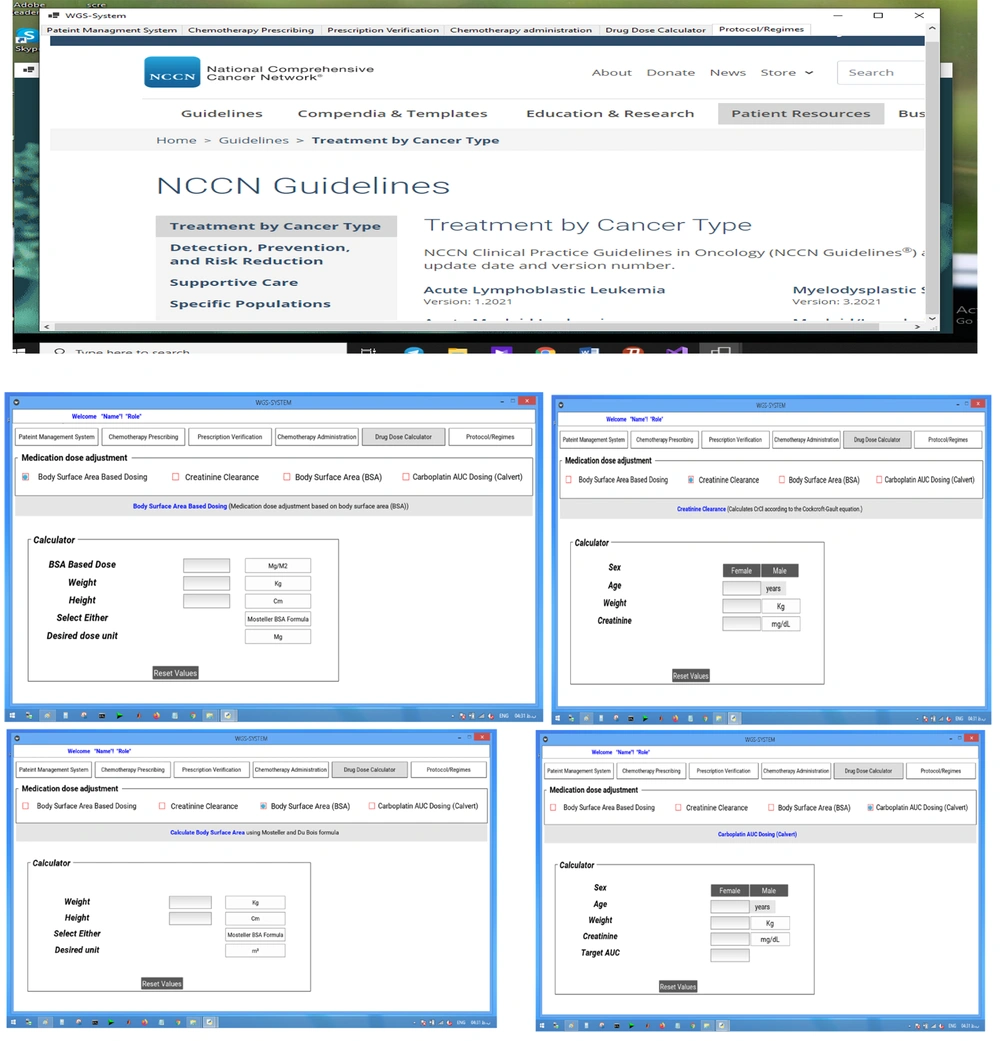
The Chemotherapy Council Group reviewed all available protocols and guidelines, such as the international guidelines, including the American Society of Clinical Oncology (ASCO), National Comprehensive Cancer Network (NCCN), Oncology Nursing Society (ONS), Cancer Care Ontario (CCO), Clinical Oncological Society of Australia (COSA), American Society of Health-System Pharmacists (ASHP), literature evidence, and local regulatory organizations of healthcare products (31-35). All existing chemotherapy protocols were compared with the local protocols. Finally, the structured chemotherapy order sets were standardized and developed by the chemotherapy council group from November 1, 2020, to January 1, 2021, and used as standard care guidelines for developing a clinical decision support system to be integrated in the CPOE system.
The structured order set identifies the chemotherapy regimen or protocol, the basic dose for automatic patient-specific dose calculation, protocols for hydration orders and pre-chemotherapy medications, cycle frequency, number of cycles, treatment criteria, and other information pertinent to administration. The structured templates also supports the clinical pharmacy workflow for verification of orders and the chemotherapy nursing unit for the administration phase. The council group matches each step of chemotherapy prescriptions in terms of the drug, dose, and sequence of guidelines for prescribing chemotherapy and antiemetic recommendations in the pre-chemotherapy stage.
3.5. Design and Implementation of a Guideline-Based Workflow Software System
Before system design and implementation, a team including three informaticians and oncologist, were formed to drive system design and implementation. The design of the guideline-based workflow software system was based on the needs assessment conducted by the participation of oncologists, clinical pharmacists, and nurses. All the chemotherapy work routines, including the reception phase, prescription phase, prescription verification stage, and administration, were assessed and documented on paper.
The first step in the design of the guideline-based workflow software system was establishing a minimum dataset that included demographic, clinical, and laboratory data in the patient’s profile in the patient management system. The conceptual model (use case, activity, class, and sequence diagram) of our system and database was developed by the committee, based on the processes identified in the previous steps, using an object-oriented analysis and design with the UML language. Specific forms for documenting patient information, safety checklists, templates for nursing administration, chemotherapy prescription verification forms for clinical pharmacists, and minimum datasets were developed by designing questionnaires in an iterative interdisciplinary collaboration.
The NCCN chemotherapy protocols for the hydration and chemotherapy prescription phases were planned and developed in the CPOE system. The design and implementation principles included all factors found to improve communication between healthcare providers, quality of chemotherapy and its safety (e.g., individualized dose adjustment based on BSA or kidney function, checking drug-drug interactions, and creating alerts), use of drop-down menus, and automatic elimination of interpretation and transcription errors.
After approval of the system design by our team, a guideline-based workflow software system was developed by the Python programming language as the front-end language and SQL as the back-end language. Our designed system generally consisted of a patient management system, a prescription verification system, and a nurse administration system, integrated in the CPOE system.
4. Results
4.1. Design Phase of the Guideline-Based Workflow Software System
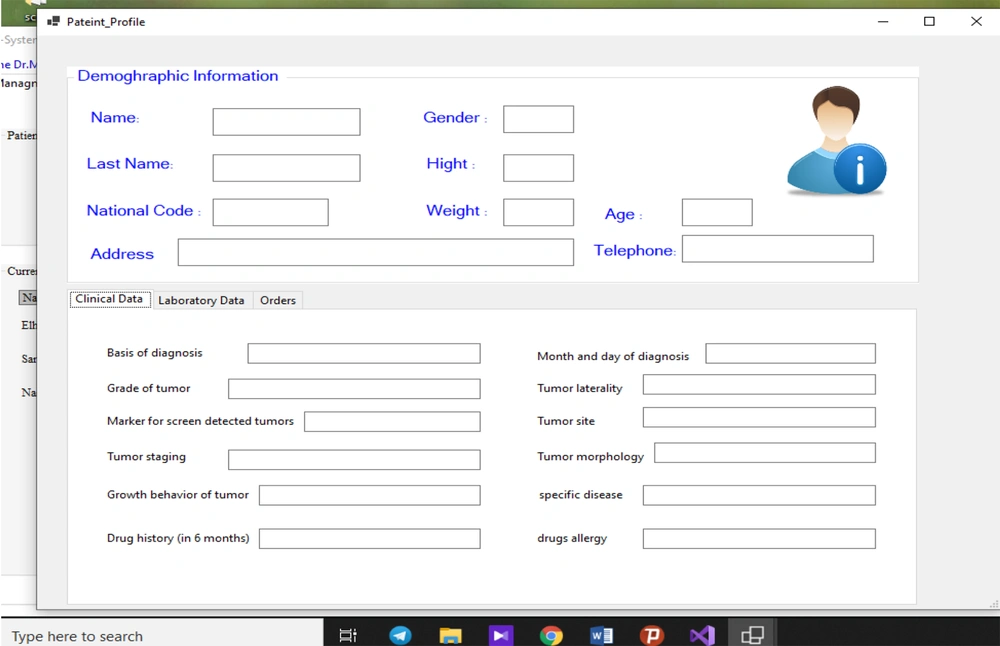
We initially developed a minimum data elements for patients undergoing chemotherapy and designed the respective examination forms for the electronic health records. The forms comprised of three sections, including demographic, clinical, and laboratory findings in the patient’s profile in the management system by developing questionnaires in an iterative interdisciplinary collaboration. Figure 2 shows the patient’s profile with the identified minimum data elements. The protocols were verified by two oncologists and three fellowships from November 1, 2020, to January 1, 2021.
The design for the guideline-based workflow software system is presented in four disparate diagrams, including a structural diagram (class diagram), functional diagrams (use case diagram, activity diagram), and behavioral diagram (sequence diagram), using the unified modeling language (UML). Three clinical staff were involved in the chemotherapy process, including an oncologist, a clinical pharmacist, and a nurse who played a critical role in different parts of the chemotherapy process. The use case diagram (Appendix 1A) and the chemotherapy prescription sequence diagram (Appendix 1B), the class diagram (Appendix 2A and database of proposed guideline-based workflow software system (Appendix 2B) are available in Supplementary File.
4.1.1. Database
The minimum data elements for patients undergoing chemotherapy were identified, and the database was designed based on the chemotherapy process and user activity, such as patient registration, chemotherapy prescription, and administration. The MySQL database was used for the development of backend database in our system. Appendix 2 presents the class diagram and database of the guideline-based workflow software system.
4.2. System Implementation
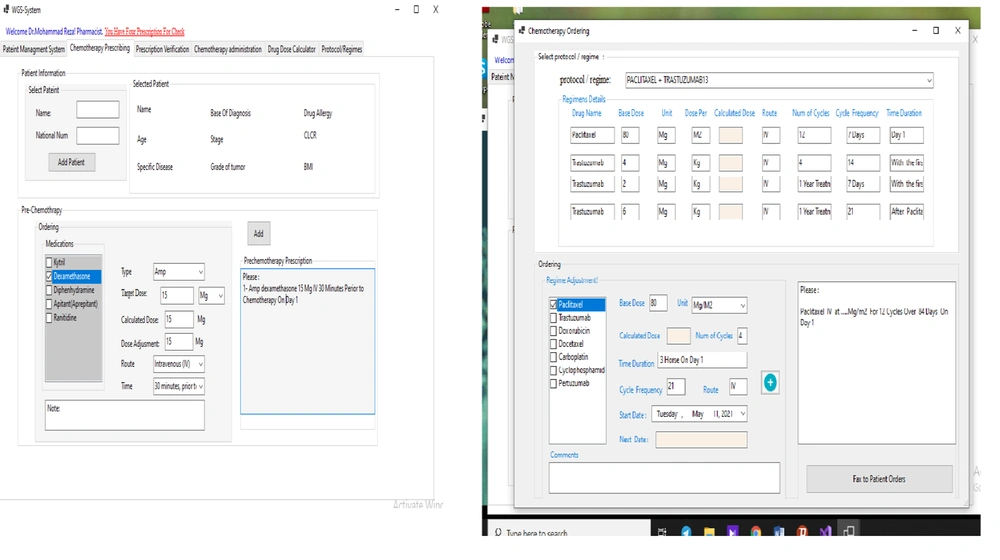
After establishing the system design, our team developed the guideline-based workflow software system. The system was implemented during February and April 2021. The system programing consisted of three types of implementation codes: codes for the user interface implementation, codes for the logic layer implementation, and codes for the database implementation. All drug dose calculations (dose adjustment based on weight, BSA, or the Calvert/Chatelut’s formula) were automatized in each phase by the clinical decision support system, which was integrated in the CPOE system. Our system addressed all chemotherapy processes from prescription orders to administration (prescription and verification of administration) for every anti-cancer medication. Each drug order was secured by several checkpoints and alarms: dose errors; drug-drug interactions; repetitive treatment; exceeding the specific maximum dose or cumulative dose; incorrect values for BSA, weight, or size; and route or cycle errors. Figures 3 - 7 show the screenshots of our developed guideline-based workflow software system.
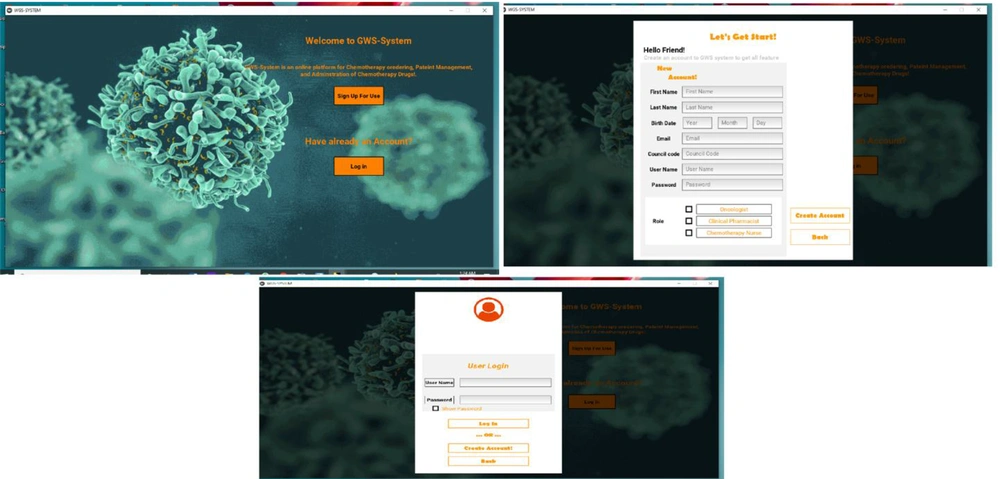
The user interface in our study comprised of 25 pages: (1) welcome page (sign up and log in page); (2) main page, and patient management module (7 pages); (3) chemotherapy prescription interface (3 pages); (4) pharmacist control interface (2 pages); (5) chemotherapy administration interface (3 pages); (6) drug dose calculator interface (4 pages) and update protocol/regimen or guideline interface (1 page). The workflow software system was developed by Python programming language as the front-end language and SQL as the back-end language.
5. Discussion
The main goal of healthcare delivery systems and healthcare providers is to offer patient care in the best and safest possible way. Chemotherapy is a complex, multidisciplinary, and error-prone process. Today, information technology is being increasingly used to enhance the quality and safety of complex processes, especially in a complex environment with complex work procedures, such as healthcare. In this study, we developed a guideline-based workflow software system for improving the chemotherapy process at Taleghani Teaching Hospital in Tehran, Iran.
The advantages of developed system in this study can be explained by: support of the chemotherapy workflow process; potential positive impacts on the safety of chemotherapy process and also on communication and coordination of care between different service providers (oncologists, nurses, and pharmacists); electronic prescription; and directing orders based on standardized protocols.
Transition from paper-based chemotherapy ordering approach to electronic prescribing method by implementation of CPOE system with a clinical decision support system is intrinsically associated with the reduction or prevention of errors through prescription, transcription, and administration of orders. The reason is: all drug dose calculations (dose adjustment based on weight, BSA, or the Calvert/Chatelut formula) were automatized in each phase by the clinical decision support system, which was integrated in the CPOE system. Our system also allowed for use of evidence-based oncology practice and standardization of chemotherapy workflow by directly connecting the prescriptions to the standardized guideline database in the clinical decision support system (CDSS) module.
The GWS system addressed all chemotherapy process tasks from prescribing orders to the administration phase (route, infusion timing, duration, and fluid). It includes all anti-cancer medications, and verification module for use of oncology pharmacist in the prescription validation phase. Each prescribed orders is secured by several alarms: dose error; drug-drug interactions; repetitive treatment; exceeding the specific maximum dose or cumulative dose; inaccurate values of BSA, weight, or size; and route or cycle errors. Oncologists, nurses, and pharmacists had full access to the patient’s profile and treatment history.
Generally, long waiting time and delays in chemotherapy process are associated with heightened dissatisfaction in both healthcare providers and patients. Also, the non-optimal use of the chemotherapy chair capacity reduces the safety of patients. A guideline-based workflow software system can enable the user to have early access to demographic, clinical, and laboratory information; preview the treatment for confirmation; change or conceal each patient treatment program; prepare the chemotherapy drugs before the administration time and check the nursing plan sheet a day before administration, and have a more precise appointment scheduling based on resources. This system allows pharmacists to have more time to control the prescriptions (e.g., drugs, doses, frequency, regimens, drug-drug interactions, and patient allergies). Therefore, long waiting time or delays in the old chemotherapy process workflow, unnecessary steps in the previous chemotherapy workflow, and poor coordination between healthcare providers can be eliminated by this new system.
The use of standard protocols can guarantee the accurate ordering and administration of chemotherapy (36). The implementation of our guideline-based system provide an opportunity for the use of standard protocols in the chemotherapy prescription module, facilitating patient-specific chemotherapy prescriptions by directly connecting the prescriptions to the standard guideline database in the CDSS module, automatically proposing standard drug doses based on BSA, or computing the creatinine clearance (Cr-Cl) based on the Cockcroft-Gault formula.
The GWS system also provided a guideline-based verification module and a special interface for administration of chemotherapy drugs based on the developed guidelines for the optimal control of chemotherapy prescriptions, verification of prescriptions, and onset of the administration phase. Once the clinical pharmacist confirms the prescribed orders in the verification module, an administration checklist, based on chemotherapy nursing protocols, is spontaneously accessible on the nursing interface for the administration of chemotherapy drugs in the chemotherapy nursing unit.
The developed guidelines in the nursing administration interface consist of items for patient identification, confirming the patient’s eligibility for chemotherapy, prescription order check, endorsement of patient satisfaction, confirmation of the route of chemotherapy administration, documentation of administration time, and confirmation of the success or failure of administration phase. Also, the checklist in the administration module prevents the elimination of any steps or sub-steps in the administration phase. From a safety and technological point of view, all guidelines used in the GWS system must be easily adapted to new research results, as the chemotherapy guidelines are subject to continuous change (so is the study of outcomes). Therefore, we developed an updated protocol/regimen interface.
An overview of previous research showed that a limited number of studies had focused on the design and implementation of workflow software systems for the chemotherapy process in cancer care units. The majority of previous studies developed an automatic chemotherapy dose calculation system and a CPOE system for the reduction of medication errors in the chemotherapy prescribing phase. The CPOE and automatic dosage systems in these studies were associated with a significant reduction in the chemotherapy prescription errors, inappropriate dosing, and also adverse drug events (19, 27, 36-45).
Generally, chemotherapy is a complex, multidisciplinary, and error-prone process. Therefore, development of a workflow-based software system is particularly important when the process is multidisciplinary and prone to error; therefore, it is essential to develop a workflow software system for improving the chemotherapy process. This is a secondary report of our pilot study. However, there is a great need to conduct well-designed post-implementation research to study the long-term effects of workflow software systems on the overall safety of the chemotherapy process in cancer care units. Also, in future studies, it is essential to evaluate the usability. Nevertheless, our early findings suggest that this new guideline-based workflow software system could be effective in enhancing the safety and efficacy of the chemotherapy process.
5.1. Conclusion
A patient management system, a clinical pharmacist control system, a nurse administration system, and a protocol-based clinical decision support system were integrated in the CPOE system for improving the chemotherapy process in cancer care settings. Elimination of iterations and unnecessary steps in the old chemotherapy workflow system, enhancement of patient safety, improvement of communication and coordination between healthcare providers, and use of updated evidence-based medicine in direct chemotherapy orders justify the incorporation of these systems in cancer care settings.