1. Introduction
Aneurysm formation of internal carotid arteries (ICA) in patients with mucormycosis is a scarce phenomenon (1-11). Meanwhile, the prevalence of rhino-cerebral mucormycosis has been reported to increase after the Coronavirus disease 2019 (COVID-19) pandemic (12-18). Mucormycosis is an extraordinarily uncommon cause of cerebral aneurysms. Accordingly, diagnostic and therapeutic approaches are also not entirely evidence-based. Hence, there is crucial to publish even case series of this rare association.
Herein, we present three patients with stroke and subarachnoid hemorrhage due to ICA aneurysm after the involvement of adjacent paranasal sinuses (PNS) with mucormycosis. Two of them had a history of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS‑CoV‑2) infection. All patients were recruited from Namazi and Khalili hospitals affiliated with Shiraz University of Medical Sciences in Iran from April 2021 to May 2021. They are high-volume referral centers for stroke and COVID-19 in southern Iran. Table 1 summarizes the clinical and radiological characteristics and outcomes of the patients. This study was approved by the Ethics Committee and Institutional Review Board of Shiraz University of Medical Sciences (IR.SUMS.REC.1400.270). Written informed consent was obtained from the patients or next-of-kin.
| Case No. | 1 | 2 | 3 |
|---|---|---|---|
| Age (y)/Sex | 40/M | 47/M | 54/M |
| Time of disease (month of 2021) | April | April | April |
| Mucormycosis Clinical Syndrome b | Rhinocerebral | Rhinocerebral | Rhinocerebral |
| Confirmatory test for mucormycosis | Histopathology | Histopathology | Histopathology |
| Mucormycosis tempo c | Subacute | Subacute | Subacute |
| Corticosteroid use | Yes (DEX) | Yes (DEX) | Yes (DEX) |
| Radiological features of brain | CT: Extensive SAH. | MRI: Right-sided watershed infarct | MRI: Left MCA territory massive infarction |
| Radiological features of PNS | CT: Opacification of the left sphenoid sinus and the left ethmoidal air cell. MRI: Mucosal thickening of all paranasal sinuses | MRI: Pan sinusitis | CT: Mucosal thickening of the left maxillary sinus and ethmoidal air cells. MRI: Mucosal thickening of the left frontal sinuses and sphenoid sinuses, and ethmoidal air cells. |
| Mucormycosis predisposing factors | DM, cirrhosis | DM | DM, GIST, using imatinib |
| Treatment strategy | FESS, Liposomal amphotericin | FESS, Liposomal amphotericin | FESS, Liposomal amphotericin |
| Aneurysm (site/ size/configuration) | Clinoid part of left ICA/0 × 7 × 11 mm/irregularly- shaped globoid aneurysm dissecting type | Terminal part of right ICA/15 × 10 × 9 mm/ fusiform aneurysm/ dissecting type | MRA: 15 × 8 × 9 mm irregular fusiform aneurysm in the cavernous portion of the left ICA with severe narrowing of supraclinoid part of left ICA just after aneurysm. DSA( two days later): Irregular fusiform aneurysm of cavernous portion of the left ICA/total occlusion of left ICA |
| Significant laboratory data | Glucose:343 mg; Leucocytes: 5%; LDH: 617 u/L; ALT: 66 u/L; AST: 48 u/L; ESR: 32 mm/h; CRP: 85 mg/L; SARS COV-2 RT-PCR: Positive | Glucose:398mg; ALT: 46 u/L; Ferritin: 1468; ESR: 111 mm/h; CRP: 58 mg/L | Glucose:398 mg; Leucocytes: 10.4%; LDH: 1890 u/L; ALT: 59 u/L; ESR: 9 mm/h; SARS COV-2 RT-PCR: Positive |
| Interval between mucormycosis and aneurysm (days) d | 10 | 24 | 19 |
| Endovascular intervention | No/subarachnoid hemorrhage before any intervention | Yes/sacrifice/coiling | No/total occlusion of left ICA before intervention |
| Follow-up duration | 18 days | Four month | 55 days |
| Clinical 667 outcome | Death after 18 days of evolution of mucormycosis | Alive (MRS = 1) | Death after 55 days of evolution of mucormycosis |
Clinical and Radiologic Features and Outcomes of Three Patients with Mycotic Aneurysms and Mucormycosis a
2. Case Presentation
2.1. Patient 1
A 40-year-old male with a past medical history of unexplained thrombocytopenia and smoking was admitted to an outside hospital following a four-day history of cough, fever, and shortness of breath. In the initial evaluation, the real-time reverse transcription-polymerase chain reaction (RT-PCR) of nasopharyngeal and oropharyngeal for SARS-CoV-2 was positive, with blood sugar 343 mg (normal: 74 - 99 mg) and platelet count 43 × 103/mm3. He was treated with intravenous (IV) remdesivir (200 mg IV on day one and 100 mg daily for four days), dexamethasone (8 mg IV daily for seven days), and IV insulin. Abdominal sonography was consistent with liver cirrhosis. On day fourth of admission, he developed headache, binocular diplopia, left eye ptosis, and decreased vision in the left eye. Computed tomography (CT) of the brain was normal, and the PNS CT indicated thickening of the mucosa in the left sphenoid and ethmoidal sinuses. Because of the clinical suspicion of mucormycosis rhinosinusitis, amphotericin B deoxycholate (1 mg/kg/day IV) was added to the treatment regimen. The patient was transferred to our facility eight days after his admission.
On arrival at our center, the patient has normal vital signs but mild tachycardia and tachypnea. He had left eye ptosis, mild proptosis, and swelling associated with a mild chemosis in the left eye and a mild conjunctival injection in the right eye. Complete ophthalmoplegia of the left eye (III, IV, and VI nerve palsy) and partial ophthalmoplegia of the right eye (III and IV nerve palsy) were indicated. Both pupils were dilated and fixed, and there was no light perception on both sides. He had hypoesthesia involving the first and second branches of the left trigeminal nerve (V1, V2). Tenderness was observed on the frontal sinus. No significant findings were found in the oral and nasal cavity assessments.
A chest high-resolution computed tomography (HRCT) revealed an opacity in the upper lobe of the left lung with a central ground-glass opacity compatible with COVID-19 infection. The brain magnetic resonance imaging (MRI) showed no abnormalities in the brain parenchyma but identified mucosal thickening of all PNS, mild proptosis, periorbital edema, and extraconal fat stranding, which was more prominent on the left side. Additionally, the MR venogram showed thrombosis of the left cavernous sinus and prominence of the posterior aspect of the left superior ophthalmic vein. Besides, MR angiography (MRA) showed no ICA aneurysm.
The patient was diagnosed with invasive mucormycosis rhinosinusitis and cavernous sinus thrombosis and treated with vancomycin, meropenem, liposomal amphotericin B (5 mg/kg/day IV), and therapeutic heparin. Abdominal ultrasound and gastrointestinal endoscopy were performed later, which confirmed splenomegaly and esophageal varices. Then, an additional diagnosis of liver cirrhosis was performed. Therefore, albumin was added, and anticoagulation therapy was discontinued.
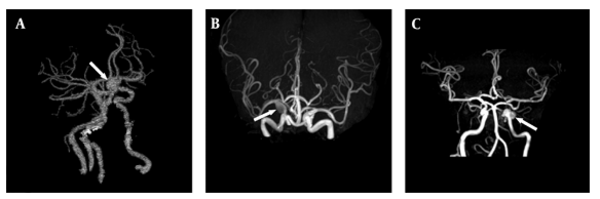
He underwent functional endoscopic sinus surgery (FESS) and debulking on the second day of admission with histological findings compatible with mucormycosis infection. On the sixth day of admission, the patient developed a sudden loss of consciousness. The urgent brain CT showed a disseminated subarachnoid hemorrhage (Fisher Grading scale: 4, Hunt and Hess scale: 5). Brain CT Angiography (CTA) revealed a 10 × 7 × 11 mm irregularly shaped globoid aneurysm in the clinoid portion of the left ICA (Figure 1A). On the eighth day of admission, he developed hypotension, electrocardiographic abnormalities, and a rise of troponins, all consistent with myocardial infarction. The patient passed away on day 10 after admission.
2.2. Patient 2
A 47-year-old male with a six-year history of diabetes mellitus (DM) on metformin and glibenclamide came to our hospital with right eye ptosis and right ocular pain. He complained of body pain and cough 21 days before admission. Four days later, he experienced a severe throbbing headache in the bilateral frontotemporal area associated with nausea and numbness on the right side of his face and palate for 10 days. He was treated with dexamethasone 8 mg IV daily for two days in an outpatient clinic. Three days prior to admission, he developed right eye ptosis, proptosis, blurred vision, and periorbital pain irradiated to the right ear.
On admission, his vital signs were normal. On physical examination, he had right eye ptosis associated with proptosis and severe chemosis (mild on the left eye), with mydriatic and non-reactive pupils. The cranial nerve examination showed a right frozen eye (III, IV, and VI cranial nerves palsy) and right-face hypoesthesia (V1 and V2 branches of trigeminal nerve).
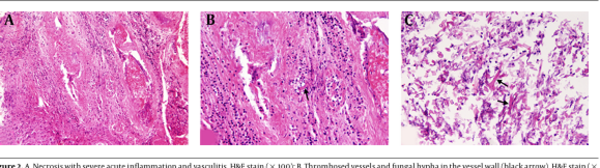
Initial blood sugar was 398 mg (normal: 74 - 99 mg), and the chest HRCT was negative for SARS-CoV-2 infection. The brain MRI did not show any parenchymal abnormalities but confirmed right exophthalmos with extraconal fat stranding and indicated mucosal thickness in all sinuses. The brain MRA revealed no aneurysm. The chest HRCT was normal. The sinus biopsy confirmed mucormycosis (Figure 2). Liposomal amphotericin B 300 mg daily was started, followed by FESS and debulking surgery.
On the 21st day of admission, he experienced right peripheral facial palsy associated with mild left-sided weakness. The brain MRI revealed multiple foci of diffusion restrictions in the right side cortical-subcortical and deep white matter, suggesting anterior and posterior watershed ischemia. The brain MRA showed a fusiform aneurysm measuring approximately 15 × 10 × 9 mm in the terminal portion of the right ICA (Figure 1B). Caspofungin 50 mg IV was added to amphotericin B daily.
The day after, he developed a generalized tonic-clonic seizure. The urgent brain CT confirmed a subarachnoid hemorrhage in the right Sylvian fissure and inferior to the right frontal lobe. The patient underwent digital subtraction angiography three days later, showing a mild increase in the size of the aneurysm.
He had a competent and complete circle of Willis; therefore, a parent artery occlusion was considered. A balloon occlusion test was conducted, occluding the cervical portion of the right ICA for 20 minutes using an 8 Fr Cello balloon catheter (EV3 Endovascular, Inc., Plymouth, USA), developing no neurological deficits during the procedure. As the patient had no change in these examinations, the aneurysm and parent artery coiling was done without complications. The patient was discharged home 45 days after. In the last follow-up, performed four months after onset, he had a normal neurological assessment (modified Rankin scale = 1), but the Visual Acuity (VA) was counting fingers at one meter in the right eye and five meters in the left eye.
2.3. Patient 3
A 54-year-old male with a medical history of a gastrointestinal stromal tumor (GIST) on imatinib and high blood glucose readings (no treatment) was admitted to our facility with a painful oral cavity lesion, nasal hemorrhage, periorbital edema, and binuclear diplopia, as well as left facial paresthesia. He had respiratory symptoms 14 days before and a COVID-19 infection confirmed by PCR, with a blood sugar of 398 mg (normal: 74 - 99 mg). Insulin, remdesivir, and corticosteroids (dexamethasone 8 mg three times a day for two days, followed by 250 mg methylprednisolone pulse daily for six days) were administered in an outside hospital.
On arrival, he was tachypneic, with an oxygen saturation level of 84% in room air. The physical assessment showed left periorbital edema, ptosis, proptosis, and chemosis of the left eye. There was a left sixth nerve palsy associated with left face hypoesthesia (V1 and V2 branches of trigeminal nerve). There was a painful white lesion in the oral cavity and necrotic tissue in the left nasal cavity. The chest HRCT demonstrated bilaterally diffuse ground-glass opacity, and the PNS CT was consistent with mucosal thickening of the left maxillary sinuses and ethmoidal air cells.
A biopsy sample of nasal turbinates confirmed mucormycosis, and the nasal cavity was debrided; consequently, the patient was treated with liposomal amphotericin B (5 mg/kg/day IV). The patient underwent FESS and debulking surgery on the seventh day of admission.
On day nine of admission, he suffered a sudden onset of aphasia, right-sided weakness, and a frozen left eye. The brain MRI demonstrated several foci of left parieto-occipital diffusion restriction suggestive of an acute ischemic infarct. The frontal, ethmoid, and sphenoidal sinuses also exhibited opacification and mucosal thickening. The brain MRA revealed a 15 × 8 × 9 mm irregularly shaped fusiform aneurysm in the cavernous portion of the left ICA with severe narrowing of the supraclinoid portion just after the aneurysm (Figure 1C). Two days later, the patient underwent DSA, which showed complete occlusion of the left ICA. He developed a decreased level of consciousness and left-sided weakness on the 13th day of hospitalization. A second brain MRI indicated several high-intensity T2 FLAIR signals with diffusion restriction in the left temporoparietal lobe and basal ganglia, consistent with acute infarction. The patient's condition deteriorated, and he developed vasogenic edema secondary to the ischemic stroke associated with a 4 mm midline shift. He underwent a hemicraniotomy but did not improve clinically and was deceased 55 days after the initial diagnosis.
3. Discussion
Herein, we presented three patients with mucormycosis and ICA aneurysm. All of them had a history of uncontrolled diabetes and corticosteroid use, and one was treated with a tyrosine kinase inhibitor, imatinib. Two patients were infected with SARS-CoV-2 before developing mucormycosis. There was a rapid progression of mucormycosis vasculopathy to aneurysm formation or complete occlusion. Although all patients had received antifungal treatment and surgical debridement and controlled their diabetes and COVID-19 infection, two out of three died. One could not have endovascular intervention due to unstable conditions, but the other two had diagnostic angiography before endovascular intervention. One patient underwent no therapeutic intervention due to total artery occlusion, whereas the other patient experienced a successful parent artery occlusion by coiling. Interestingly only survived this patient. Patient 2 had a normal brain MRA in the early course of mucormycosis. However, 15 days later, a second brain MRI revealed a sizeable carotid aneurysm. This indicates the rapidly growing nature of mucormycosis-associated internal carotid aneurysms. The configuration of ICA aneurysm in the current series was mostly irregularly shaped and fusiform aneurysm, which is more similar to dissecting aneurysm rather than saccular aneurysm.
Bacterial pathogens cause most infection-associated mycotic aneurysms in the context of endocarditis. An aneurysm develops in the setting of antecedent systemic infections with bacteremia or through the direct local invasion of the vessel wall (e.g., IV drug users) in the pre-existing aneurysm or atheromatous plaques (19). The bacterial infection causes the release of pro-inflammatory cytokines, polymorphonuclear (PMN) leukocyte infiltration, and activation of matrix metalloproteinases, resulting in the focal vessel wall disintegration (20).
Although fungal germs are a relatively uncommon cause of cerebral aneurysms, they can occur in immunocompromised patients due to diabetes, hematological malignancy, systemic chemotherapy, and human immunodeficiency virus (HIV) infection, or fungal dissemination (21). Fungal agents that can cause mycotic aneurysms mainly include Candida species and Aspergillus species (22).
Fungal aneurysms of carotid arteries are extremely rare (9). Fungal aneurysms pose challenges for diagnosis and management because they are rare, unpredictable, and often occur in a clinical context that is neither specific nor alarming. The treatment strategy is controversial owing to the risk of complications associated with surgery on the cavernous sinus. Pathologic investigations demonstrate that fungal aneurysms typically impact the circle of Willis and the proximal arterial tree. They tend to develop and expand, moving long segments of the vascular wall, and they are friable and poorly defined. As a result, endovascular or surgical therapy is challenging, if not impossible, and has a very high mortality rate (23, 24).
An increase in mucormycosis cases has been reported since the outbreak of the COVID-19 pandemic. There are several reports of mycotic associated with COVID-19 infection (12-18) in patients with a history of diabetes and other risk factors. Diabetes (DM), even without diabetic ketoacidosis, is the most critical risk factor for mycotic aneurysms. New studies have also found poorly controlled DM predicts complications such as COVID-19 infection severity and hospitalization. On the other hand, it has been reported that SARS-CoV-1 could result in acute diabetes and diabetic ketoacidosis (25). Besides, high expression of angiotensin-converting enzyme 2 receptors in pancreatic islets, along with increased insulin resistance because of cytokine storm (26), may explain the diabetogenic possibility of SARS-CoV-2 infection. While current guidelines recommend using corticosteroids to treat severe or critical COVID-19, the evidence suggests that the frequent use of steroids exacerbates glucose homeostasis and makes patients susceptible to mucormycosis (27, 28). Hence, in the COVID-19 pandemic, corticosteroids in diabetic patients should be cautiously prescribed. Also, the physicians should be highly suspicious of COVID-19-associated mucormycosis, as the convergence of SARS-CoV-2 infection and uncontrolled DM can cause a mucormycosis storm.
A review of mucormycosis cases associated with an intracranial aneurysm was performed, and it was shown that mucormycosis has a high mortality [11 out of 14 patients (78.6%)] (Table 2). The mean age was 48 years. Aneurysm formation after involving adjacent PNS with mucormycosis occurs in a matter of days. The most common predisposing factors were DM, using steroids, hematopoietic and GI cancer, chemotherapy, and transsphenoidal surgery. The most common aneurysm sites were the internal carotid artery which occurred in about half of the patients. Aneurysm caused complications via rupture and subarachnoid hemorrhage in eight patients and artery to artery embolism and ischemic infarction in four patients. In two patients, aneurysms were found in imaging workups in patients who developed cavernous sinus syndromes. These aneurysms were rapidly growing as they developed between 10 and 68 days after the evolution of mucormycosis.
| Case No. | Age (y)/Sex | Mucormycosis Clinical Syndrome a | Confirmatory Test for Fungal Infection | Mucormycosis Tempo b | Corticosteroid Use (Type) | Radiological Features | Mucormycosis Predisposing Factors | Treatment Strategy | Aneurysm (Site/ Size/Configuration) | Interval Between Mucormycosis and Aneurysm (Days) c | Endovascular or Surgical Intervention | Clinical Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Price et al. (8) | 38/M | Rhino-orbito-cerebral | Histopathology (mucor) | Chronic | No | Sinus radiography: Gross opacification of the right ethmoid and sphenoid sinuses and moderate opacification of the left ethmoid and sphenoid sinuses, bone destruction of the right side of the sphenoid body. DSA: Irregular narrowing of the right ICA over a distance of 1.5 cm. | DM with DKA | Amphotericin B, paranasal surgical debridement | Proximal part of the right ICA /(1.8 cm in diameter)/ ruptured aneurysm | 68 days | NA | Death |
| Glass et al. (4) | 4/ F | Cerebral | Autopsy Phycomycetes (Mucor) | Acute | Yes (NM) | Brain scan using 99TC-pertechnetate: Clearly visualized ventricles, the left lateral ventricle appearing larger. | Prolonged steroid use | NA | Left ACA/ruptured aneurysm | NM | NA | Death |
| Ho (5) | 48 / F | Rhinocerebral | Histopathology (mucor) | Subacute | No | DSA: An ample avascular space on the right side, compatible with subdural hematoma. | Early DM with DKA | penicillin G potassium, chloramphenicol, nystatin Intravenous amphotericin B | ACOM/ 1.0 × 0.8 0.6-cm/ruptured saccular aneurysm | 13 days | Surgical clipping | Death |
| Kikuchi et al. (7) | 61/ M | Cerebral | Autopsy Phycomycetes (Mucor) | Subacute | Yes (HC) | DSA: Four aneurysms originating from the left pericallosal artery and complete disappearance of the right ACA and MCA | Craniotomy, Prolonged steroid use | NA | Left pericallosal artery/ four unruptured aneurysms | 30 day | NA | Death 33days |
| Thajeb et al. (11) | 62/M | Rhino-orbito cerebral | Histopathology (mucor) | Subacute | No | MRI: Lesions in the left orbital apex and the inferior part of the left cavernous sinus; CT: Severe SAH with hydrocephalus and cerebral infarctions in the left frontal lobe, left pontomesencephalon, left cerebellum, and bilateral thalami. | DM, spontaneous infection of the left orbital. Apex and cavernous sinus | Amphotericin B, paranasal surgical debridement | Presumed Ruptured aneurysm (SAH) | NM | NA | Death |
| Kasliwal et al. (6) | 61/M | Cerebral | Histopathology (mucor) | NM | NO | MRI: Postoperative changes with a small amount of residual tumor and a left basal ganglia infarct. CT: SAH. DSA: Bilateral, almost mirror image-like, fusiform aneurysms of the right ACA. | DM, non-functional pituitary macroadenoma, transsphenoidal surgery, prolonged administration of antibiotics | Amphotericin B, liposomal amphotericin, Sur paranasal surgical debridement | Bilateral ACA /7.45 × 6.9 mm/ Ruptured fusiform aneurysms (right: With the neck of 6.9 diameter left with no obvious neck) | NM | Surgical clipping | Death |
| Alvernia et al. (1) | 38 /M | Rhino cerebral | Histopathology (mucor) | Subacute | No | CT: Inflammatory process involving the paranasal sinuses with extension into the left cavernous sinus and left petrous bone. DSA: 50% stenosis of the left ICA at its petrous portion and a bilobulate pseudoaneurysm originated at the same level. | DM | Topical clotrimazole, parenteral liposomal amphotericin B, hyperbaric oxygen, paranasal surgical debridement, atorvastatin | Petrous; segment of the left ICA/ unruptured bilobulate pseudoaneurysm | NM | Successful endovascular coiling | Alive |
| Dusart et al. (3) | 64/M | Rhinocerebral | Autopsy(mucor) | Chronic | Yes (HC) | MRI: An extensive sphenoid sinusopathy, a massive fusiform aneurysmal dilatation of the right intracavernous ICA, a suprasellar extension of the mass, spontaneous thrombosis, right thalamic infarction. T: Inflammatory-induced bone modifications, bone defects between sphenoid and cavernous sinuses | Somatotropic macroadenoma (treated by transsphenoidal surgery and radiotherapy 21 years ago) | No effective treatment | Right intracavernous ICA/ uge fusiform aneurysm | 30 days | No | Death |
| Azar M et al. (2) | 71/F | Rhinocerebral | Histopathology (mucor) | Subacute | No | CT: Extensive right frontal, sphenoid, ethmoid, and maxillary sinusitis with extraosseous spread into the orbital area and pterygopalatine fossa, and possibly a cavernous sinus thrombosis. MRI: Enlargement of the signal void at the distal cavernous segment of the right ICA. | DM, AML, using chemotherapy | Liposomal amphotericin B, meropenem, vancomycin, voriconazole, moxifloxacin paranasal surgical debridement | Cavernous part of the right ICA/1.2 × 0.8 cm/ bilobed aneurysm | NM | Yes/sacrifice (coil embolization of the aneurysm) | Alive |
| Sasannejad et al. (10) | 57/M | Rhinocerebral | Histopathology (mucor) | subacute | NO | CT: Extensive SAH. CTA: Two consecutive fusiform aneurysms in an SCA. MRI: Infarction of the cerebellum in the territory superior cerebellar artery. | DM | Amphotericin-B, wide spectrum antibiotics, Nasal cavity debridement | SCA / 5.17 × 5.50 mm and 4.17 × 5.55 mm/ ruptured fusiform aneurysms | 21 days | NA | Death |
| Rangwala et al. (9) | 27/F | primary pulmonary mucormycosis developed with cerebral mucormycosis | Histopathology (mucor) | Subacute | Yes (NM) | CT & CTA: Intraparenchymal hemorrhage of the left temporoparietal lobe measuring 2.6 × 2.6 × 3.7 cm, with an underlying multilobulated aneurysm of the distal left MCA. | Systemic lupus, erythematous, using steroid | Amphotericin B | Distal of left MCA/ ruptured fusiform mycotic aneurysm | NM | Microsurgical aneurysm excision | Death |
An Overview of Clinical Features and Outcomes of Published Cases Diagnosed with Mycotic Aneurysms and Mucormycosis
The absence of a fungal culture should be mentioned as a major limitation of the current study.
In conclusion, in patients with rhino-cerebral mucormycosis, aneurysm evolution should be promptly and meticulously investigated by MRA and CTA. Unlike other types of mycotic aneurysms, these aneurysms occur in more proximal portions, and their configuration resembles dissecting aneurysms rather than saccular ones. As this type of aneurysm is very fast-growing, as soon as the involvement of the sphenoid sinus is detected, the possibility of ICA aneurysm formation should always be kept in mind. If the patient develops an aneurysm, prompt intensive antifungal therapy and therapeutic endovascular interventions such as stenting, coiling, or sacrificing should be considered as soon as possible to optimize outcomes.