1. Background
Diabetes mellitus is a group of metabolic diseases with an increasing incidence throughout the world. It is known as one of the leading causes of mortalities and underlying conditions for cardiovascular diseases (1). Despite the growing success of conventional medicine, many diseases, especially chronic disorders, have been left untreated or inadequately treated and are sometimes associated with complications in the involved patients (2). This led some patients to take advantage of a variety of complementary and alternative therapies in order to be treated (3).
Recently, special attention has been paid to “personalized medicine” within the framework of conventional medicine. This branch of medicine aims to achieve an appropriate treatment for each person individually (4). Researchers in this field believe that treatment methods should focus on the hosts of diseases instead of the diseases themselves. Personalized medicine tries to establish a health/therapeutic model to enter the psychological, environmental, mental, and biological aspects of treatments for patients and help them maintain their health (5).
TPM is a traditional medical system highly used by patients with diabetes in Iran. TPM, like other traditional medical approaches, applies a specific type of diagnostic model for patients’ individualization, known as syndrome differentiation in the literature (6). The syndromes in TPM are defined based on a concept which is called Mizaj.
Mizaj is a phenomenon created by the interactions of various elements in the body. Mizaj affects the phenotypic characteristics and physiological functions of the body (7, 8). Each person has a special Mizaj, which is evaluated based on the two spectra of warm/cold and wet/dry qualities (7). According to the theory of Mizaj, any treatments should be selected based on the Mizaj of the patient and the disease, using a drug with reverse Mizaj (hot drugs for cold diseases, dry drugs for wet diseases, and vice versa) (9). Therefore, different treatments may be considered for the same disease (such as diabetes) based on the patient’s Mizaj (10).
Cinnamon (Cinnamomum verum) is a popular medicinal plant mostly investigated in the field of diabetes. Cinnamon induces auto-phosphorylation and de-phosphorylation of the insulin receptors and synthesis of the glucose transporter GLUT4 receptors. Cinnamon also alters hepatic glucose metabolism through modulation in phosphoenolpyruvate carboxykinase and pyruvate kinase. Inhibition of intestinal glucosidases is another suggested mechanism responsible for the anti-glycaemic effect of cinnamon. Cinnamon’s anti-diabetic effects have been evaluated through numerous human and animal studies, leading to conflicting results (11-15). According to the TPM syndrome differentiation model, cinnamon has a hot and dry Mizaj and is more effective on diabetic patients with cold and wet Mizaj.
2. Objectives
Accordingly, this study was designed to evaluate the effect of applying the syndrome differentiation model based on TPM to identify the patients who benefit more from cinnamon supplementation.
3. Methods
3.1. Study Setting
This study was a randomized, triple-blinded, placebo-controlled clinical trial. All the T2DM patients were invited to the Diabetes Research Center, Yazd University of Medical Sciences, Iran, from April to October 2017. After the research method was explained to the participants, all of them signed an informed consent form. Afterwards, they were selected based on the eligibility criteria. The patients’ demographic and clinical features were recorded. Moreover, their physical activities and dietary intakes were registered according to the International Physical Activity Questionnaire and Food Frequency questionnaire, respectively. Their baseline blood samples were taken in the laboratory.
The study was approved with the reference number: IR.SUMS.REC.1395.176 by the Local Medical Ethics Committee of Shiraz University of Medical Sciences. Registration of the trial protocol with the registration ID: IRCT2017031133015N1 was carried out in the Iranian Registry Database of Clinical Trials.
3.2. Inclusion and Exclusion Criteria
This study was performed on both male and female patients, aged 30 - 80 years, and diagnosed with T2DM by an endocrinologist based on the American Diabetes Association (ADA) criteria and a fasting blood glucose (FBG) level of 126 - 250 mg/dL. Those who consumed only oral hypoglycemic agents for their diabetes, and did not have any uncontrolled co-morbid diseases, were included in this study. The patients who were reporting a change of treatment during the study, having any allergic reactions to cinnamon, consuming less than 80% of the prescribed capsules and women with pregnancy or lactation were excluded from the study. No limitation on the duration of diabetes was applied for patient inclusion.
3.3. TPM Syndrome (Mizaj) Evaluation
All participants completed the 10-item Mizaj Questionnaires of Mojahedi et al. (10) based on a 3-point Likert scale (Table 1). The questions were asked by the researcher, and the results were recorded in the pre-designed forms. The researcher tried to equalize the perceptions of the items. The first (1-8) and the second (9, 10) domains of the questions were related to the warmness and wetness dimensions of Mizaj, respectively. In the mentioned domains, the total scores of ≤ 14, 15 - 18, and ≥ 19 indicated cold, moderate, and warm Mizaj conditions, and those of ≤ 3, 4, and ≥ 5 were considered as wet, moderate, and dry Mizaj states, respectively (10).
| Question | 1 | 2 | 3 | |
|---|---|---|---|---|
| 1 | When others touch your skin, what do they say about its warmness or coldness? | Cold | Not cold, not warm | Very warm |
| 2 | How big is the palm of your hand? | Small | Not small, not big | Big |
| 3 | How fast are you influenced by warmness and coldness? | I feel cold, fast. | I feel the same in both cases. | I feel warm, fast. |
| 4 | How do you pronounce several consequent sentences? | Articulate | Not articulate, not continuous | Continuous |
| 5 | How is your rage and anger? | I get angry late | I get angry no late no fast | I get angry fast |
| 6 | How fast are you influenced by warm nature foods like honey, spices, pepper or cold nature foods like buttermilk, yogurt and cucumber? | I feel cold, fast by cold nature foods | I feel the same in both cases. | I feel warm, fast by warm nature foods. |
| 7 | How is your voice power compared to others? | Weak | Not weak, not strong | Strong |
| 8 | How is your physical movements compared to others? | Very slow | Not slow, not fast | Fast |
| 9 | Are you fat or thin compared to others? | Very fat | Not fat, not thin | Very thin |
| 10 | How is the condition of your skin’s softness and coarseness? | Very soft | Not soft, not coarse | very coarse |
aThe score of the hot-cold scale could be 8 to 24. Hot ≥ 19, cold ≤ 14.
bThe score of the wet-dry scale could be 2 to 6. Dry ≥ 5, wet ≤ 3.
3.4. Drugs
Cinnamon bark was purchased from a store in Shiraz, and then was confirmed by a herbalist (Mrs. Khademian), and a voucher sample (PM-996) was deposited in the herbarium of Shiraz pharmacy school. After being rinsed, dried, and powdered, it was filled into 500 mg capsules. The placebo capsules with a similar amount of heated starch (turned into brown color) were prepared. The bottles of packaged cinnamon and placebo capsules were numbered and then were selected randomly. The cinnamon essential oil was extracted and analyzed by Gas Chromatography/Mass Spectrometry (GC/MS).
3.5. Intervention
The capsules were administered to the patients twice a day (after fasting overnight in the morning and before going to bed at night) for three months. During the first month, the supplements were delivered to the patients, and through a follow-up phone call every two weeks, they were asked whether they were taking the drugs regularly and if they were experiencing side effects. After the first month, only the patients who had used more than 80% of the capsules were treated using the same procedure for another two months; the rest of the patients were excluded from the study. Upon completing the three months of treatment, the patients’ biochemical markers were re-assessed.
3.6. Outcome Measures
Measurements of the biochemical outcomes were conducted at the admission and the end of the three-month intervention, in accordance with the following procedures: the patients’ FBG levels were measured by taking 5 ml of their blood after 12 hours of fasting by glucose peroxidase method using an assay kit (Pars Azmoon Company, Tehran, Iran) and an auto-analyzer device (Echo Plus Inc Co, Italy). Glycated hemoglobin (HbA1C) was measured after its primary separation via the ion-exchange chromatographic method (biosystems). Insulin level and resistance were determined using ELISA kits (Diametra Corporation, Milan, Italy) and Homeostasis Model Assessment of Insulin Resistance (HOMA-IR) through the equation of [FBG (mg/dL) × serum insulin (mU/mL)]/405, respectively (16, 17).
3.7. Randomization and Blinding
This study was a randomized, triple-blinded, placebo-controlled clinical trial. Computer-generated block randomization lists using a 1:1 allocation ratio and block size of 4 were used. An independent person, who did not have any contacts with the patients, filled the bottles of the supplement and placebo capsules, labelled as the categories of A and B, the contents of which were not revealed for any of the patients or the responsible physician and the person allocating the groups. The patients could receive any randomized bottle decoded from the researcher according to the randomization table obtained from the original form of the randomization results.
Finally, a statistician analyzed the results obtained from the cinnamon (A) and placebo (B) groups.
3.8. Sample Size Calculation and Statistical Analyses
A sample size of 120 patients was estimated for a one-sided testing at a 0.05 significance level with a power of 0.80. Given the probability of 15% for the dropped-out patients, a total of 140 patients were enrolled in this study. The patients’ demographics and clinical features were represented as mean ± SD or percentage where appropriate. Chi-square test and ANOVA were applied in the statistical analyses using the Statistical Package for the Social Sciences, version 18.0 (SPSS Inc., Chicago, IL, USA). A P value of < 0.05 was considered significant.
4. Results
4.1. Drug Analysis
Gas chromatography/mass spectrometry (GC/MS) analysis revealed that cinnamaldehyde and 4-methoxycinnamaldehyde (3.26%), α-copaene (0.86%), δ-Cadinene (1.61%) and α-Muurolene (0.68%) are the major constituents of cinnamon bark essential oil, respectively.
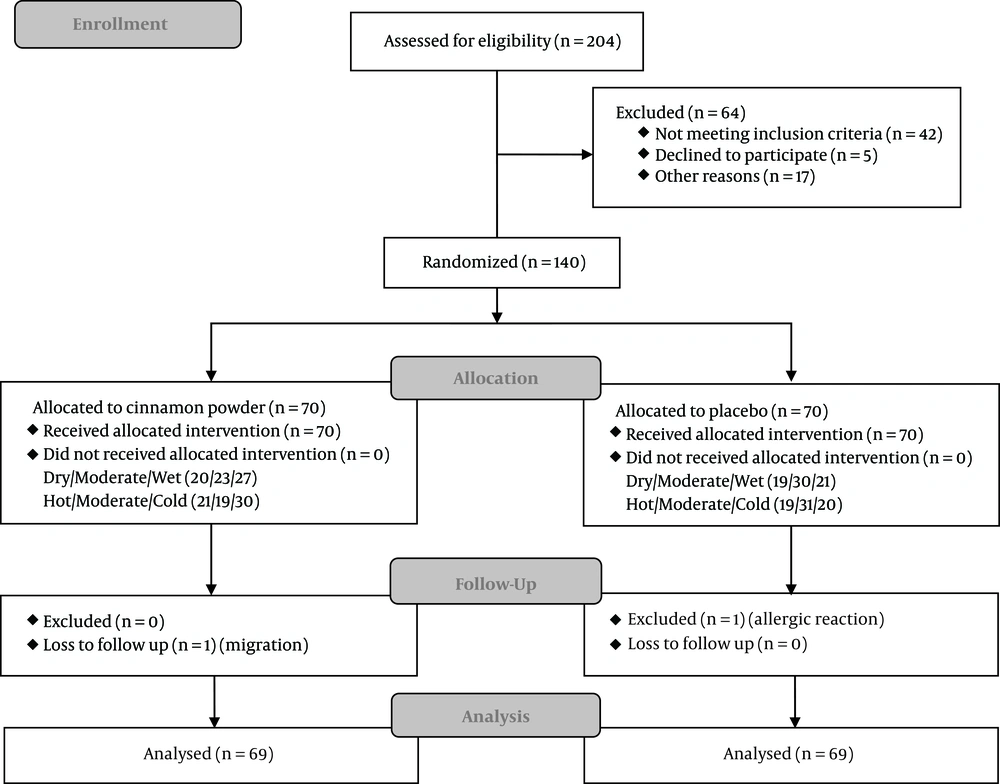
4.2. Study Flow
140 patients out of 204 T2DM patients, who had agreed to take part in this study, were randomly assigned to the cinnamon and placebo groups during April to October 2017. According to the Mizaj distribution in each group, patients were categorized into six subgroups (wet, moderate, dry, cold, moderate, and hot). One patient in the cinnamon group was omitted due to migration (loss of follow-up). Another patient in the placebo group was also excluded due to self-reporting an allergic reaction which led to discontinuation of the medication. 138 patients were included in the data analysis (Figure 1).
4.3. Baseline Characteristics
As shown in Table 2, there was no statistically significant difference between the patients’ demographic and clinical features in the two groups. No significant difference in Mizaj distribution was present in two groups (Table 3).
| Characteristics | Placebo | Cinnamon | P Valueb |
|---|---|---|---|
| Age, y | 53.20 ± 8.5 | 52.17 ± 9.7 | 0.51 |
| Gender (female/male) | 26/43 | 37/32 | 0.06 |
| Physical activity | 1.10 ± 0.30 | 1.13 ± 0.33 | 0.59 |
| Duration of diabetes, y | 5.27 ± 3.8 | 5.66 ± 4.1 | 0.56 |
| Calorie intake, Kcal/d | 501.92 ± 118.25 | 2471.82 ± 316.85 | 0.46 |
| Weight, kg | 74.37 ± 11.6 | 75.72 ± 11.6 | 0.49 |
| BMI, kg /m2 | 29.33 ± 17.11 | 29.92 ± 12.3 | 0.81 |
| HbA1C, mg/dL | 7.43 ± 0.82 | 7.36 ± 0.71 | 0.59 |
| FBS, mg/dL | 159.39 ± 35.3 | 162.57 ± 30.06 | 0.56 |
| 2hpp, mg/dL | 208.21 ± 47.8 | 210.47 ± 44.4 | 0.77 |
| Insulin, mg/dL | 11.46 ± 4.50 | 10.64 ± 4.26 | 0.27 |
| Insulin resistance (HOMA-IR) | 4.51 ± 2.13 | 4.24 ± 1.79 | 0.42 |
Abbreviations: BMI, body mass index; FBS, fasting blood glucose; 2hpp, two hour postprandial blood glucose; HOMA-IR, Homeostatic Model Assessment of Insulin Resistance.
aValues are expressed as mean ± SE.
bChi-square test for categorical variable and t-test for quantitative variable.
| Mizaj | Cinnamon | Placebo | P Valuea |
|---|---|---|---|
| Cold | 29 | 19 | 0.079 |
| Moderate (coldness/warmness) | 19 | 31 | |
| Warm | 21 | 19 | |
| Wet | 27 | 21 | 0.427 |
| Moderate (wetness/dryness) | 23 | 30 | |
| Dry | 19 | 18 |
aNo significant.
4.4. Glycemic Outcomes
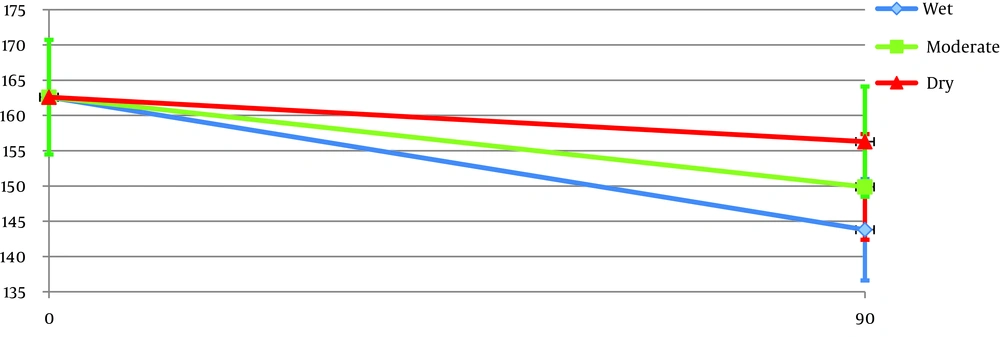
Patients in the cinnamon group showed significantly higher improvement of the mean glycemic indices (FBS, HbA1c), insulin levels, and resistance (HOMA index) compared with the placebo group (Table 4). All the outcomes were improved in patients with cold and wet syndrome more than the hot and dry ones (Tables 4 and 5, Figure 2). This difference was statistically significant in patients with wet compared to dry syndrome.
| Characteristics | Mizaj | Cinnamon (N = 27/23/19)b | Placebo (N = 21/30/18)b | Mean Difference | P Valuec |
|---|---|---|---|---|---|
| Fasting blood glucose, mg/dL | Wet | -18.18 ± 2.69 | 0.76 ± 1.18 | 18.94 ± 3.4 | < 0.001 |
| Moderate | -12.73 ± 3.19 | -2.23 ± 1.5 | 10.50 ± 3.2 | 0.002 | |
| Dry | -6.31 ± 2.26 | -3.6 ± 0.56 | 2.70 ± 2.7 | 0.328 | |
| Pd | 0.016 | 0.201 | |||
| Total | -13.1 ± 1.7 | -1.7 ± 1.9 | < 0.001 | ||
| Glycosylated hemoglobin (HbA1c), % | Wet | -0.366 ± 0.03 | 0.05 ± 0.017 | 0.42 ± 0.04 | < 0.001 |
| Moderate | -0.33 ± 0.09 | 0.013 ± 0.015 | 0.43 ± 0.100 | < 0.001 | |
| Dry | -0.063 ± 0.053 | -0.088 ± 0.056 | -0.025 ± 0.06 | 0.688 | |
| P | 0.003 | 0.012 | |||
| Total | -0.27 ± 0.04 | 0.001 ± 0.02 | < 0.001 | ||
| Insulin, mIU/L | Wet | -2.19 ± 0.21 | -0.003 ± 0.04 | 2.18 ± 0.22 | < 0.001 |
| Moderate | -2.06 ± 0.54 | -0.006 ± 0.02 | 2.05 ± 1.54 | 0.001 | |
| Dry | -0.85 ± 0.16 | 0.03 ± 0.05 | 0.892 ± 0.18 | < 0.001 | |
| P | 0.026 | 0.709 | |||
| Total | -1.77 ± 0.41 | 0.005 ± 0.02 | < 0.001 | ||
| HOMA-IR | Wet | -1.35 ± 0.14 | -0.008 ± 0.05 | 1.34 ± 0.16 | < 0.001 |
| Moderate | -1.04 ± 0.24 | -0.020 ± 0.05 | 1.02 ± 0.22 | < 0.001 | |
| Dry | -0.50 ± 0.10 | -0.070 ± 0.04 | 0.43 ± 0.11 | 0.001 | |
| P | 0.006 | 0.766 | |||
| Total | -1.01 ± 0.11 | -0.03 ± 0.03 | < 0.001 |
aValues are expressed as mean ± SE.
bN (Cinnamon): wet = 27, moderate = 23, dry = 19/N (placebo): wet = 21, moderate = 30, dry = 18.
cPost Hoc tests (multiple comparisons).
dANOVA (between groups).
| Characteristics | Mizaj | Cinnamon (N = 29/19/21)b | Placebo (N = 19/31/19)b | Mean Difference | P Valuec |
|---|---|---|---|---|---|
| Fasting blood glucose, mg/dL | Cold | -14.06 ± 3.37 | 2.00 ± 1.34 | 16.06 ± 4.3 | 0.001 |
| Moderate | -14 ± 2.4 | -3.12 ± 0.37 | 10.87 ± 2.5 | < 0.001 | |
| Warm | -10.9 ± 2.18 | -3.00 ± 2.1 | 7.95 ± 3.04 | 0.013 | |
| Pd | 0.708 | 0.056 | |||
| Total | -13.1 ± 1.7 | -1.7 ± 1.9 | < 0.001 | ||
| Glycosylated hemoglobin (HbA1c), % | Cold | -0.32 ± 0.08 | 0.05 ± 0.025 | 0.38 ± 0.106 | 0.001 |
| Moderate | -0.31 ± 0.04 | 0.019 ± 0.018 | 0.29 ± 0.03 | < 0.001 | |
| Warm | -0.15 ± 0.03 | -0.021 ± 0.05 | 0.13 ± 0.06 | 0.042 | |
| P | 0.154 | 0.012 | |||
| Total | -0.27 ± 0.04 | 0.001 ± 0.02 | < 0.001 | ||
| Insulin, mIU/L | Cold | -2.24 ± 0.41 | -0.03 ± 0.03 | 2.20 ± 0.51 | < 0.001 |
| Moderate | -1.53 ± 0.32 | -0.005 ± 0.03 | 1.52 ± 0.32 | < 0.001 | |
| Warm | -1.36 ± 0.27 | 0.06 ± 0.04 | 1.42 ± 0.28 | < 0.001 | |
| P | 0.177 | 0.224 | |||
| Total | -1.77 ± 0.41 | 0.005 ± 0.02 | < 0.001 | ||
| HOMA-IR | Cold | -1.23 ± 0.19 | 0.05 ± 0.044 | 1.29 ± 0.20 | < 0.001 |
| Moderate | -1.91 ± 0.17 | -0.06 ± 0.045 | 1.84 ± 0.14 | < 0.001 | |
| Warm | -0.80 ± 0.17 | -0.052 ± 0.084 | 0.75 ± 0.19 | < 0.001 | |
| P | 0.220 | 0.287 | |||
| Total | -1.01 ± 0.11 | -0.03 ± 0.03 | < 0.001 |
aValues are expressed as mean ± SE.
bN (Cinnamon): cold = 29, moderate = 19, warm = 21/N (placebo): cold = 19, moderate = 31, warm = 19.
cPost Hoc tests (multiple comparisons).
dANOVA (between groups).
However, the patients’ glycemic outcomes, except 2hpp, showed a significant improvement regardless of the coldness and warmness of Mizaj (Table 5).
No adverse effects were reported by any of the patients in the cinnamon group. An allergic reaction to the placebo was reported by one patient of dry Mizaj syndrome, leading to the discontinuation of the intervention. After one week, his allergic symptoms disappeared with no need for further intervention. The patients reported no other adverse effects.
5. Discussion
In this study, supplementation with a one-gram daily dose of cinnamon for three months led to improved glycemic indices (FPG and HbA1C) and insulin resistance compared with placebo. All the outcomes were more enhanced in patients with wet syndrome compared with the dry ones. These findings were compatible with Persian syndrome differentiation.
The improvements of the diabetic patients’ glycemic outcomes were compatible with those observed in the previous studies in terms of reducing FBS (12, 18, 19), 2hpp (20, 21), HbA1c (11, 22), and IR (23, 24) and increasing insulin secretion (22). In contrast, some other studies did not report similar results for diabetic patients’ glycemic outcomes (12, 15, 25, 26). Therefore, this study tried to address this discrepancy by differentiating patients responsive to cinnamon supplementation based on the traditional Persian medicine syndrome differentiation method. The findings show that cinnamon supplementation in patients with a wet Mizaj may be more effective on improvement of the glycemic parameters. To the best of our knowledge, the effects of Mizaj, as a predictor of diabetic patients’ responses to supplementation of cinnamon, have not been investigated previously.
Applying syndrome differentiation model based on TPM to identify the patients who will benefit more from cinnamon supplementation was reported in multiple previous studies. Kamali et al. (27) investigated the effects of pomegranate extract on patients with ulcerative colitis. They reported significantly better outcomes in patients with hot syndrome compared with cold syndrome. Another study by Niazi et al. (28) assessed the effects of topical rose oil on migraine and reported that the oil leads to more short-term pain relief in hot type patients compared with the cold ones. TPM-based syndrome differentiation also successfully differentiated patients responding to the lipid-lowering effects of Sumac in another study carried out by Hajmohammadi et al. (29).
No previous study was found on syndrome differentiation based on traditional Persian medicine in patients with diabetes. However Chinese medicine based syndrome differentiation theories such as QI stagnation due to liver depression, liver and stomach heat stagnation, phlegm and heat stasis, excess heat in the stomach and intestine, intestinal damp and heat syndromes, and dual deficiency of Qi and Yin are previously used to describe differences in patients with diabetes (30).
The most important limitation of this study was the instrument used for Mizaj evaluation. Mojahedi questionnaire, which was used to differentiate patients with different Mizaj in our study, has been used and validated in multiple studies. However, it has not been previously approved in patients with diabetes.
5.1. Conclusions
The results of the current study, along with the previous studies applying the syndrome differentiation model of TPM, showed the potential of this theory to differentiate patients responding to herbal supplements. This personalized approach can especially be beneficial in the explanation of the discrepancies in results of clinical studies on specific herbal drugs for a particular disease, for instance, cinnamon for diabetes mellitus.