1. Background
Acute respiratory infection (ARI) is one of the most deadly infectious diseases in children worldwide (in both developed and developing countries), causing discomfort, frequent healthcare visits, and deaths (1). It is estimated that between 1.6 and 2.2 million children die each year from ARI (2). The mortality of ARI varies in different regions of the world (3) and accounts for up to 50% of visits of children to health facilities globally (4). Several socio-cultural, demographic, and environmental risk factors, such as female sex, age, comorbid diseases, nutrition, low-income status, maternal lower age, maternal lower education, place of residence (urban or rural), and wet season, predispose children younger than 5 years to ARI (5). Approaches to control ARIs according to the pneumonia severity usually include 4 basic classes: immunization against specific pathogens, early detection, and therapy of disease, enhancements in nutrition, and appropriate environments (6). Assessments of the World Health Organization (WHO) indicate that improvements in nutrition may decrease the risk of ARI incidence or mortality in children of developing countries (7). Therefore, any nutritional interventions can ameliorate child survival from infectious respiratory.
Glutamine (Gln) is the most plentiful free amino acid in plasma and tissue, synthesized in the lungs, liver, brain, skeletal muscles, and adipose tissue and secreted into the circulation (8). Most consumers of Gln are the small intestine, leukocytes, liver, and kidneys (9). Gln has important and regulatory functions in metabolism (as the lipogenic and glucogenic precursor and oxidative energy), protein synthesis and degradation, cell survival and growth, and expression of genes associated with metabolism. Moreover, Gln, as a precursor of glutathione and attenuation of pro-inflammatory cytokines, has a vital role in the antioxidant and anti-inflammatory defense of the body (10-14). As a conditionally essential amino acid, Gln is an essential element in the proliferation and function of immune cells; hence, Gln deficiency may have an intense impact on the immune system and may elevate the risk of respiratory infections. In catabolic conditions, Gln levels fall below normal, mainly in the muscle and liver (15, 16). Endogenous Gln synthesis does not provide the human body’s needs in catabolic conditions, including severe and long-term physical exercise, trauma, cancer, surgeries, sepsis, and infections. Under physiological conditions, Gln is efficiently synthesized in the liver and skeletal system. However, under catabolic situations and oxidative stress, concentrations of Gln in tissues decrease swiftly to assist the further demands of the body, resulting in energy metabolism disruptions and a weakened immune system (16). These disturbances can be lessened by supplementation with Gln; accordingly, it is currently a component of clinical nutrition supplementation practices and/or introduced for patients with immune suppression (17).
Given the increase of oxidative stress (18-20) and inflammatory cytokines in respiratory diseases (21, 22) and the high prevalence of ARI in children, we sought for the first time to investigate the effects of Gln supplementation on serum levels of tumor necrosis factor α (TNF-α), interleukin 1 beta (IL-1β), high-sensitivity C-reactive protein (hs-CRP), malondialdehyde (MDA), and total antioxidant capacity (TAC) in hospitalized children with ARI.
2. Methods
2.1. Study Design
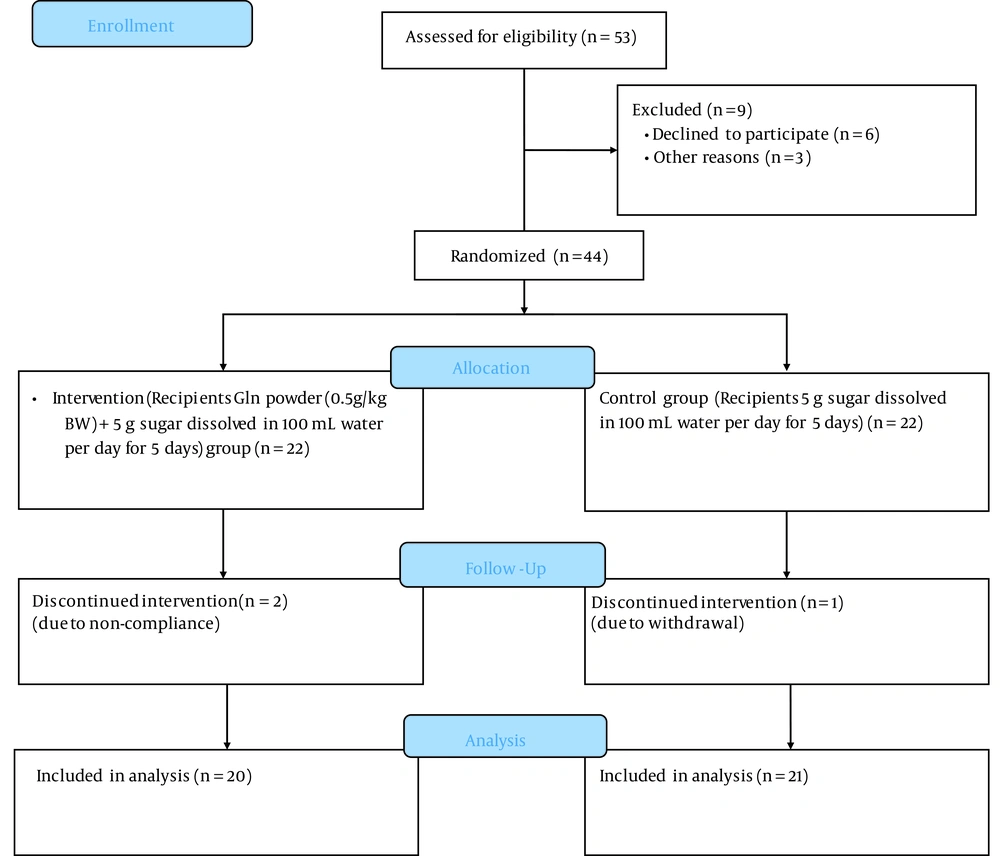
We conducted a 5-day parallel-group, randomized controlled trial. This clinical trial was carried out at the Bu Ali Hospital, Ardabil University of Medical Sciences, for 5 days to determine the efficacy of the 0.5 g/kg body weight Gln, along with medical therapy, in hospitalized children with ARI (Figure 1).
2.2. Ethics and Trial Registration
The study protocol was approved by the Ethics Committee of Ardabil University of Medical Sciences (code: IR.ARUMS.REC.1398.122), and it was conducted in accordance with the Helsinki Declaration. This clinical trial was registered on the Iranian Registry of Clinical Trials website (registration number: IRCT20210913052455N1).
2.3. Participants and Intervention
By referring to the Bu Ali Hospital, Ardabil, Iran, 44 children with ARI who met the inclusion criteria were recruited in this trial, and written informed consent was obtained from their parents (May 2020 to July 2020). Inclusion criteria were age between 2 to 6 years, having ARI, and not having any chronic disease such as congenital heart diseases, chronic liver or renal diseases, immune deficiency, or malignancy. Exclusion criteria were refusal to continue and consumption of less than 10% of total administered powders. The diagnosis of ARI and the general management of ARI were made by pediatric physicians. The sample size was determined based on serum MDA as the primary outcome obtained from our previous study (23). The sample size was computed using the following formula:
2.4. Assessment of Anthropometric Parameters
A trained dietitian measured anthropometric variables, including body weight, height, and body mass index (BMI), after overnight fasting (27). Body weight was measured by a Seca scale with an accuracy of 100 g at the beginning and end of the trial. Height was assessed in a relaxed position using a Seca portable stadiometer with an accuracy of 0.5 cm. BMI was calculated as body weight in kilograms divided by the square of height in meters at baseline and the end of the study (28).
2.5. Assessment of Appetite
Appetite was evaluated by the Council on Nutrition Appetite Questionnaire (CNAQ), which was adapted by Wilson et al. (29). CNAQ contains 8 items, and the scales of each item range from 1 to 5. Thus, the range of the total score is 8 to 40 points. A score of less than 28 is a cause for concern. The validity of CNAQ has been confirmed in the Iranian population (Cronbach α = 0.77) (30).
2.6. Assessment of Biochemistry Variables
Before and after the trial, venous blood samples (2 mL) were collected after 10-12 hours of overnight fasting. Enzyme-linked immunosorbent assay (ELISA) kits (Zell Bio GmbH) were used to determine serum levels of IL-1β, TNF-α, MDA, and TAC. Moreover, an ELISA kit (Pars Azmoun Co, Karaj, Iran) was applied to determine serum levels of hs-CRP (31).
2.7. Statistical Analysis
The data analyst was blinded after assignment to interventions. Statistical analysis was performed using SPSS version 23 (SPSS Inc, Chicago, Ill, USA). P values less than 0.05 were considered statistically significant. The Kolmogorov-Smirnov test was used to assess the normal distribution of the data. A chi-square test was applied to compare the categorical variables between the 2 groups at the baseline. The independent samples t test and Mann-Whitney U test were used to compare differences in parametric continuous and nonparametric data between the 2 groups, respectively. To compare within-group changes in variables, the paired-samples t test or Wilcoxon signed-rank test were used. To control confounding variables (TNF-α, IL-1β, hs-CRP, MDA, and TAC baseline values), analysis of covariance (ANCOVA) was applied to identify any differences between the groups after the intervention, adjusting for baseline values and covariates.
3. Results
Two patients in the Gln group and 1 patient in the placebo group were lost to follow-up due to non-compliance and withdrawal, respectively. The Gln and placebo groups were homogenous regarding the economic status, gender, place of residence, fever at the beginning, dyspnea age, height, weight, BMI, and appetite (P > 0.05; Table 1). However, the study groups had a significant difference in days of cough (2.45 [0.76] vs 1.8 [0.61]; P = 0.005).
| Variables | Gln Group (n = 20) | Placebo Group (n = 21) | Statistical Indicators |
|---|---|---|---|
| Economic status b | 0.629 | ||
| Equal income and expense | 8 (40.0) | 6 (30.0) | |
| Income more than expense | 7 (35.0) | 10 (50.0) | |
| Income less than expense | 5 (25.0) | 4 (20.0) | |
| Gender b | 0.501 | ||
| Girl | 15 (75.0) | 12 (60.0) | |
| Boy | 5 (25.0) | 8 (40.0) | |
| Fever at the beginning b | 0.366 | ||
| Yes | 15 (75.0) | 13 (65.0) | |
| No | 5 (25.0) | 7 (35.0) | |
| Dyspnea b | 0.698 | ||
| Yes | 18 (90.0) | 18 (90.0) | |
| No | 2 (10.0) | 2 (10.0) | |
| Age c | 4 (2.25 - 4.75) | 3.75 (2.25 - 4) | 0. 741 |
| Weight (kg) d | 15.36 (3.12) | 15.15 (2.63) | 0.817 |
| Height (cm) c | 98.2 (7.61) | 98.88 (6.96) | 0.796 |
| BMI (kg/m2) c | 15.49 (2.01) | 15.23 (1.74) | 0.660 |
| Duration of cough (day) d | 2.50 (2 - 3) | 2 (1 - 2) | 0.005 |
| Appetite c | 23.2 (9.09) | 18.9 (5.95) | 0.086 |
Abbreviations: Gln, glutamine; BMI, body mass index.
a Independent t test or Mann-Whitney U test for numeric variables and Pearson chi-square test for categorical variables.
b Values are expressed as No. (%).
c Values are expressed as mean (SD).
d Values are expressed as Median (percentiles)
As presented in Table 2, there were no significant differences between the Gln and placebo groups in terms of biochemical measurements (P > 0.05) at the baseline, except for hs-CRP (P = 0.004). At the end of the intervention, the serum values of IL-1β, TNF-α, and hs-CRP significantly decreased in the Gln group (P < 0.05). In the placebo group, there were no significant changes in serum levels of biochemical variables at the end of the trial compared to baseline (P < 0.05). The difference in hs-CRP between the Gln and placebo groups after 5 days of intervention was significant (analyzed by ANCOVA after adjusting for the duration of cough and biochemical baseline values). Moreover, at the end of the study, there was no significant difference regarding the duration of hospitalization between the Gln and placebo groups (3.25 [1.37] vs 3.35 [0.8], respectively; P = 0.70).
| Variable | Gln Group (n = 20) | Placebo Group (n = 21) | P Value |
|---|---|---|---|
| IL-1β (mIU/mL) a | |||
| Before | 2917.18 (2403.70) | 4206.25 (2989.75) | 0.183 b |
| After | 2000.11 (1493.45) | 2934.29 (2190.28) | 0.391c |
| MD, pd | -917.05, 0.028 e | -1271.96, 0.117 | |
| TNF-α (ng/L) f | |||
| Before | 433.2 (231.10 - 924.65) | 358.1 (152.3 - 873.52) | 0.277b |
| After | 216.8 (143.60 - 503.52) | 219.20 (175.77 - 649.50) | 0.798c |
| MD, pd | -239.22, 0.002 e | -110.17, 0.167 | |
| hs-CRP (mg/L) | |||
| Before | 18.37 (7.02) | 11.53 (7.11) | 0.004b,e |
| After | 10.67 (7.77) | 14.04 (6.57) | 0.005c,e |
| MD, pd | -7.69, < 0.001 e | -2.51, 0.341 | |
| MDA (nmol/mL) | |||
| Before | 2.08 (0.70) | 1.99 (0.63) | 0.779b |
| After | 1.84 (0.35) | 1.81(0.25) | 0.930c |
| MD, pd | -0.24, 0.136 | -0.122, 0.465 | |
| TAC (nmol/mL) f | |||
| Before | 0.85 (0.74 - 1.07) | 0.84 (0.73 - 1.17) | 0.373b |
| After | 0.86 (0.74 - 1.10) | 1.1 (0.96 - 1.37) | 0.100c |
| MD, pd | 0.06, 0.395 | 0.185, 0.769 |
Abbreviations: IL-1β, interleukin 1 beta; TNF-α, tumor necrosis factor α; hs-CRP, high-sensitivity C-reactive protein; TAC, total antioxidant capacity; MD, mean difference; MDA, malondialdehyde.
a Values are expressed as Mean (SD).
b Independent t test or Mann-Whitney U test.
c Analysis of covariance (adjusted for duration of cough and biochemical baseline values).
dPaired t test or Wilcoxon signed-rank test.
e P values of statistical significance (P < 0.05).
f Values are expressed as median (percentiles).
4. Discussion
According to the best of our knowledge, no study has examined the effects of Gln supplementation, along with medical therapy, in hospitalized children with ARI. It was shown that in the intervention group, hs-CRP was significantly reduced compared to the placebo group. Higher hs-CRP levels were significantly associated with poor respiratory function in children (32). In ARI, periodic assessment of CRP can instruct the true recovery or deteriorating phases of infection independently from viable signs and symptoms (33). In a recent study, reduced immunoglobulin A and CD4+ CD25+ T cells percentage, as well as elevated hs-CRP, IL-10, and procalcitonin, were related to pneumonia in children with COVID-19 (34). Abdollahi et al. concluded that hs-CRP measurement could be effective in the prediction of early neonatal sepsis (35). Moreover, according to the findings of studies by Xia et al. (36) and Lu et al. (37), the detection of hs-CRP or CRP is helpful for differential diagnosis and estimating the therapeutic effect of ARI in children. Since the elevation of hs-CRP and other inflammatory biomarkers is related to pneumonia in children, a decrease of inflammatory biomarkers through safe supplements such as Gln, along with medicine, can help to cure ARI and reduce infectious morbidity rates.
In the randomized controlled trial by Cai et al., Gln supplementation and recombinant human growth hormone for 14 days induced a significant reduction in CRP in critically ill elderly patients (38). In another study, parenteral Gln decreased CRP levels and infectious morbidity rates in patients with severe acute pancreatitis (39). Moreover, the combination of normal saline, hydroxyethyl starch, and Gln in severe acute pancreatitis resulted in a reduction in serum TNF-α, IL-8, and CRP concentrations (40). In the study by Wischmeyer et al. parenteral Gln administration in severe burn patients for 14 days reduced CRP in the Gln group, although this reduction was not statistically significant compared with the control group (41). We failed to show any significant reduction in other inflammatory indices compared to the placebo group. However, in the study by Ameho et al., Gln contributed to reducing the concentrations of the pro-inflammatory biomarkers, including IL-8 and TNF-α, in inflamed colonic tissues, leading to disease amelioration in experimental trinitrobenzene sulfuric acid-induced colitis in rats (42). Various studies have revealed that nutrition support supplemented with specific immunonutrients such as Gln may improve intestinal integrity and modulate acute phase responses (43-45), but the precise mechanism of Gln’s protection is unknown.
In the present study, the Gln administration did not change MDA and TAC levels. In line with our findings, enteral Gln administration of 45 g/d for 5 days did not change serum levels of MDA compared to the control group in patients with peritonitis or abdominal trauma (24). Seven days of Gln supplementation (0.15 g/kg) did not improve oxidative stress indices (MDA and TAC) in young, healthy men (46). Gln supplementation for 5 days increased the levels of 70-kd heat-shock protein as an oxidative stress marker and did not affect IL-10 and IL-6 levels in critically ill children (26). We presume that the differences between the conducted studies may be related to various patient groups, doses, and durations of Gln administration. The strengths of our study were the use of a placebo, regular follow-up of participants for supplement consumption, and evaluation of the effect of Gln on inflammatory and oxidative stress biomarkers levels in ARI for the first time. Limitations of this trial were lack of nutritional intake evaluation, short duration of Gln supplementation, and small sample size.
4.1. Conclusions
The effect of Gln supplementation on the reduction of hs-CRP in children with ARI was demonstrated in this study. Further studies are needed to determine the exact effects of Gln on inflammatory and oxidative stress biomarkers in children with ARI.